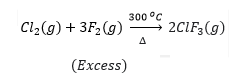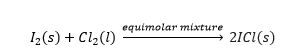-
Call Now
1800-102-2727
Interhalogen Compounds Preparation - Definition of Interhalogen Compounds, Practice Problems and FAQs
If you have siblings, you know that you play with them, compete with them, fight with them and most importantly live happily. Do you know how these attributes are related to the chemical elements we see in the periodic table? Definitely! There are elements in the periodic table which can react with their own member present in the family. These elements are present in the same group together in group-17 and are known as the halogen group which includes -fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I) and two other elements that are radioactive in nature Astatine (At) and Tennessine (Ts) respectively. Let’s discuss what kind of compounds are these and how these compounds can be prepared.
Table of contents
- What are Interhalogen Compounds?
- Preparation of Interhalogen Compounds
- Practice Problems
- Frequently Asked Questions-FAQs
What are Interhalogen Compounds?
Interhalogen compounds are defined as compounds which are formed by combining two or more distinct halogen atoms when allowed to react with one another. Most of the halogen compounds are binary in nature (i.e composed of two different halogen components). It is generally represented by the formula (XYn), where n = 1, 3, 5 and 7 and ‘X' represents the less electronegative halogen atom, ‘Y' represents the more electronegative element.
The possible interhalogen compounds are listed below:
|
AX |
AX3 |
AX5 |
AX7 |
|
IF(unstable) |
IF3(unstable) Yellow |
||
|
ClF(g) Colourless |
ClF3(g) Colourless |
ClF5(g) Colourless |
|
|
BrF(g) Pale brown IBr(s) Black |
BrF3(l) Pale yellow |
BrF5(l) Colourless |
|
|
ICl(s) Ruby red |
(ICl3)2 Bright yellow |
IF5(l) Colourless |
IF7(g) Colourless |
Preparation of Interhalogen Compounds
The formation of interhalogen compounds is influenced by a variety of situations in which distinct compounds are generated. These compounds could be made by halogen reacting with each other directly or by halogen reacting with the lowest interhalogen compound. Any type of interhalogen chemical synthesis is dependent on different conditions which include elements' characteristics, the temperature at which the reaction is taking place, and the composition of different reactants.
Chlorine, for example, becomes ClF when it interacts with fluorine of equal volume at 437 K. This approach is frequently used in the synthesis of group 17 fluorides.
Recommended video link: https://www.youtube.com/watch?v=esGy1sWRUnU
Practice Problems
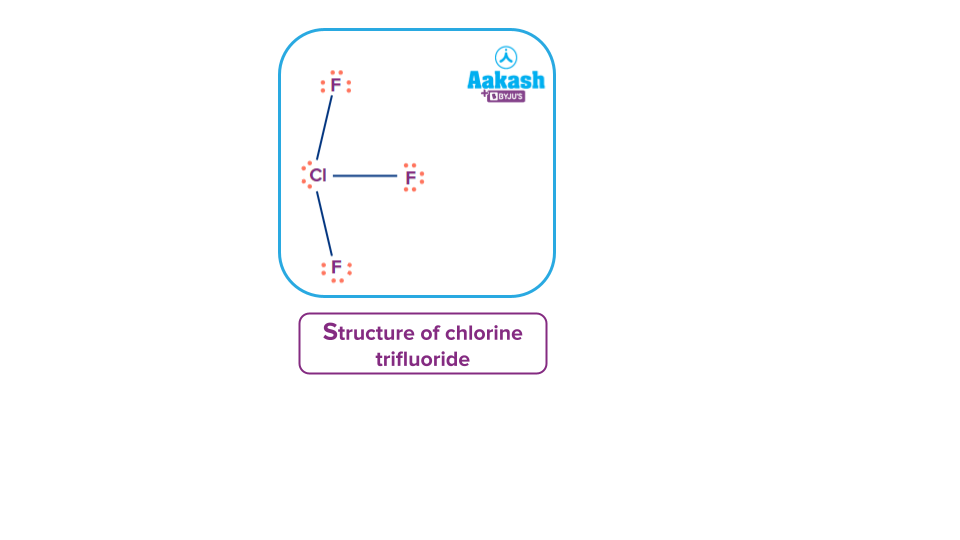
Q. Select the correct option for the number of lone pairs present in the compound with formula ClF3.
a. 12
b. 10
c. 11
d. 2
Answer: (C)
Solution: In the case of chlorine trifluoride molecule, there are two lone pairs on the central atom and each fluorine atom contain 3 lone pairs, therefore, a total number of lone pairs in the complete molecule of chlorine trifluoride becomes 11. But the number of lone pairs present on the central atom is only 2.
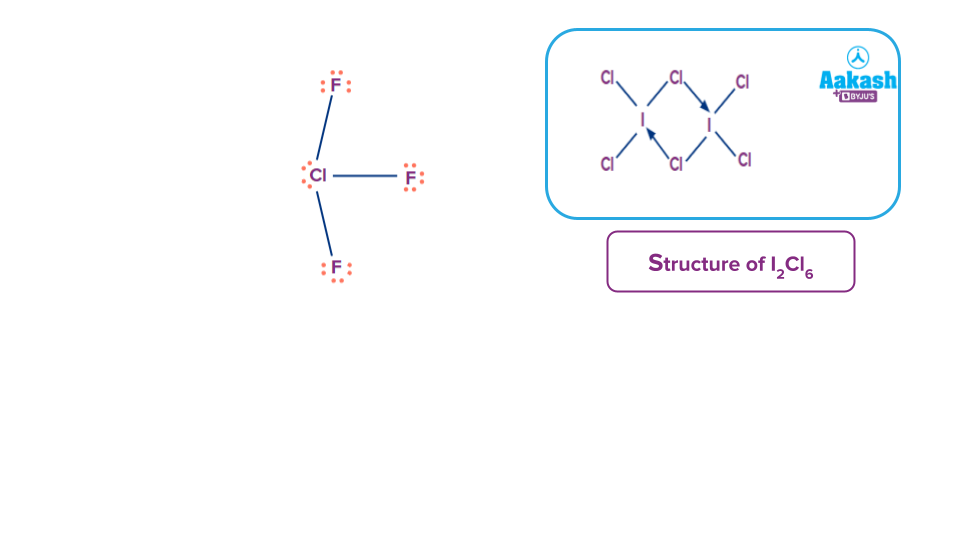
Q. Select the correct option which is unstable in nature and has the tendency to get dimerise.
a. ClF3
b. ICl3
c. BrF5
d. IF5
Answer: (B)
Solution: ICl3 is unstable and has a tendency to get dimerise and form I2Cl6 compound which is bright yellowish colour solid compound and contains two 3C-4e bonds and known as banana bonds or bridge bonds and four 2c-2e bonds which are also known as the terminal bonds.
Q. Select the incorrect option which represents the incorrect geometry of given interhalogen compounds.
a. ClF3 (Trigonal bipyramidal)
b. IF7 (octahedral)
c. ICl2+(Tetrahedral)
d. IF5 (octahedral)
Answer: (B)
Solution: Geometry of the compound depends upon the hybridisation of the compound which in turn depend upon the number of bond pair and a number of lone pair present in the central atom .
The geometry and hybridisation of the compounds given in the options are listed below:
|
Compound |
Hybridisation |
Geometry |
|
ClF3 |
sp3d |
Trigonal bipyramidal |
|
IF7 |
sp3d3 |
Pentagonal bipyramidal |
|
IF5 |
sp3d2 |
Octahedral |
|
ICl2+ |
sp3 |
Tetrahedral |
According to the above table, option (B) is incorrect.
Q. Select the correct option for the interhalogen compound which has self ionisation tendency.
a. BrF5(l)
b. I2Cl6(l)
c. IF5(l)
d. All of the above is correct
Answer: (D)
Solution: Generally interhalogens can be used as a non-aqueous ionising solvent due to their self ionisation tendency.
Frequently asked questions - FAQs
Q. Why generally interhalogen compounds are more reactive than halogens?
Answer: Interhalogen compounds are more reactive than halogens except for fluorine because in the case of interhalogen compounds bond energy is less and is polar in nature as compared to the bond energy of halogen molecules. In case of fluorine reactivity increases due to its small size, highest electronegativity value and less bond energy, it readily reacts with other molecules forming compounds.
Q. What are the different types of interhalogen compounds that can be formed?
Answer: Four different types of interhalogen compounds can be formed:
- XY type: In these types of interhalogen compounds there is one bond pair of electrons present between two different halogen atoms. For example, IF, ClF, ICl.
- XY3 type: In these types of interhalogen compounds there are three bond pairs of electrons present between two different halogen atoms X and Y. For example, IF3, ClF3, BrF3 .
- XY5 type: In these types of interhalogen compounds there are five bond pairs of electrons present between two different halogen atoms X and Y. For example, IF5, ClF5, BrF5 .
- XY7 type: In these types of interhalogen compounds there are seven bond pairs of electrons present between two different halogen atoms X and Y. For example, IF7.
|
AX |
AX3 |
AX5 |
AX7 |
|
IF(unstable) |
IF3(unstable) Yellow |
||
|
ClF(g) Colourless |
ClF3(g) Colourless |
ClF5(g) Colourless |
|
|
BrF(g) Pale brown IBr(s) Black |
BrF3(l) Pale yellow |
BrF5(l) Colourless |
|
|
ICl(s) Ruby red |
(ICl3)2 Bright yellow |
IF5(l) Colourless |
IF7(g) Colourless |
Q. Mention some significant uses of interhalogen compounds.
Answer: Some important uses of interhalogen compounds includes:
- BrF5 is used as an oxidiser for propellants.
- Compounds like ICl, ClF3, BrF3 are used for the preparation of polyhalides.
- Compounds like ClF3, BrF3 are used as fluorinating agents to prepare a number of metal fluorides.
- ClF3 and BrF3 are used to make UF6, which is used in the enrichment of U-235.
Q. List some important properties of interhalogen compounds.
Answer: Some important properties of interhalogen compounds includes:
- These compounds are diamagnetic and covalent in nature.
- They form more reactive bonds than diatomic halogen bonds except for fluorine molecules.
- The physical properties of interhalogen compounds are transitional between their constituent halogen elements.
- Interhalogens can be used as a non-aqueous ionising solvent due to their self-ionisation tendency.
For example- Liquid IF5 self-ionises and therefore conduct electricity
- Interhalogen compounds are made up of lighter group 17 elements which are generally colourless, whereas compounds containing halogens having higher molecular mass are darker in colour.