-
Call Now
1800-102-2727
Intermolecular Bonds - Definition, Types of Intermolecular Bonds, Practice Problems & FAQs
Have you ever seen a magnet? Have you ever tried playing with magnets?
We know, that when we bring the north pole of a magnet close to the south pole of another magnet, they attract each other and this happens due to the magnetic forces between them.
Just like magnetic forces, we have many other forces/bonds in chemistry which hold atoms/molecules together and can cause changes to the atoms/molecules.
Let’s find out what kind of interactions we are talking about.
Table of Contents
- What are Intermolecular Bonds?
- Ion-Dipole Interactions
- Ion-Induced Dipole Interactions
- Dipole-Dipole Interactions
- Dipole-Induced Dipole Interactions
- London Dispersion Forces
- Practice Problems
- Frequently Asked Questions - FAQs
What are Intermolecular Bonds?
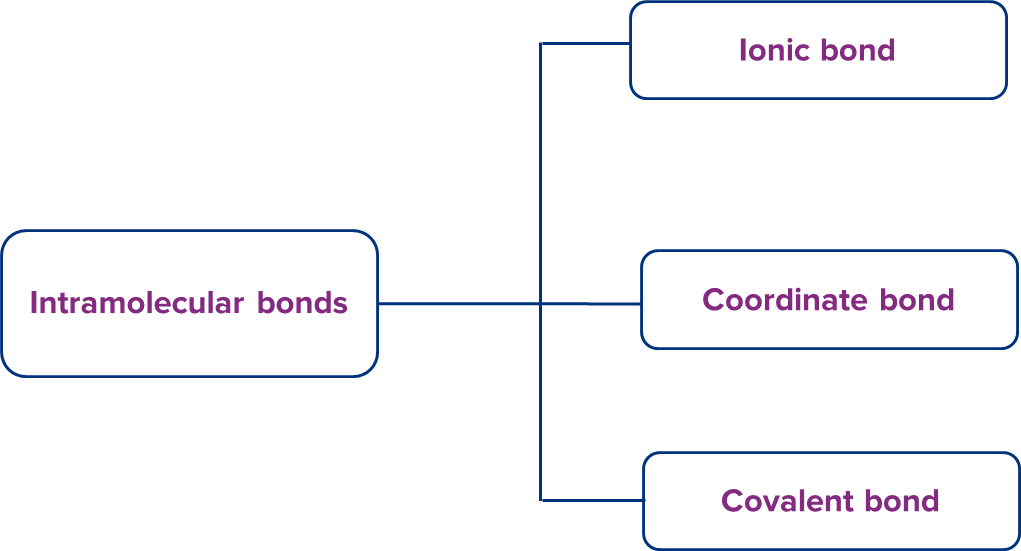
Intramolecular and intermolecular chemical bonding are the two types of chemical bonding, which are further classified into groups as follows:
Intramolecular bonds are chemical bonds that exist between the atoms of a molecule or substance.
Intramolecular bonds can be divided into three categories:
Intermolecular bonds are attractive interactions that exist between two particles that are close together (atoms, molecules or ions). They have lower strength than intramolecular bonds. Intermolecular bonds can be divided into different categories:
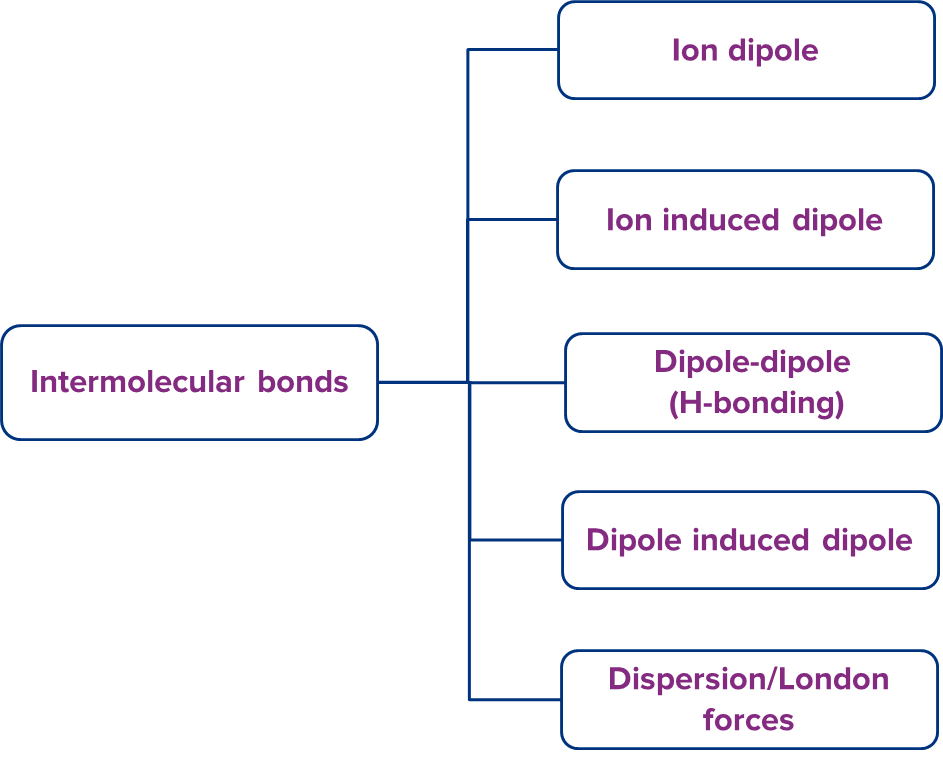
We will be discussing everything about intermolecular bonds.
Ion-Dipole Interactions
Ion-dipole forces are the result of electrostatic interactions between an ion and the charged ends of a dipole, and their name accurately depicts what they are.
In solutions containing an ionic compound dissolved in a polar solvent, such as a solution of table salt
(NaCl) in water, ion-dipole Forces are present because they involve two separate species (an ion and a polar molecule).
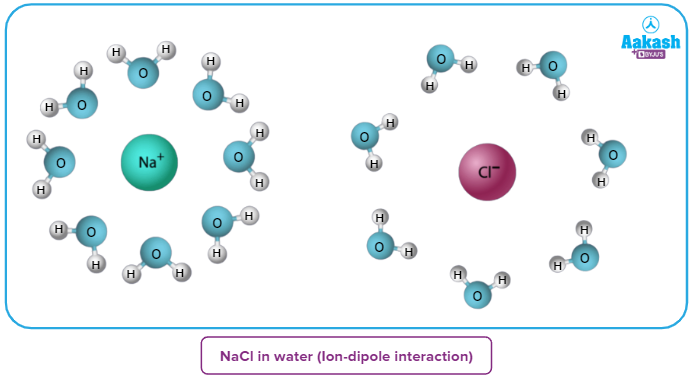
As we can clearly see in this example, water is a polar molecule and NaCl is an ionic compound. When NaCl is dissolved in water, Na+ is the positively charged ion in it whereas Cl- is the negatively charged ion in it. When Na+ and Cl- ions come in contact with H2O, which is a polar molecule, ion-dipole interactions are produced.
Ion-Induced Dipole Interactions
When a nonpolar molecule interacts with an ion, there will be a formation of ion-induced dipole interactions, which are weak attraction forces. When a charged ion comes closer to a non-polar molecule, the molecule gets polarised as electrons will be unevenly distributed to two different ends of the molecule.
A negatively charged ion repels the electron cloud of the molecule and polarises it. Whereas, a positively charged ion attracts the electron cloud of the molecule and polarises it.


We can clearly see above that when an ion is approaching towards the atom, it is changing into a dipole which is a clear illustration of how dipoles can be induced with the help of an ion. This can be seen in nitrate ion and iodine molecule interaction.
Dipole-Dipole Interactions
Between two permanent polar molecules, the electrostatic forces which are present are known as dipole-dipole interactions. The positive end of one molecule is generally attracted to the negative end of another molecule. As a result, the two molecules come closer, increasing their stability. The dipole-dipole interaction is different from a conventional bond because here electrons are not transferred or shared.
The dipole-dipole interactions are caused by the dissimilar distribution of electrons in the molecule. Usually, for a molecule which can show dipole-dipole interaction, the electrons get collected at one end. Due to this, partial negative and positive charges are developed on the ends having more and less number of electrons respectively. Now, these two molecules which are polar in nature with opposite charges naturally attract each other.
This dipole-dipole interaction is a type of Van der Waals force also known as Keesom forces.


For stationary polar molecules;
Dipole-dipole interaction energy
For rotating polar molecules;
Dipole-dipole interaction energy
Where, r is the distance between the polar molecules.
Dipole-Induced Dipole Interactions
A polar molecule can generate a dipole in a molecule/atom which is non-polar by disturbing the arrangement of electrons in the non-polar molecules. Now, attraction forces can be generated between polar and non-polar molecules and these attraction forces are known as dipole-induced dipole interactions. The existence of a polar molecule is required for the dipole–induced-dipole interaction to occur.
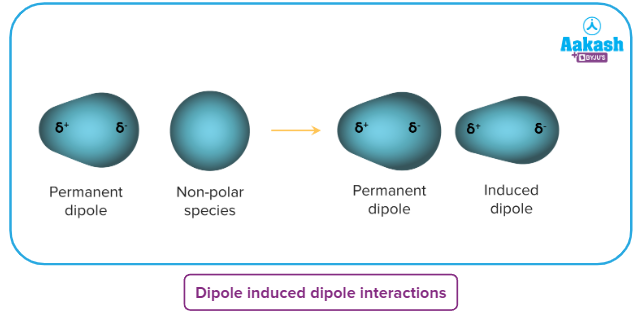
We can see when the permanent dipole of a water molecule interacts with a chlorine molecule (non-polar species). The electric field provided by the permanent dipole of the water molecule induces the dipole in the non-polar chlorine molecule. Induction (or polarisation) interaction refers to the permanent dipole-induced dipole interaction.
This dipole-induced dipole interaction is a type of Van der Waals force also known as Debye forces.
For dipole induced dipoles;
Dipole-induced dipole interaction energy
Where, r is the distance between two molecules.
Dispersion Forces

When electrons are dispersed asymmetrically around the nucleus, an atom or molecule might produce a temporary (instantaneous) dipole due to the continuous mobility of the electrons. Because electrons repel each other, the development of the dipole in the first atom or molecule might distort a second atom or molecule, resulting in an electrostatic attraction between the two atoms or molecules.
This dispersion force is a type of Van der Waals force also known as London forces or London dispersion forces.
These types of interactions are present in the molecules which are non-polar like F2, N2, H2, etc. All these molecules are electrically symmetrical and have no permanent dipole. These forces are applicable only at short distances (≈ 500 pm) when they are almost touching each other.


London dispersion forces are typically produced:
- between molecules that are easily polarised and the interactions in those cases are stronger.
- between molecules that aren't easily polarised and the interactions in those cases are weaker.
Magnitude of London Dispersion Forces ∝ Polarisability of the specie
Also,
London Dispersion interaction energy
Where, r is the distance between two molecules.
Practice Problems
Question: Which of the following pairs has dipole-induced dipole interactions?
A. SiF4 and He
B. H2O and CH3OH
C. HCl and He
D. Cl2 and CCl4
Answer: (C)
Solution: Dipole-induced dipole interaction is observed when one molecule is polar while another molecule is nonpolar in nature. Due to the polar nature of the molecule, it induces the dipole in the nonpolar molecule. HCl molecule is polar in nature whereas He is non-polar in nature and therefore dipole-induced dipole interaction is present.
So, the correct answer is option (C).
Question: What kind of intermolecular attraction develops between temporary dipoles and their induced temporary dipoles?
A. London dispersion
B. Ionic bond
C. Hydrogen bond
D. Metallic bond
Answer: (A)
Solution: When two nonpolar molecules come closer to each other one molecule induces a temporary dipole in the molecule known as London dispersion forces or induced dipole-induced dipole interaction. London dispersion forces are present between two nonpolar molecules. Therefore, option A is correct.
Question:F2 and Cl2 are gases at room temperature, Br2 is a liquid, and I2 is solid. This happens because:
A. the strength of a dipole-dipole interaction increases as the molecular size increases.
B. with increasing molecular size, polarity increases.
C. with increasing molecular size, dipole-induced dipole interactions increase.
D. dispersion interactions increase as molecular size increases, and polarity increases as well.
Answer: (D)
Solution: Halogen molecules are nonpolar in nature. When two nonpolar molecules come closer to each other one molecule induces a temporary dipole in the molecule known as London dispersion forces or induced dipole-induced dipole interaction. London dispersion force is a very weak interaction force which is directly proportional to the molecular mass of the molecule.
On moving down the group, the molecular mass and size of halogen molecules increase which results in an increase in the London dispersion forces. Therefore iodine exists in the solid state, bromine in the liquid state, and chlorine and fluorine exist in the gaseous state.
Question: Solid CuSO4 dissolves in water because of many reasons, one of the reasons for this is:
A. Molecules' ion-dipole forces between the ions and the water.
B. Cu2+ and ions have instantaneous dipole-induced dipole forces (dispersion or London forces).
C. Cu2+ and ions are attracted to one another electrostatically.
D. the hydrogen bonds between the water and copper ions.
Answer: (A)
Solution: When copper sulphate ( CuSO4) is dissociated into ions by supplying the energy proportional to the lattice energy of the copper sulphate. It results in the dissociation of ions. Once the molecule dissociates into ions on adding water both the ions formed (Cu2+ and ) is surrounded by the water molecule. A water molecule is a polar molecule and when it interacts with the ions it results in the ion-dipole interaction which releases energy in the form of hydration energy and helps the solid copper sulphate to dissolve in water. Therefore, option A is correct.
Frequently Asked Questions-FAQs
Question: How do we figure out how strong molecules' intermolecular forces are?
Answer: The nature of the interacting molecules determines intermolecular forces. A nonpolar molecule, for example, can be polarised by the presence of an ion nearby, causing it to become an induced dipole. Ion-induced dipole interactions are the result of their interaction. So, by figuring out what kind of molecule (polar or nonpolar) we are dealing with, we can understand how strong are the intermolecular forces.
Question: What's the difference between an ion-induced dipole and an ion-dipole?
Answer: A fully charged ion interacts with a partially charged dipole in an ion-dipole interaction. The ion-dipole force's strength is proportional to the ion charge. On the other hand, a fully charged ion interacts with a temporarily charged dipole in an ion-induced dipole interaction. The existence of the ion causes the temporary dipole to form.
Question: What creates dipole-dipole interactions in the first place?
Answer: The basic need for dipole-dipole interactions between molecules is that both molecules contain dipoles. The dipoles we are talking about here might be permanent or temporary. Dipoles are generated by unequal distribution of electrons in the molecule.
Question. What is the difference between dipole-dipole interactions and hydrogen bonding?
Answer: Hydrogen bonding is a special type of dipole-dipole interaction which is observed in the molecule when a lone pair present on the electronegative elements like fluorine oxygen, nitrogen and in some cases chlorine atom interact with the hydrogen attached with the more electronegative element like- fluorine, oxygen and nitrogen. Hydrogen bonding is stronger than dipole-dipole interaction because more energy is released during the hydrogen bond formation. Whereas dipole-dipole interaction takes place between the polar molecules except for the molecules in which more electronegative elements (fluorine, oxygen and nitrogen) are present and are bonded with the hydrogen atom.


