-
Call Now
1800-102-2727
Irreversible Reactions- Definitions, Examples, Practice Problems and FAQs
Changes are unpredictable phenomena that might be for the better or worse, depending on your point of view. Every day, we observe several things in our environment changing, such as ice melting, food cooking, baking, and crafting. Some changes occur naturally and quickly, while others are slow. You may receive the original stuff back in some circumstances, but sometimes you may not. Have you ever considered the possibility of reversing the process that turns milk into cheese if we can manufacture cheese from milk? We all know, it is not possible. Right???
So, this kind of process is known as an irreversible process. There are many more examples of this kind, particularly chemical changes that result in new compounds and can not be reversed into their original state. Let's talk about the science that underlies these types of processes and reactions.
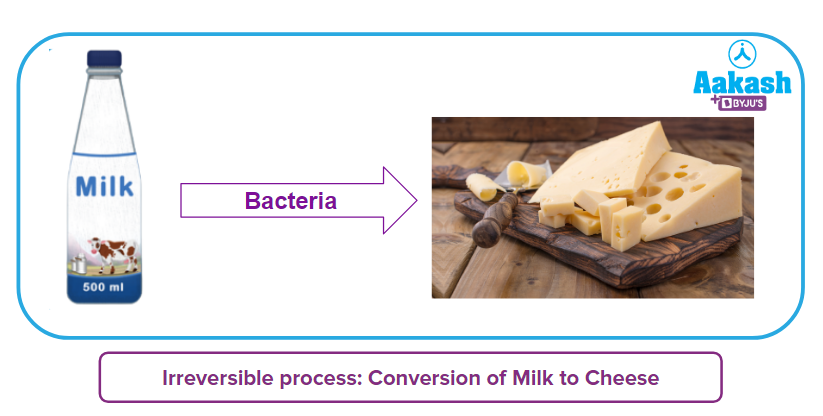
Table of content
- Irreversible reaction
- Types of Irreversible reactions
- Practice Problems
- Frequently Asked Questions
Irreversible reaction
Chemical reactions occur where reactants are converted to generate products.
Irreversible reactions are unidirectional processes in which the reactants transform into products completely.
Always remember that a reversible reaction is bidirectional, meaning that it can proceed forward as well as backward, whereas an irreversible reaction is unidirectional and proceeds only forward, consuming the entire amount of reactants. Changes that are irreversible are permanent.
Key points
- A compound created by the reaction of one or more reactants cannot be converted back into the original reactant.
- A one-step reaction that is irreversible is depicted by a single arrow.
- In irreversible reactions, equilibrium cannot be achieved.
- Reactions that are irreversible undergo completion.
- Heating, burning, mixing, and powdering are a few processes that lead to irreversible changes. Cooked eggs that can't be turned back to their original state are frequently encountered cases of cooking. Ash created by the burning of paper or other items is another illustration.
Conditions of Irreversible reactions
- Reactions involving gaseous products occurring in open system under atmosphere
pressure conditions are always irreversible.
All natural processes are irreversible.
- Removal of products by way of precipitates or (immiscible liquids) or solids facilitate the
irreversible reactions
- All exothermic reactions will be irreversible.
Types of Irreversible reactions
- Precipitation reaction:
- Combustion reaction:
Practice Problems
1. When ethanol is ignited, two different gasses combine. What inference could be drawn from this observation?
a. The reaction is irreversible.
b. It's an exothermic reaction.
c. It's an endothermic reaction.
d. The reaction can be reversed.
Answer
When a material is ignited, it burns in the air. To put it another way, we assume it is combusting with the oxygen in the air. When ethanol is burned, two distinct gasses are produced, according to our sources. We can conclude that a reaction occurred because the two gasses differ from one another and from the two starting constituents (ethanol and oxygen).
Furthermore, because the original component, ethanol, was not collected at the end of the reaction, we can deduce that all of the reactants were converted into products and that products did not combine to produce ethanol.
We can deduce that the reaction is still ongoing. Because these are experimental observations, it is impossible to tell whether the reaction is exothermic or endothermic.
The correct answer is A, indicating that the reaction is irreversible.
2. Which of the following reactions is by its nature irreversible?
a.
b.
c.
d. H2O(l) ⇌H2O(g)
Answer: The reactants in a reversible process can be recovered from the byproducts. In order to depict reversible reactions, a two-sided half arrow is employed. Reversible reactions include
N2(g)+3H2(g) ⇌ 2NH3(g)
H2O(l) ⇌H2O(g)
The reactants that cannot be formed from the products in an irreversible reaction. In order to depict irreversible reactions, a one-side full arrow is employed. Irreversible reactions include
BaCl2(aq)+Na2SO4(aq)BaSO4(s)+NaCl(aq)
Hence, the correct answer is an option (A).
3. Which of the following process is not irreversible in nature?
a. Precipitation reaction
b. Baking a muffin
c. Wood burning at the campfire
d. Making sugar syrup
Solution: The correct answer is an option (D).
It is not possible to reverse a precipitation reaction because once a product gets separated from the ration, it is not possible to reverse that particular reaction. Similarly, when flour is used to make muffins, it is not possible to reverse it. When wood is burnt in a campfire, that is a combustion reaction which is an irreversible reaction. Hence, all three are irreversible reactions. Sugar syrup can be reversed into sugar and liquid by boilng which makes it a reversible process.
4. Which of these changes cannot be reversed?
a. ice melting
b. wax melting
c. water boiling
d. wax burning
Solution: Wax is burned during the burning process—neither the liquid wax nor the solid wax. When the wax is burned, light and heat are produced. The wax cannot be recovered after being burned. Wax burning is a chemical process, hence it cannot be stopped. Hence, this process cannot be reversed and a correct answer is an option (D).
Frequently Asked Questions
Q1. Is burning a candle a reversible process?
Answer: No, burning a candle is an irreversible process. See, melting wax is a reversible process but when wax comes close to a flame, it burns and turns into carbon dioxide. This carbon dioxide cannot be turned back into wax.
Q2. Why is the precipitation reaction known as an irreversible reaction?
Answer: A precipitation reaction is an irreversible process because once precipitate forms, there are insoluble solids present in the solution. Once precipitates leave the solution, you can not retrieve the reactant by heating the solution. These are permanent changes.
Q3. Compare reversible and irreversible reactions.
Answer: Reactants in a reversible reaction combine to create new products, and the original reactants can be recovered. On the other hand, it is impossible to return to the initial reactants in the case of irreversible reactions.
In reversible reactions, the changes happen gradually through a series of intermediary phases during which the system is in an equilibrium state, whereas in irreversible reactions, there is no such equilibrium state.
Q4. Please respond to the sentence "Every change has a cause."
Answer: Until and unless it is affected by an external element like heat, motion, wind, etc., a substance remains in either its physical condition or chemical state. As a result, every change has a cause.
Q5. How is burning a piece of paper an irreversible change?
Answer: When we burn paper or wood, we produce ash and smoke. We will never be able to make paper or wood from ash again. As a result, the change is irreversible.


