-
Call Now
1800-102-2727
ISOTOPES-DEFINITION, TYPES, OCCURRENCE, APPLICATIONS, FAQS AND PRACTICE PROBLEMS
We look at the night sky and see spots of brilliance amid the vast expanse of darkness. It is known that the night sky has been historically and eternally fascinating to human beings. It has evoked many questions, often probing the depths of the universe’s secrets. It also begs the knowledge of time and space.
We know that the earth is very old. How old you ask? Well, it is 4.543 billion years old if we’re being nitpicky. Earth’s closest neighbour is the moon, no matter where you look from. The moon is almost at a similar age, some 85 million years younger than the earth. However, the scientific community is still debating the birth of our moon. How did it happen? When exactly did it happen? And more importantly, where did it come from? Some theories suggest that the moon is actually a part of the earth. Almost sounds like a conspiracy theory, doesn’t it? As bizarre as it may seem, this idea stems from the geochemical similarities between the two bodies. Let me make it simpler for you. The isotopes of common elements that we see in our surroundings like carbon and silicon are the same on the moon too. This is a huge deal because the stability of a certain nucleus is fine-tuned to the pressure and atmospheric conditions (or maybe a lack thereof).
Today let us try to dive deeper into the idea of nuclei and isotopes as we know it and see where the quest takes us!
TABLE OF CONTENTS
- Definition and Notation
- Radioactive and stable isotopes
- Occurrence in nature
- Applications
- Practice Problems
- FAQs
DEFINITION AND NOTATION
Isotopes are nuclei that have the same atomic number (number of protons) but different mass numbers (number of nucleons). For instance, Carbon-12 and Carbon-14. Both these nuclei have the same number of protons i.e., 6 but their mass number is different. This essentially means that their number of neutrons is different.
It gets cumbersome to have to write the full name of the element every time we need to refer to its isotope so we have a notation in place.
An atom is generally denoted by:
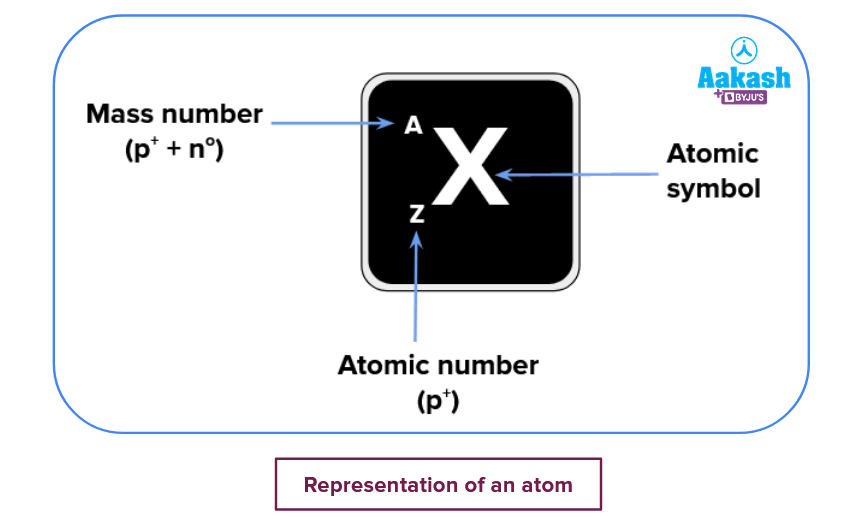
So we can replace A, X and Z with the respective mass number, atomic symbol and atomic number for a species like carbon-12, we get 612C. For carbon-14, we can write it as614C.
For the sake of understanding, let’s take another famous set of isotopes.
- Hydrogen-1 i.e., Protium has one proton and no neutrons. It can be denoted as11H.
- Hydrogen-2 i.e. Deuterium has one proton and one neutron. It can be denoted as 12H.
- Hydrogen-3 i.e. Tritium has one proton and two neutrons. It can be denoted as13H.
RADIOACTIVE AND STABLE ISOTOPES
“Radioactive” is a word that is thrown around without much thought but it can have disastrous implications for scientists. It is an important characteristic property exhibited by some nuclei which entail decay and emission of radiation. Radioactivity is generally a good indication of instability in a nucleus and hence the isotope.
While some isotopes, known as radioisotopes, have been observed to decay radioactively, other isotopes, known as stable isotopes, have never been observed to do so. For instance, while612C and613C are stable isotopes of carbon,614C is a radioactive version of carbon. On Earth, there are 339 naturally occurring nuclides, 286 of which are primordial nuclides, which means they have been around since the Solar System's creation.
According to theory, many supposedly "stable" isotopes and nuclides are inherently radioactive and seem to have extraordinarily long half-lives (discounting the possibility of proton decay, which would make all nuclides ultimately unstable). The term "observationally stable" refers to isotopes that are generally known to undergo any of the multiple forms of decay. There are 31 known radioisotopes with half-lives that are longer than the age of the universe, and the expected half-lives for these nuclei frequently far exceed the estimated age of the universe!
Did you know there are currently 3,339 radioactive isotopes known to exist, not counting those produced intentionally? However, only 905 of these nuclei are stable or have half-lives of more than 60 minutes. In fact, a commonly used nuclide-Technetium-99, which has a lot of medical usages, is known to be useful because of its radioactivity.
OCCURRENCE IN NATURE
There are 2 kinds of elements present:
- Mononuclidic elements: These are those elements that only contain one isotope. Eg: Vanadium, Iridium, Rubidium.
- Polynuclidic elements: These are those elements that include multiple naturally occurring isotopes. Eg: Oxygen, Radon, Calcium
More than one stable isotope of any stable element can be found naturally on Earth. Tin has the most stable number of isotopes which is ten.
Fast facts:
- About 94 elements are naturally occurring on Earth, however, some, like plutonium-244, are only found in very small quantities. Only 252 naturally occurring isotopes are stable, that is their decomposition has never been seen as of yet.
- There are 286 known primordial nuclides in total, 34 of which are radioactive and have half-lives of more than 100 million years, which existed before the Solar System was formed.
- By the use of nuclear reactors and particle accelerators, an extra 3000 radioactive isotopes that are not present in nature have been produced.
- By using spectroscopic analysis, it has also been possible to observe numerous short-lived nuclides that are naturally produced in stars and supernovae but are not typically present on Earth. Example: aluminium-26, which is abundant on an astronomical scale but is not naturally found on Earth.
APPLICATIONS OF ISOTOPES
A lot of properties and applications of elements are contingent on the properties of different isotopes of a given element. Let us explore some of them.
Purifying the isotopes: A key technological problem is isotope separation, especially for heavy materials like uranium or plutonium. Lithium, carbon, nitrogen, and oxygen are lighter elements that are frequently separated from one another by the process of gas diffusion of compounds, like CO and NO. The Girdler sulphide technique, for instance, uses chemical qualities rather than physical ones to separate hydrogen and deuterium, which is unique. Gas diffusion, gas centrifugation, laser ionisation separation, and a particular form of production mass spectrometry (during the Manhattan Project) have all been used to separate uranium isotopes in bulk.
Isotope analysis: The identification of an element's isotopic signature, or the relative abundances of its isotopes in a given sample, is done by isotope analysis. Isotope ratio mass spectrometry is a common method for isotope analysis. It is possible for C, N, and O isotopes to vary significantly in biogenic compounds in particular. Numerous uses for the analysis of such differences include determining a product's geographic origins using isoscapes or the identification of adulteration in food goods. The isotopic signature of the trace gases found in some meteorites allows us to identify them as having come from Mars.
Isotopic substitution: It is a method that can be used to identify the mechanism of a reaction by applying the kinetic isotopic effect.
Isotope labelling: Isotopic labelling, the use of uncommon isotopes in chemical reactions as tracers or markers, is another typical application. A given element's atoms are typically indistinguishable from one another. However, employing isotopes of various masses, mass or infrared spectroscopy may differentiate even various stable isotopes that are not radioactive. For instance, stable isotopes are employed to measure proteins in "stable isotope labelling using amino acids in cell culture" which is called the SILAC method. Radiation emissions can be used to identify radioactive isotopes if they are present.
Isotope dilution: Using the isotope dilution method, which involves mixing known quantities of isotopically substituted compounds with the samples and determining the isotopic signatures of the resulting mixtures using mass spectrometry, isotopes are frequently used to measure the concentration of different elements or substances.
Nuclear applications: Nuclear power as an unconventional energy source is gaining popularity in a lot of developed nations. Nuclear weapons are an important tool in war and are capable of wiping out entire civilisations in a matter of minutes when used at scale. However, more commonplace examples include oncological radiation and chemotherapy.
PRACTICE PROBLEMS
Question: Are Sulphur-40 and Chlorine-40 isotopes?
Answer: No, isotopes are species which have the same atomic number and different mass numbers. In the case of the above example, Sulphur has atomic number 16 and Chlorine has atomic number 17 so they are not isotopes. However, they are called isobars as they have the same mass number but different atomic numbers.
Question: What are some mononuclidic elements?
Answer: An element that is essentially found naturally on Earth as a single nuclide is referred to as a mononuclidic element or monotopic element.
The atomic mass of this particular nuclide will be distinctive.
Thus, one isotope that is stable or has a very long half-life makes up the majority of the element's natural isotopic abundance. Examples include Beryllium, Fluorine, Sodium and Aluminium.
Question: How many nucleons are present in CO?
A. 28
B. 26
C. 14
D. 8
Answer: (A)
Solution: Since nothing has been mentioned about the mass number, let us assume that we are dealing with Carbon-12 and Oxygen-16. Hence we can say that the number of nucleons present = Mass number of C + Mass number of O = 12 + 16 = 28
FREQUENTLY ASKED QUESTIONS-FAQs
Question: What is radiotherapy?
Answer: Radiation therapy, often known as radiotherapy, is a treatment that uses ionising radiation to control or destroy cancerous cells. It is typically administered as part of cancer treatment and is typically delivered via a linear accelerator. If cancer is restricted to a single part of the body, radiation therapy may be curative. It can also be used as adjuvant therapy to stop tumour recurrence following primary malignant tumour removal surgery.
Question: How do we make nuclear bombs?
Answer: Chemically, the most important part of a nuclear weapon deployment is the enrichment of fissile material. Choosing this nuclear material carefully is important, as one would want the maximum release of energy. Generally, U-235 and Pu-239 are chosen as fissile materials. There are various mechanisms to initiate the nuclear reaction and thereby propagate the chain reaction which leads to the release of massive amounts of energy.
Question: What are the isotopes of uranium?
Answer: Uranium is known to have three naturally occurring isotopes. They have mass numbers 234, 235 and 238. It is noteworthy that Uranium-238 is about 99% abundant in the Earth’s crust. Isotopes with mass numbers ranging from 214 to 242 (barring 220 and 241) have also been synthesised for uranium.
Question: Is Pu-239 a stable isotope?
Answer: Plutonium-239 is actually a radioactive element with a half-life greater than 24,000 years. However, it is important to know that it is one of the three active fissile agents in nuclear bombs. This is because of its highly exothermic nuclear fission, as well as, an industrial convenience of synthesis of the same.
Question: Are primordial isotopes rare?
Answer: Not at all! Because of their very high stability, primordial isotopes have indeed been around before the Earth. Some relatable examples include Calcium-48, Vanadium-50, Zirconium-90 and Barium-130.


