-
Call Now
1800-102-2727
Individual and Average Oxidation states - Individual oxidation states, Average Oxidation states, Practice Problems and FAQs
Let's imagine there are three chemistry coaching classes in your neighbourhood.
‘Alpha Classes’
‘Beta Classes’
‘Gamma Classes’
Five students from both Beta and Gamma classes switched to the Alpha classes because of the great topic knowledge of the teacher in Alpha classes. How many students will switch from beta classes if two students switch from gamma classes?
Simple solution: Three students; Right???
The oxidation state is a related idea. According to their nature and electronegativity, all atoms either lose or gain electrons. If a compound is made up of three atoms and you know the identities of two of them, you can easily calculate the third atom's oxidation number.
TABLE OF CONTENT
- Individual Oxidation states
- Rules for Calculating Oxidation State
- Average Oxidation states
- Practice Problems
- Frequently Asked Questions
Individual Oxidation states:
An atom's hypothetical charge, assuming all of its bonds with other atoms were totally ionic in nature, is known as its oxidation state.
Some important points regarding the oxidation states are:
(i) The amount of electrons an atom lost or acquired depends on its oxidation state in a chemical molecule.
(ii) The amount of electrons an atom can receive or lose during a redox reaction is determined by its oxidation state.
(iii) In a redox reaction, the oxidation state is utilized to determine which species are reduced and which are oxidized.
(iv) An atom is said to be oxidized if its oxidation state increases. On the other hand, an atom is reduced when its oxidation state decreases.
Rules for Calculating Oxidation State:
To ascertain an atom's oxidation state in a compound, we must know and follow a set of rules.
- In either its free or uncombined state, each atom in an element maintains a zero oxidation state. Hence, oxidation state of each element in is zero.
- In their compounds, fluorine and other halogens have an oxidation state of (-1). But when combined with oxygen, iodine, chlorine, and bromine all have positive oxidation states (+1,+3,+5,+7).
- Alkali metals (Li,Na,K,Rb,Cs) always have (+1) oxidation state and Alkaline earth metals (Be,Mg,Ca,Sr, and Ba)always have a (+2) oxidation state when they combine to form a compound.
- Single-atom ions have an oxidation state according to their real charge. For instance, Zinc has an oxidation state of +2 in the form of Zn2+.
- Typically, oxygen is found in molecules in an oxidation state of –2. There are three exclusions.
- Oxygen exists in peroxides form, has an oxidation state of (-1).
Example: K2O2, the oxidation state of oxygen =x
- Oxygen exists in superoxides and has an oxidation state of (-1/2).
Example: RbO2, the oxidation state of oxygen is:
- When oxygen is bonded to an atom which is more electronegative than oxygen.
Example: Only fluorine is more electronegative oxygen. Hence, the oxidation state of oxygen in O2F2 = x
- When bonded to metals, hydrogen has an oxidation state of (-1) while when bonded to non-metals, it has an oxidation state of (+1).
For example, the oxidation state of H in BeH2
The oxidation state of H in H2S
When the oxidation states of the constituent atoms of a molecule are added together, the algebraic sum must be equal to zero.
For example, the oxidation state of Mn in KMnO4.
In the case of polyatomic ions, the algebraic sum of the oxidation states of the constituent atoms of the ion must be equal to the charge of the ion.
For example, the oxidation state of Cr in
Average Oxidation states:
The general guidelines assign an average oxidation number to some atoms in some compounds where sometimes the oxidation number appears to be different from what we have assumed. For instance, Fe’s oxidation number in Fe3O4 , comes out to be or S’s oxidation number in , comes out to be . Since electrons are never shared or transferred in fractions, this is unusual. Right?
The average oxidation number of the element in the compound is actually represented by this fractional oxidation number.
Example: Fe3O4 (Iron Oxide)
For example, Fe3O4 is composed of a mixture of FeO and Fe2O3, with the formula FeO.Fe2O3.
As a result, the oxidation number of Fe in FeO = x
Oxidation number of Fe in Fe2O3 = y
Fractional or Average Oxidation number of Fe in
Example: H2S4O6
The compound contains four atoms of sulfur. Consequently, the average sulfur oxidation state can be calculated as:
The oxidation state of the First sulfur
The oxidation state of the second sulfur Because the sulfur atoms are surrounded by sulphurs only and where there is a bond with the same atom, the contribution from them is taken to be zero.
The oxidation state of the fourth sulfur
Average oxidation state of S =
Related Topic Link : Paradox of Fractional Oxidation Number | CHEMISTRY | JEE | Shishir Mittal Sir | Concept of the Day
Practice Problems:
Q1. Calculate average oxidation number for Br in Tribromine Octaoxide radical?
Answer: (D)
Solution: Three bromine atoms are present in the given compound [Br3O8].
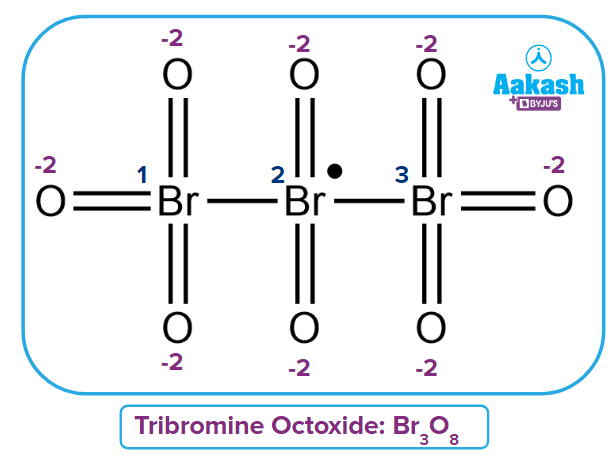
The average oxidation number of Bromine can be calculated as
Oxidation number of the First Bromine
Oxidation number of the second Bromine
Their contribution is taken to be zero because they are bonded to the same atom by a bond.
Oxidation number of the third Bromine
Fractional or Average Oxidation number of
Hence, option (D) is the correct answer.
Q2. Calculate the oxidation state of K in ?
A. +4, +2, +1
B. +2, +2, +2
C. +2, +4, +1
D. +1, +1, +1
Answer:(D)
Solution: Potassium (K) is an alkali metal and hence, has the tendency to donate an electron. Thus, it exists in +1 oxidation state.
In , oxygen exists in a peroxide form as . The oxidation state of oxygen in peroxide is -1.
Thus, the oxidation state of potassium in is
In KO2, oxygen exists in a superoxide form as . The oxidation state of oxygen in superoxide is .
Thus, the oxidation state of potassium in KO2 is
In K2O, oxygen exist in an oxide form as . The oxidation state of oxygen in oxide is -2.
Thus, the oxidation state of potassium in K2O is
So, option D is the correct answer.
Q3. Calculate fractional oxidation number for Mn in Mn3O4 ?
Answer: (C)
Solution: Mn3O4 exist as MnO and MnO2, with the chemical formula 2MnO.MnO2.
Oxidation number of Mn in MnO = x
Oxidation number of Mn in MnO2 = y
Fractional or Average Oxidation number of Mn in Mn3O4 (2MnO.MnO2) =
Hence, option (C) is the correct answer.
Q4. Pick the correct statement out of the following statements?
A. Chromium has the same oxidation state in both CrCl3 and CrCl2
B. Fluorine has the same oxidation state in both OF2 and O2F2
C. Chlorine has the same oxidation state in both HClO4 and HClO3
D. Phosphorus has the same oxidation state in both H3PO4 and H3PO2.
Answer: (B)
Solution: Cr in CrCl3:
Cr in CrCl2:
Thus, the oxidation state of chromium in CrCl3 and CrCl2 is not the same. Therefore, the statement given in option A is incorrect.
Fluorine is the most electronegative element in the periodic table. So, it always has -1 oxidation state in all its compounds.
F in OF2:
F in O2F2:
Thus, the oxidation state of fluorine in OF2 and O2F2 is the same. Therefore, the statement given in option B is correct.
Cl in HClO4:
Cl in HClO3:
Thus, the oxidation state of Chlorine in HClO4 and HClO3 is not the same. Therefore, the statement given in option C is incorrect.
P in H3PO4:
P in H3PO2:
Thus, the oxidation state of phosphorus in H3PO4 and H3PO2 is not the same. Therefore, the statement given in option D is incorrect.
Thus, the statements given in options A, C and D are correct, but the statement given in option B is correct.
So, option B is the correct answer.
Frequently Asked Questions-FAQs:
Q1. Is the oxidation state of fluorine positive?
Answer: The periodic table's most electronegative element is fluorine. Fluorine always has a negative oxidation state and cannot have a positive one.
Q2. Why does an apple become brown after being cut?
Answer: Oxygen gets into the plant tissue that has been damaged when you cut an apple. The phenolic compounds that are naturally present in apple tissues are immediately oxidized by the polyphenol oxidase (PPO) enzymes in the chloroplasts to o-quinones, which are colorless precursors to secondary products that are brown in color.
Q3. What real-world uses does oxidation have?
Answer: Either oxygen addition or electron loss occurs during oxidation processes. Oxidation is a process that occurs frequently in daily living, including breathing, combustion, and plant photosynthesis. As a result of this process, fats and oils become rancid and oxidized.
Q4. Are reduction and oxidation simultaneous processes?
Answer: Yes, oxidation and reduction happen at the same time. This is due to the need that another element acquires or takes the electrons that the oxidized element loses. As a result, both oxidation and reduction take place concurrently.


