-
Call Now
1800-102-2727
Ionic mobility and transport number
Come the festive season, of Raksha Bandhan, Diwali, Eid, Christmas, we connect with our love ones and show our love and affection by sending them goodies through courier. Or you must have ordered pizza or something online of books, gadgets, or clothes.

Whenever we place an order on courier service, they give us a unique tracking number so that we can track our packages. Sometimes packages arrive before the expected date and sometimes it reaches us, way after the expected delivery date.
What are the factors that affect these delivery systems?
Well, there are a lot of factors such as public holidays, unexpected weather conditions, etc.
Similarly, during the electrolysis of an electrolyte, different ions move in a different manner. Certain ions have faster mobility than others. Several factors hinder the mobility of ions. It is vital to understand how different ions move in a different atmosphere for their application in the respective field.
Let’s understand the concept of ionic mobility and transport number.
Table of content
- Definition of Ionic mobility
- Factor affecting ionic mobility
- Relationship between ionic mobility and electrolytic conduction
- Definition of transport number
- Factors affecting transport number
- Relationship between transport number, electrolytic conductance and Ionic mobility
- Determination of cell constant
- Determination of electrolytic conductance
- Practice problems
- Frequently asked questions (FAQs)
Definition of Ionic mobility (μ )
Ionic mobility describes the speed of an ion in an electrolyte moving on passage of electricity current between two electrodes with a unit value of potential gradient. It is denoted by a symbol ‘ ‘
Speed
Speed is the distance travelled in the unit time.
Potential gradient
Potential gradient
Ionic mobility describes the distance travelled by an ion of an electrolyte in uint time with respect to the unit value of potential gradient.
Unit of ionic mobility
Speed must be in
Factor affecting ionic mobility
Mobility of ions are generally affected by the following factors
- Temperature.
- Concentration ions
- size of ion
- Charge of the ion
- Viscosity of the solvent
Temperature
We know that when the temperature is increased ions will gain kinetic energy and due to this their motion will be increased to a greater extent. In simple terms, we can say that greater the value of temperature, greater will be the kinetic energy of the ions and the faster will be the mobility of ions.
Concentration of ions.
Mobility of ions increases with dilution. Strong electrolytes dissociate completely and so exhibits higher conductance due to the larger number of ions. On dilution, mobility increase slightly due to more room for movement of ions.
Weak electrolytes are partially dissociated and posses smaller number of ions and hence exhibit lesser conductance due to smaller number of ions compared to strong electrolytes. But on dilution, ionization of weak electrolytes increases preleasing more ions. So, the number and mobility increases with dilution significantly higher than the stronger electrolytes.
Size of the ion
Size of the ion has an interesting impact on mobility of ions. Greater the size of ions, sluggish the ion to move and lesser will be the mobility of ions. Size of the ions and mobility of the ions are inversely proportional to each other.
Remember one thing that you have to check whether the ion is in gaseous or in hydrated state. As we know that order of size of hydrated ions is inversely proportional to the size of gaseous ion.
Charge of the ion:
Ions are solvated by the solvent molecules. Higher the charge of the ion, higher the number of oppositely charged ion around it, and there by increasing the size and reducing the mobility of ions..
Viscosity of solvent:
Ions shall be dragged behind by the viscous solvent and hence will so decreasing mobility with increasing viscosity of the solvent.
Relationship between ionic mobility and electrolytic conduction
When a voltage is applied to electrodes dipped in an electrolytic solution, the electrolyte's ions starts moving, causing electric current to flow through the solution. Conductance or conductivity refers to the ability of electrolytes to conduct electric current. Ohm's law also applies to electrolytic solutions.
Every substance offers resistance to the flow of electricity to a small or large extent. The law that gives the exact value of the resistance is known as Ohm's law. It states that
If to the ends of a conductor is applied a voltage 'E' and a current 'I' flows through it, then the
resistance 'R' of the conductor is E/I.
Voltage is often measured in volts, whereas current is typically measured in amperes. If one ampere current flows through a conductor when a voltage of one volt is applied to it, the resistance of the conductor is taken as I ohm .
Thus, according to Ohm's law,
V = IR
Where I =current flowing through the conductor
V = potential difference across the conductor
R = resistance offered by the conductor
Electrolytic conductance(
Mathematically,
Example: the correct order of the electrolytic conductance at infinite dilution for LiCl, NaCl and KCl. .
Solution: We have to remember that
Ionic mobility is inversely proportional to the size of the ion. The larger the size the of the ion, lesser will be the mobility of ions.
Given: LiCl, NaCl and KCl
Chloride ion is common in all three electrolytes. Hence, the mobility of the ion will depend on the cations of these electrolytes. That is size of Li, Na and K will determine its ionic mobility and thus its electrolytic conductance.
The order of ionic radius for hydrated atoms of Li, Na and K is
Li >Na >K
As all these elements belong to group 1 of the periodic table and we know that down the group ionic radius increases for gaseous atoms whereas it decreases for hydrated atoms. Hence, Li ion will be the largest among these three.
Now the order of ionic mobility will be in this order
LiCl< NaCl< KCl
As we know
Hence, the electrolytic conductance order of the above the electrolytes will be
Definition of transport number (t)
It is also called as ion transport number or transference number. Transport number is described as a fraction of current carried by cation or anion to that of total current passed through the electrolyte. It is represented by’ t’.
Mathematically,
We must remember that the sum of transport number of cation and transport number of anion is always 1.
mathematically,
ta + tc = 1
Factors affecting transport number
- Temperature
- Concentration of the electrolyte
- Hydration of ions
Temperature
A rise in temperature tends to bring the cation and anion transport numbers closer together.
Nature of the ions present in the solution
The speed of the cation affects the transport number of the anion, and vice versa. The transport number of the Cl -ion in NaCl is 0.0004, but it is 0.16 in HCl. This is due to the fact that H+ moves faster than Na+.
Hydrated of ions
In general, as an ion's degree of hydration decreases, its transport number rises.
Relationship between transport number, electrolytic conductance and Ionic mobility
Transport number of cation or anion can be related to electrolytic conductance as follows:
Similarly, transport number of cation or anion can be related ionic mobility in the following manner:
Determination of cell constant
It is represented by
Cell constant can be determined by the above three formulas but most commonly it is determined through either K/G or KR
Determination of electrolytic conductance
Electrolytic conductance can be measured from the Wheatstone bridge.
The conductivity of an electrolytic solution is determined by using a Wheatstone bridge arrangement in which one resistance is replaced by a conductivity cell filled with the electrolytic solution of unknown conductivity.
The apparatus used in this method are
Rheostat, resistance, conductivity cell, detector, and oscillator
Rheostat
A rheostat is a variable resistor that regulates current flow. They can change the resistance in a circuit without breaking it.
Detector
Here galvanometer is used to detect the small amount of current flowing through the circuit.
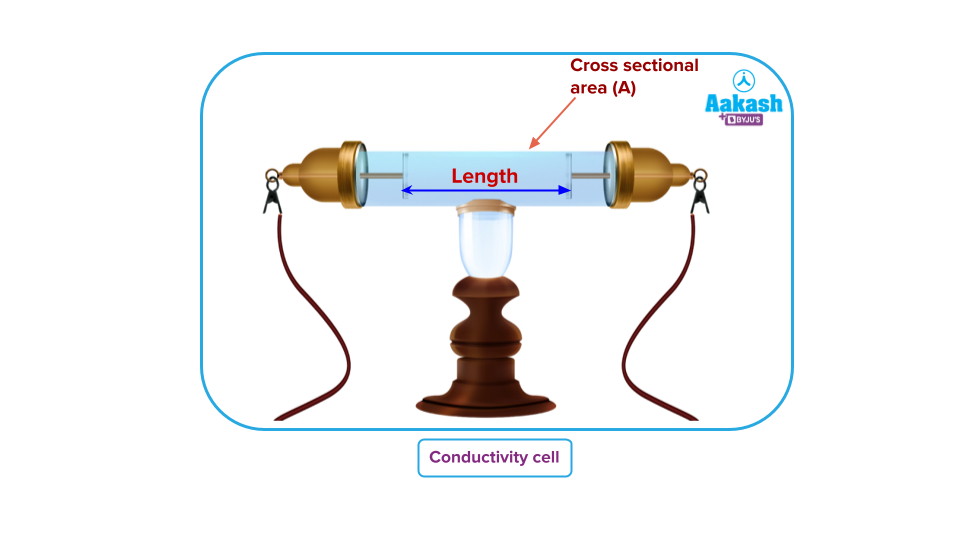
Conductivity cell
It is a device in which the conductivity of the electrolyte is unknown. The conductivity cell has two platinised electrodes which are connected to the terminals. These terminal ends are connected with the arms of wheatstone bridge.
When the detector detects a null point. At this point
Where R1= resistance of the rheostat
R2=resistance of the conductivity cell
R3=resistance of resistor in arm 3
R4=resistance of resistor in arm 4
From the above equation we can calculate the resistance of the conductivity cell and thus conductivity of the electrolyte can be calculated.
Practice problems
Q.1 Resistance of 0.1 M KCl solution in a conductance cell is 300 ohm and conductivity is 0.013 S cm-1. The value of cell constant is :
- 3.9 cm-1
- 39 m-1
- 3.9 m-1
- None of these.
Answer: (A)
Solution: most commonly cell constant is determined through either K/G or KR
Q.2 If the transport number of K+ is 0.225 (dilute solution of KCl in methanol), the transport number of H+ (dilute solution of HCI in methanol) is: (Given, Molar conductance of KCl in methanol = 98.6 ohm-1 cm2 mol-1 and Molar conductance of HCI in methanol = 182 ohm-1 cm2mol-1
- 0.38
- 0.58
- 0.78
- 0.98
Answer: (B)
Solution:
Transport number of a cation or anion can be determined by the following formulas
We know that
sum of transport number of cation and transport number of anion is always 1
ta + tc=1
Transport number of cation or anion can be related to electrolytic conductance as follows:
or
So,
From the above data now we can determine the molar conductivity of ions
Q.3 Resistance of a decimolar solution between two electrodes 0.02 meter apart and 0.0004 m2 in area was found to be 50 ohm. Specific conductance (κ) is
- 0.01 S m-1
- 1 S m-1
- 0.1 S m-1
- 2104 S m-1
Answer: (B)
Solution:
Given data : l=0.02 m and a=0.0004 m2
Q.4 What is the unit of ionic mobility?
- cm2 sec-1 volt-1
- cm2 sec-2 volt-1
- cm2 sec-1 volt-2
- cm2 sec-1 volt-1
Answer: (A)
Solution: Ionic mobility describes as the distance traveled by any ion of an electrolyte in uint time with respect to the unit value of potential gradient.
Unit of ionic mobility
Speed must be in cm Sec-1
Frequently asked questions (FAQs)
Q.1 In measuring conductance, why direct measurement of cell constant by is generally discarded?
Answer: We know that the value of cell constant can be determined by three formulas:
From , from KR and from . but formula is discarded due to the unreliability and an inconvenient in calculation after a certain use.
Q.2 Why do we use ac source instead of DC source during the determination of of electrolytic conductance of a conductivity cell?
Answer: when DC current is applied to this circuit the electrolyte in the conductivity cell start dissociation process and split into ions and the absolute conductivity of the respective electrolyte becomes difficult to determine. Hence, AC current is generally applied in the wheatstone bridge to calculate the value of conductivity of the electrolyte more precisely.
Q.3 Does potential difference across the electrolyte has any influence on the mobility of ions?
Answer: The ion movement is accelerated by increasing the current (due to increased Coulombic attractions). Because current is influenced by voltage, the rate of migration accelerates as the potential difference between the electrodes widens.


