-
Call Now
1800-102-2727
Isomerism in Coordination Compounds - Structural Isomerism- Ionization Isomerism, Hydrate Isomerism, Coordination Isomerism, Linkage Isomerism, Stereo Isomerism. Geometrical Isomerism, Practice Problems and FAQs
You might have heard about twins, but it is said that there will be six people who are almost physically similar in the world. Some chemical substances also are similar in chemical formula but different in properties and are called isomers. Coordination compounds exhibit varieties of such isomerism.
The coordination compounds [Co(NH3)6][Cr(CN)6] and [Cr(NH3)6][Co(CN)6] can present in many different combinations or isomers. Can you guess the number?
Can you tell me how geometrical isomerism can exist in coordination compounds? Let’s brush up on the concepts with a quick revision and a fine answer to such questions.
Table of Contents
- Isomerism in coordination compounds
- Structural isomerism
- Ionization Isomerism
- Hydrate isomerism
- Coordination isomerism
- Linkage isomers
- Stereoisomers
- Geometrical isomerism
- Practice Problem
- Frequently Asked Questions
Isomerism in coordination compounds
Isomers are two or more compounds that have the same molecular formula but different atom arrangements. Isomers are two or more compounds that have different atomic arrangements but the same molecular formula.This phenomenon is known as isomerism. Isomers differ in one or more physical or chemical properties Isomers differ in chemical and physical properties due to differences in atom arrangement.
Isomers can be divided into two broad categories:
- Structural isomers
- Stereoisomers
Structural isomerism
Structural isomers are isomers that have the same molecular formula but a different structural arrangement of atoms or groups of atoms around the central metal ion different structural arrangement of group of atoms or atoms bonded with the central metal ion but have a same molecular formula. It is further subdivided into four categories.
- Ionization isomerism
- Hydrate isomerism
- Coordination isomerism
- Linkage isomerism
1. Ionization Isomerism
Ionization isomers are compounds that have the same molecular formula but produce different ions in solution. Ionization isomers are compounds that produce different ions in the solution but have the same molecular formula. The difference in this type of isomerism results from the interchange of groups within or outside the coordination entity due to the exchange of groups outside or within the coordination sphere.
As an example:
(i) and
(ii) and
- Hydrate isomerism
Solvate isomers are compounds that have the same molecular formula but differ in whether or not a solvent molecule is directly bonded to the metal ion. Solvate isomers are compounds that differ in bonding between the solvent molecule and the metal ion, but have the same molecular formula.
It is also referred to as hydrate isomerism when water is used as a solvent.
Water molecules can exist both inside and outside the coordination sphere as a coordinated group or as water of hydration outside and within the coordination sphere as a coordinated group or as water of hydration in this type of isomerism.
Example
(i) and
]
(ii) and
]
- Coordination isomerism
Isomerism of this type occurs in compounds that contain both cationic and anionic entities, and the isomers differ in the distribution of ligands in the coordination entity of cationic and anionic parts both anionic and cationic entities, and isomers are there due to different distribution of ligands in the coordination sphere of anionic and cationic parts of coordination compound. The exchange of ligands between cationic and anionic entities of different metal ions present in the complex causes this type of isomerism.
The following are some examples:
(i) and
(ii) and
- Linkage isomers
Linkage isomers are compounds that have the same molecular formula but differ in the mode of attachment of a ligand to a metal atom or ion have the same molecular formula but differs in the way the ligand is attached to the central metal atom. That kind of ligands are known as Ambidentate ligands.
As an example: ONO nitrito-O, NO2 nitrito-N
The nitrogen and oxygen atoms in NO2 ion can both donate their lone pairs. One compound will be formed if nitrogen donates its lone pair. When oxygen donates its lone pair, a different compound (with the same molecular formula) is formed.
Examples of ligands are:
-CN Cyano (through C)
-NC Isocyano (through N)
-SCN Thiocyanato (through S)
-NCS Isothiocyanato (through N)
Stereoisomers
Stereoisomers are isomers that have the same atomic or group positions but differ in their spatial arrangements around the central atom. There are two types of isomerism: geometrical isomerism and optical isomerism.
Geometrical isomerism
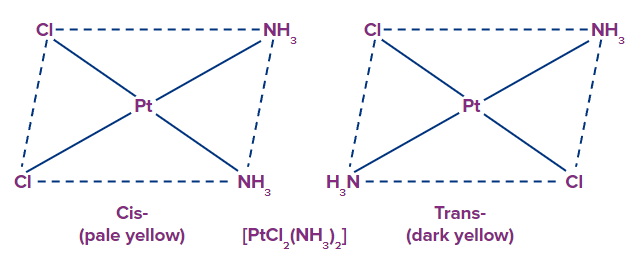
Geometrical isomerism occurs because ligands occupy different positions around the central ion different positions around the central metal atom in heteroleptic complexes,. These are known as cis-form (ligands occupy adjacent positions adjacent positions are occupied by ligands) and trans-form (ligands occupy opposite positions) (ligands occupy opposite positions opposite positions are occupied by ligands). This isomerism is also known as cis-trans isomerism.
(a) Geometric isomerism in coordination number 4 complexes
Some Important points
- Tetrahedral or square planar geometry can be used for these complexes.
- In tetrahedral complexes, geometrical isomerism is not possible.
- Square planar complexes, on the other hand, exhibit geometrical isomerism.
- Geometrical isomerism cannot occur in complexes of type MA4, MA3B, or MAB3 In complexes of type MA4, MA3B, or MAB3, geometrical isomerism is not possible because all possible spatial arrangements for any of these complexes because all the possible arrangements of these complexes are identical.
Example: [Pt(NH3)2(Cl)2]
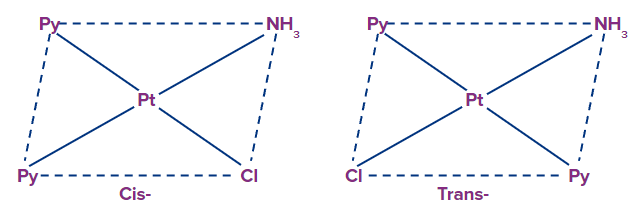
Example: [Pt(NH3)(py)2Cl]
Example: [Pt(NH3)(py)BrCl]
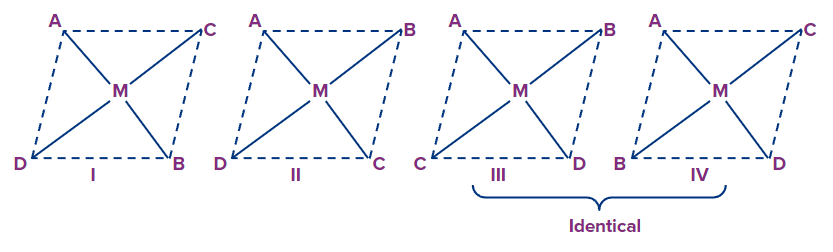
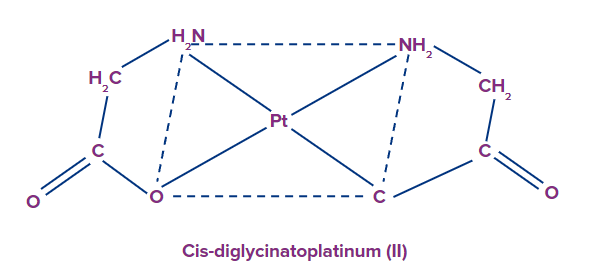
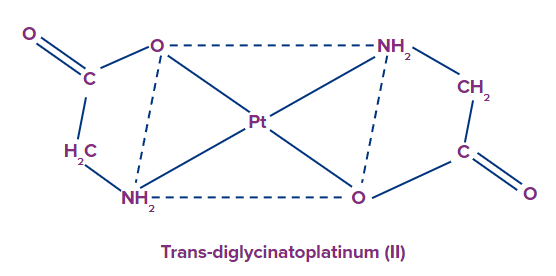
Example: [Pt(gly)2]
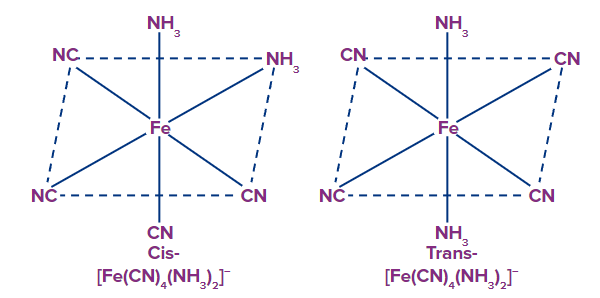
(b) Geometric isomerism in coordination number 6 complexes
Example: [Fe(CN)4(NH3)2]-, they exist as cis trans isomers.
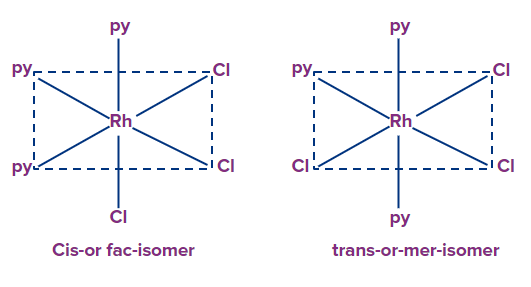
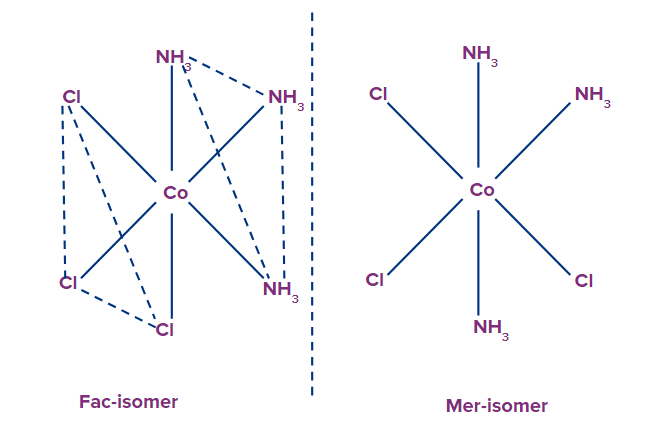
Example: [Rh(Cl)3(py)3], They exist as fac-mer isomers
Meridional and mer isomers: The isomer is called meridional or mer isomer when the three ligands are on the same equatorial plane of the octahedron, when three ligands of the octahedron are on same equatorial plane, i.e., around the octahedron's meridian.
Facial or Fac isomers: The three ligands in the facial isomer are located at the three corners of a triangular face, whereas the three ligands in the meridional isomer are located at the three corners of a square plane.
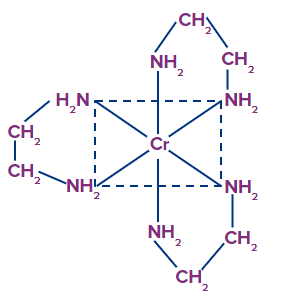
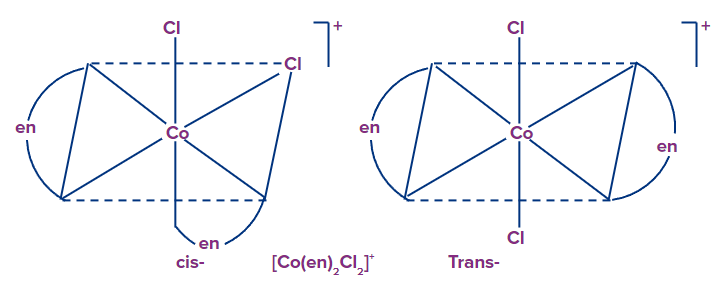
Example: [Co(en)2(Cl)2]+, they exist as cis trans isomers.
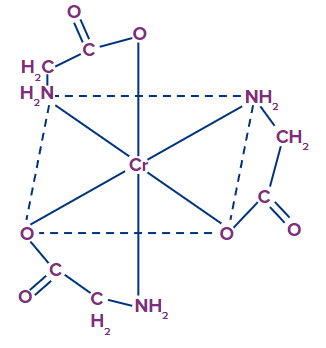
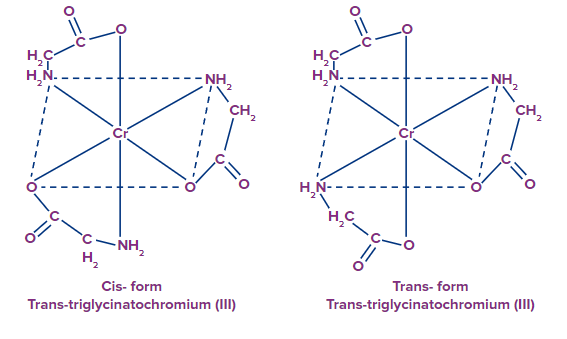
Example: [Cr(gly)3], they exist as cis trans isomers.
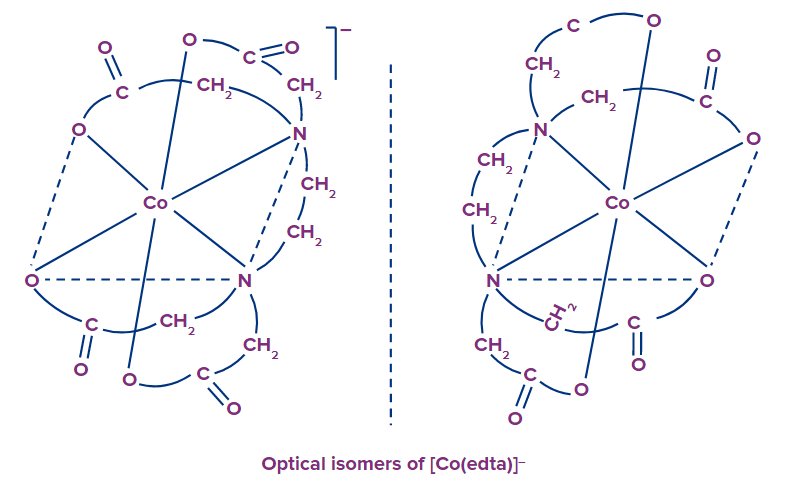
Optical Isomerism
Optically active isomers are those that rotate the plane of polarized light equally but in opposite directions.
The d and l isomers are non-superimposable mirror images of each other, just as the left and right hands are mirror images of each other, and lack the plane of symmetry.
These optical isomers also have the chirality property. The absence of a plane of symmetry in the structure of a substance is required for it to exhibit optical activity.
Example: [Co(en)3]3+
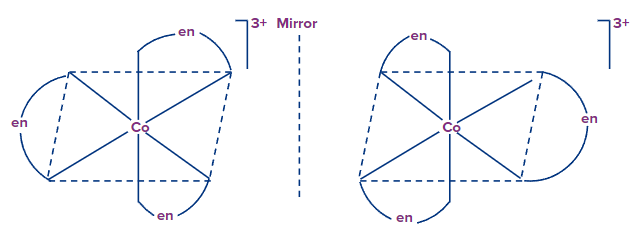
Example: [Co(edta]-
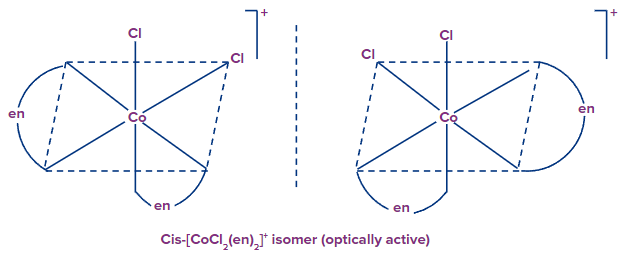
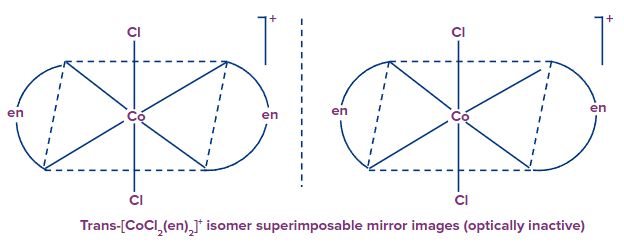
Example: [Co(en)2(Cl)2]+
The trans form lacks optical isomerism, which means it cannot be resolved into optical isomers. Because the molecule has a plane of symmetry. The cis-isomer, on the other hand, is unsymmetrical and can be resolved into optical isomers. The cis-isomer, on the contrary, is unsymmetrical and can be resolved into the individual optical isomers
Only the cis-isomers are optically active.
Practice Problem
- The compound with the formula Co(NH3)5BrSO4 has two isomers: one that is red-violet and forms a precipitate with BaCl2, and the other that is red and forms an AgBr precipitate with silver nitrate. Can you predict the compound's structure as well as its kind of isomerism?
Solution: The compound with the formula Co(NH3)5BrSO4 has two isomers.
(a) One of these, red-violet in color, forms a precipitate with BaCl2. It denotes the presence of an ionisable sulfate ion outside of the coordination entity. Hence, the compound should be [Co(NH3)5Br]SO4
(b) The second is red and does not form a precipitate with BaCl2, but does form an AgBr precipitate with silver nitrate. It denotes the presence of ionisable bromine ions outside the coordination entity. Hence, the compound should be [Co(NH3)5SO4]Br.
[Co(NH3)5Br]SO4 and [Co(NH3)5SO4]Br shows Ionization isomerism as when interchange of groups within or outside the coordination entity happens, it comes under Ionization isomerism.
- When compound A, B and C with molecular formula CrCl3(H2O)6 are treated with conc. H2SO4compound A does not lose water, B loses one water molecule and C loses two water molecules. When compound A, B and C are treated with AgNO3, three chloride, two chloride ions and one chloride ions are precipitated respectively Predict A, B and C.
,
]
,
]
,
,
,
]
,
]
,
,
Solution: When compound A is treated with conc. H2SO4, it does not lose water, and three chloride ions are precipitated with AgNO3. Hence, three ionisable chloride ions are present. The compound A should be [Cr(H2O)6]Cl3
When compound B is treated with conc. H2SO4, it loses one water molecule, and two chloride ions are precipitated with AgNO3Hence, two ionisable chloride ions are present. The compound B should be [Cr(H2O)5Cl]Cl2.H2O
When compound C is treated with conc. H2SO4 dark green, it loses two water molecules and one chloride ion is precipitated with AgNO3Hence, one ionisable chloride ion is present. The compound B should be [Cr(H2O)5Cl2]Cl.(H2O)2
So, the correct answer is option (B).
- Which of the following isomerism is exhibited by the given pair [Pt(NH3)4] [PtCl4] and [PtCl(NH3)3] [PtCl3.NH3]
- Ionization isomerism
- Hydrate isomerism
- Coordination isomerism
- Linkage isomerism
Solution: Isomerism of this type occurs in compounds that contain both cationic and anionic entities, and the isomers differ in the distribution of ligands in the coordination entity of cationic and anionic parts. The exchange of ligands Cl- and NH3 between cationic and anionic entities of different metal ions present in the complex causes coordination isomerism.
Hence, the correct answer is option (C).
4. Which of the following complexe can show fac-mer isomerism?
Solution: Fac is an abbreviation for facial, and mer is an abbreviation for meridional. The same ligands are on a common face of a polyhedral complex in a fac-isomer, but on a plane that bisects the polyhedron in the mer isomer.
At Least three unidentate must be the same to show this type of isomerism. So, Correct option should be (B), as it contains only three same unidentate ligands.
Frequently Asked Questions
- Why do tetrahedral molecules lack geometrical isomerism?
Because all of the bond angles in the tetrahedral molecule are the same 109.5, geometrical isomerism requires at least one bond angle to be different; otherwise, orientation will not differ to show isomerism.
- Why Square planar complexes generally do not show optical isomerism?
This is because all the 4 ligands and metal cations exist in the same plane and there is plane of symmetry present in every molecule which make it optically inactive.
- Is there any square planar complex which is optically active?
Every statement in chemistry is not true. So, there are some square planar complexes which are optically active due to their structural representation. For example: [Pd(EDTA)]2-
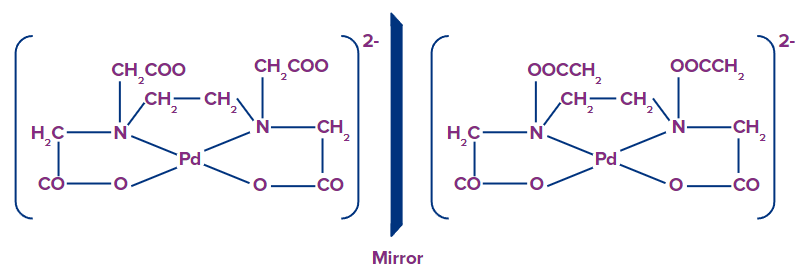
- Why [Cr(gly)3] exist in cis trans isomerism whereas [Cr(en)3]3+ does not?
In [Cr(gly)3] , there are two different atoms involved in the structure of ligand which can create cis trans isomersim whereas in [Cr(en)3]3+, same Nitrogen is present on each end which exclude the possibility of geometrical isomerism.