-
Call Now
1800-102-2727
Internal Energy of a Gas - Definition, Change in Internal Energy, Practice Problems and FAQs

While flipping the bottle, we can observe that the bottle moves through the air and because of this it possesses kinetic energy.
At the same time because of its height from the ground, it also attains a certain amount of potential energy. These are the energies with which we are familiar since our early school days. But are these the only energies we should check for? Is there something more inside or some different type of energy we see inside the bottle? Let’s check it out.
If we zoom into the water bottle, we can see the water molecules are in constant random motion and possess kinetic energy.

We know three types of motion possible and the energies associated with these motions. Translational motion, Rotational motion, and Vibrational motions and their respective energies are associated with them.
If we look further at a molecular level, we can see that there are interactions between the polar water molecules. These could be intermolecular hydrogen bonding interactions and within a molecule, there are covalent interactions between the bonded atoms.
These intermolecular and intramolecular interactions are associated with the chemical potential energy in the system. This energy is also an add-on to the system's total energy, i.e., water inside a bottle. Can you think of some more types of energies which can be associated with the system? Let’s see.
If we further zoom into the nuclear level of an atom, we know there exist neutrons and protons inside the nucleus which are held together by nuclear energy. It doesn’t end here, there are many other forms of energies stored in the system as well.
Now if we add all these energies, it gives us the total energy content of this system i.e., the bottled water.
Now, if we closely observe this equation, we get to know that apart from the KE and PE occurring at the macroscopic external level, rest of the energies are internal and inherent to the system. These energies which are inherent to the system are collectively called the internal energy of the system. Let’s study more about the internal energy of the system and see its significance of internal energy.
Table of Contents
- Definition of Internal Energy
- Internal Energy Change
- Change in Internal Energy for Different Gases
- Change in Internal Energy in a Cyclic Process
- Practice Problems
- Frequently Asked Questions – FAQs
Definition of Internal Energy
Internal energy is a type of energy that arises from the molecular state of motion of matter and is found in every system. The letter U stands for internal energy, and the joule is the unit of measurement of internal energy (J).
The entire kinetic energy attributed to the mobility of molecules, as well as the potential energy associated with the vibrational motion and electric energy of atoms within molecules, makes up the internal energy (U) of a system or a body with well-defined limits. The energy in all chemical bonds is included in internal energy. For any material or repulsion between individual molecules, internal energy can be discovered in a variety of ways at the microscopic level.
Internal energy is an extensive property and is a state function of a system. One can have a corresponding intensive thermodynamic property called specific internal energy, which is internal energy per unit mass of the substance and is often denoted by the lowercase letter (u). As a result, the SI unit of specific internal energy would be the (J/g). If the internal energy is stated in terms of the number of moles of the substance, it is known as molar internal energy, and the unit is (J/mol).
Internal energy of a gas
The internal energy of a system can be understood by examining the simplest possible system: an ideal gas. The particles in an ideal gas do not interact, hence there is no potential energy in this system. The internal energy of an ideal gas is therefore the sum of the kinetic energies of the particles in the gas.
According to the kinetic molecular theory, a gas's temperature is proportional to the average kinetic energy of its particles. For one mole of an ideal gas, kinetic energy is:

The internal energy of an ideal gas is therefore directly proportional to the temperature of the gas as it is the sum of the kinetic energies of the particles in the gas.
The internal energy of systems that are more complex than an ideal gas can't be measured directly. However, the system's internal energy remains proportional to its temperature. As a result, we can track changes in a system's internal energy by examining what happens to its temperature. When the system's temperature rises, we can deduce that the system's internal energy increased as well.
Internal Energy Change
Every substance has a definite amount of energy that is determined by its chemical constitution and current state of existence. This is known as intrinsic energy. Every substance has an internal energy value that is equal to the energies of all of its constituents, such as atoms, ions, or molecules.
The difference between the internal energies of the two states can be used to calculate the change in internal energy. .
The internal energies of a system in states A and B, respectively, are Ua and Ub. The change in the internal energy can be calculated as:
ΔU = Ub-Ua
The internal energy difference has a set value and is unaffected by the path followed between two states A and B. The difference between the internal energies of the products and the reactants can be considered the change in internal energy for a chemical reaction.
Where, Uproducts is the summation of the internal energies of all the reactants and Ureactants is the summation of internal energies of all the reactants.
Take example of a chemical reaction:
Change in internal energy for this reaction can be written as:
Change in Internal Energy for Different Gases
For ideal gases,
Where,
ΔU = Change in internal energy
n = number of moles of gas
CV= molar specific heat capacity at constant volume
T = change in temperature
R = Universal gas constant
As ,
Where,
f = Degree of freedom
For a monatomic gas,
(Since, the degree of freedom for monoatomic gas = 3)
For a diatomic gas,
(Since, the degrees of freedom for diatomic gas = 5)
For polyatomic linear gas molecules,
(Since, the degrees of freedom for polyatomic linear gas = 5)
For polyatomic non-linear gas molecules,
(Since, the degrees of freedom for polyatomic non-linear gas = 6)
For particular gas molecules, it is observed that the change in internal energy depends only on the
change in temperature from these equations. The degrees of freedom and number of moles are considered to be constant. As it depends only on the initial and the final state of the system, it is a state function (A state function is a property whose value does not depend on the path taken to reach that specific value).
Internal energy is an extensive property as it is dependent on the number of moles.
Hence, internal energy can be changed by the following:
• Temperature
• Type of the gas (monoatomic, diatomic, etc.)
Internal energy is dependent on the freedom of movement of particles. Since in a gaseous state, particles are most free to move, this state has maximum internal energy followed by liquid and solid.
Suppose as illustrated in the diagram, for an ideal gas, the gas is expanded from point A to point B along the path AB. During the transition, the gas's internal energy changes as follows:
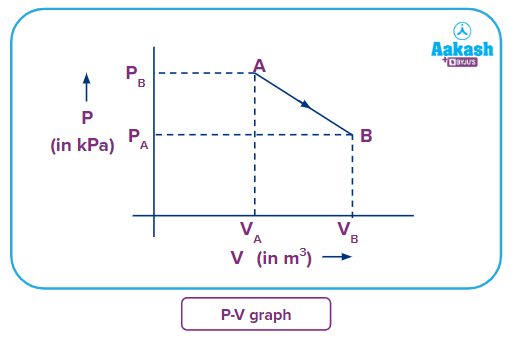
For ideal gases,
ΔU = nCVT
Where , n = number of moles
CV= molar specific heat capacity at constant volume
T= change in temperature
Applying ideal gas equation on gas at point A
PAVA= RTA
Applying ideal gas equation on gas at point B
PBVB= RTB
Change in Internal Energy in a Cyclic Process
For a closed system the internal energy can be represented using the first law of thermodynamics:
ΔU = q + W
Where,
- ΔU is the change in a system's internal energy during a process
- q is the heat transfer
- W is the mechanical work.
If an energy exchange occurs because of the temperature difference between a system and its surroundings, this energy appears as heat; otherwise, it appears like work.
After completing the cycle, a cyclic process returns the system to its initial condition.
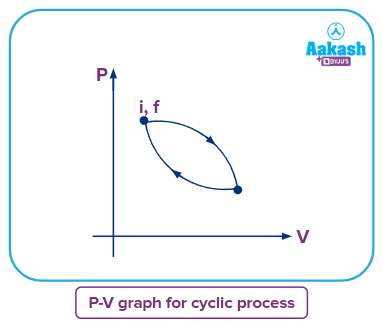
As the change in internal energy is a state function, its value depends on the initial point and final point. In a cyclic process initial and final points are the same, so the change in internal energy will be zero.
For an ideal gas,
Related Video Link : https://www.youtube.com/watch?v=5H9si8LPcjM
Time Stamp: (00:30 to 04:40)
Practice Problems
Q. Against a steady pressure of 1.0 atm, a gas expands adiabatically from 2.0 L to 14.0 L. The change in the internal energy during the process is:
(1 atm-L = 101.325 J)
A)−1215.9 J
B) 1114.9 J
C) 1215.9 J
D)−835.9 J
Answer: (A)
From the first law of thermodynamics;
∆U=q+W
As q = 0 (Adiabatic process)
∆U=W
Formula can be used to find the value of 𝛥U under irreversible adiabatic expansion is:
W = −pext(ΔV) = −pext(V2−V1)
Pressure (pext) = 1 atm
Initial volume (V1) = 2.0 L
Final volume (V2)=14.0 L
Therefore,
∆U=W=−pext(V2−V1)
∆U=−1.0 atm (14 L−2 L) = − 12 atm-L
Converting the value of 𝜟𝑼 in Joules;
1 atm-L = 101.325 J
Therefore, the value of ΔU when a gas expands adiabatically from 2.0 L to 14.0 L against a constant pressure of 1 atm is −1215.9 J.
Q. A system absorbs 600 J of heat and does work of 100 J on surroundings. What will be the change in the internal energy?
A) 300J
B) 400J
C) 700J
D) 600J
Answer: (C)
Solution:
Heat absorbed by the system (q) = 600 J
Work done by the system (W)=-100 J
Using, the first law of thermodynamics,
∆U=q+W
∆U= (600 - 100) J
∆U= 500 J
Q. What would be the value of change in internal energy if one mole of diatomic gas expands at constant pressure if the temperature of gas increased from 35°C to 300°C ? (R = 2 Cal K-1mol-1)
(A) 1125 Cal
(B) 1225 Cal
(C) 675 Cal
(D) 1325 Cal
Answer: (D)
Solution: For a diatomic gas,
(Since, the degrees of freedom = 5)
Given,
n = 1 mol
T1=35°C = (35 +273)K = 308 K
T2=300°C =(300 +273)K = 573 K
ΔT = T2-T1 = (573 - 308)K = 265K
Now,
Q. The change in internal energy of a system depends on
a. initial and final states of the system
b. path followed by the system
c. initial, final states and also on the path
d. None of these
Answer: (A)
Solution: Internal energy is a function of the state. As a result, the change in internal energy of a system is solely determined by the system's starting and final states.
Q. What will be the change in internal energy for this reaction?
UOF2 = 220 Cal
UH2O = 345 Cal
UO2 = 115 Cal
UHF = 325 Cal
A) 500 Cal
B) 400 Cal
C) 200 Cal
D) 300 Cal
Answer: (C)
Solution: Change in internal energy for this reaction will be:
Frequently Asked Questions – FAQs
Q. What will happen to the change in internal energy at constant volume?
Answer: At constant volume, no P-V work can be done, and the change in the internal energy of the system is equal to the amount of heat transferred from the system to the surroundings or vice versa.
Q.What influences the internal energy of a system?
Answer: The internal energy can be altered by modifying the object’s temperature or volume without altering the amount of particles inside the body. As a system’s temperature increases, the molecules will move faster, thus having more kinetic energy and thus the internal energy will increase.
Q. Why internal energy is a state function as well as extensive property?
Answer: The thermodynamic properties which do not depend upon the path followed by the thermodynamic process are known as state properties. State property depends only upon the initial and final state of the system.
The properties which depend upon the amount of matter are known as extensive properties. The internal energy depends upon the amount of matter. As more particles cause more collisions, so internal energy increases. So, the internal energy is an extensive property.
Q. What is the change in internal energy for an isothermal process?
Answer: For an isothermal process, the temperature remains constant. So, the change in internal energy is zero as it is a function of temperature.


