-
Call Now
1800-102-2727
Iodometric Titrations - Introduction, Examples, Practice Problems and FAQ
Swimming is a great way to exercise and keep your body in shape. Consider what it would be like to swim in a filthy, trash-filled pool. Do you believe swimming in such a pool will be healthy?
Obviously not! Many microorganisms found in filthy pools can cause a variety of ailments.
Have you ever wondered how pools are routinely cleaned to maintain their hygienic conditions?
You are correct! The pool cleaners use chlorine or bleaching powder!
When bleaching powder, Ca(OCl)2, or any other chlorine-based chemicals come in contact with water, hypochlorous acid (HOCl), a sanitiser is generated. This sanitiser is used to disinfect swimming pools.
But how can you figure out how much bleaching powder to put in a specific swimming pool?
The answer to this question is a technique called ‘Iodometry’.
Iodometry is routinely employed to determine how much hypochlorite is present in bleach that is actually performing the bleaching activity. With this method, an excessive amount of iodide is added to a known volume of material; only the active (electrophilic) species are able to convert the excess iodide to iodine. The availability of chlorine can be determined using iodometry by measuring the iodine content. The chlorine released when diluted acids and hypochlorite combine is referred to as "available chlorine."
In general, this titration can be used to calculate the concentration of oxidising agents in any sample.

![]()
TABLE OF CONTENTS
- What is Titration?
- Iodine Titration
- Iodometric Titrations
- Practice Problems
- Frequently Asked Questions - FAQ
What is Titration?
Titrimetric analysis or titration entails calculating the volume of a solution with an accurately known concentration that must quantitatively react with the measured volume of the solution of a substance whose concentration is to be calculated. The standard solution is the name given to a solution whose concentration is precisely known. The volume of the standard solution used, the chemical equation, and the relative molecular masses of the interacting compounds are used to compute the mass of the substance dissolved in the solution of unknown concentration. Titrant refers to the reagent with a known concentration, while titrate refers to the material being titrated.
Standard solution is typically added from the lengthy graduated tube known as the burette to perform titrimetric analysis. Titration is the process of gradually introducing the standard solution into a solution of uncertain concentration until the reaction is barely complete. The equivalency point, also known as the theoretical or stoichiometric end point, is the point at which a reaction is complete.
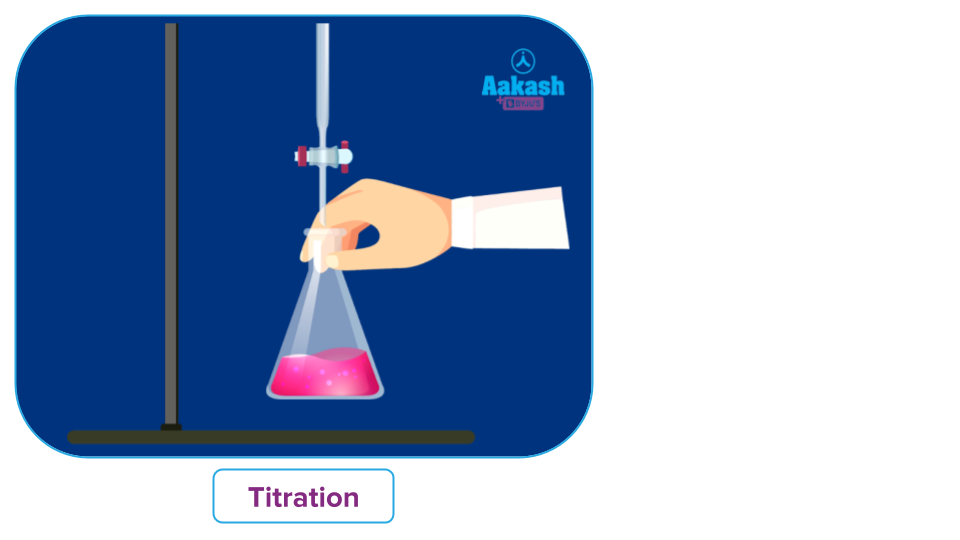
In simple terms, titration is an analytical technique used to determine the concentration of a known volume of an unknown solution (titrate) using a solution of known concentration (titrant).
Iodine Titration
This method of analysis involves the use of iodine/iodide in titration. There are two types of iodine titrations. They are the direct method, also known as ‘Iodimetry’, and the indirect method, also known as ‘Iodometry’.
In the direct method, an iodine solution is used to titrate the reducing agents, which can be quantitatively oxidised at the equivalence point. As iodine is a weak oxidising agent, only a few substances can be oxidised by iodine. Thiosulphate, one of the common reducing agents, must be determined iodimetrically.
Iodine (I2) is a mild oxidising agent.
(reduction)
In the indirect method, also known as ‘Iodometry’, the oxidising agent that has to be estimated is treated with an excess of iodide ions and the liberated iodine is estimated by titrating with a standard thiosulphate solution.
Iodide (I-) is a mild reducing agent.
(oxidation)
In this concept page article, we’ll discuss about iodometry in detail.
Iodometric Titrations
Iodometric titration is used to determine the strength or the concentration of an oxidising agent. In this method, I- is oxidised into I2 in the presence of an oxidising agent. The liberated iodine is estimated quantitatively by titrating with a standard thiosulphate solution. This method can be used to estimate almost all strong oxidising agents. Here, starch is used as an indicator which gives a dark blue colour in presence of I2.
The steps involved in an iodometric titration are as follows.
Step 1: An oxidising agent reacts with iodide ions (example: KI, potassium iodide).
From the equation,
Equivalents of oxidising agent = Equivalents of I- oxidised = Equivalents of I2 formed
Step 2: The iodine liberated is titrated with a standard hypo solution (Na2S2O3).
From the equation, Equivalents of I2 = Equivalents of S2O32-
Therefore, from the amount of iodine consumed by the thiosulphate ions, the amount of oxidising agent can be calculated. The combined schematic representation can be given as
The following are some of the widely used iodometric analyses in the laboratory.
Practice Problems
1. Find the n-factor of S in Na2S2O3 in the reaction
a. 1
b. 2
c. 3
d. 4
Answer: B)
Let the oxidation state of S in Na2S2O3 be ‘x’
Let the oxidation state of S in Na2S4O6 be ‘y’
n-factor of S = number of S atoms in Na2S4O6 ✕ |O.S. of S in Na2S4O6- O.S. of S in Na2S2O3|
n-factor of S = 2 |2.5 - 2| = 1
So, option A) is the correct answer.
2. Find the n-factor of Br in BrO3- in reaction
a. 7
b. 6
c. 5
d. 4
Answer: B)
Let the oxidation state of Br in BrO3- be ‘x’
Let the oxidation state of Br in Br- be ‘y’
n-factor of Br
= number of Br atoms in BrO3- ✕ |O.S. of Br in Br- - O.S. of Br in BrO3-|
n-factor of Br = 1 |-1-5| = 6
So, option B) is the correct answer.
3. An aqueous solution containing 0.20 g KIO3 reacted with an excess of acidified KI with HCl. The liberated I2 consumed 56.0 mL of hypo. Calculate the normality of hypo solution
a. 0.025 N
b. 0.0025 N
c. 0.25 N
d. 2.5 N
Answer: A)
Step 1: KIO3+5KI+6HCl3I2+6KCl+3H2O
Moles of KIO3 = 0.2214=9.310-4 mol
Moles of I2 formed = 3 9.310-4 mol
Step 2: I2+2Na2S2O32NaI+ Na2S4O6
Moles of Na2S2O3 required =
Molarity of Na2S2O3 =
n-factor of Na2S2O3 = 1
Normality of Na2S2O3 = =0.025 N
So, option A) is the correct answer.
4. Find the n-factor of I in IO3- in reaction
a. 5
b. 6
c. 7
d. 8
Answer: B)
Let the oxidation state of I in IO3- be ‘x’
Let the oxidation state of I in I2 be ‘y’
n-factor of I = 1 |5-0| = 5
Frequently Asked Questions - FAQ
1. In an iodometric titration, which indicator is used?
Answer: Starch is typically used as the indication for iodine titrations. Iodine and starch combine to create a deep blue complex. Iodimetry's terminal point is marked by a sharp shift in the colour of the solution to blue.
2. In iodometry, why is acetic acid added?
Answer: Glacial acetic acid's function is to maintain a pH of 3 to 4 so that starch can perform its function in the titration perfectly.
3. Why can not the starch solution be added to an iodometric titration at the beginning?
Answer: Starch cannot be added to the iodometric titration at the beginning because iodine can rapidly react with it to produce a dark blue solution, which will obstruct the end point detection. Therefore, we add starch indicator as soon as the iodine colour in the solution begins to fade, which is when it turns pale yellow and indicates a very low iodine content.
4. How can the end point of an iodometric titration be determined?
Answer: A starch solution is used as an indicator in an iodometric titration because it can absorb the I2 that is released. When titrated with standardised thiosulfate solution, this absorption causes the solution's colour to change from deep blue to light yellow. This represents the titration's end point.


