-
Call Now
1800-102-2727
Important Compounds of Alcohol, Ethers and Phenol: Methanol, Ethanol, Phenol, Anisole, Practice problems and Frequently Asked Questions(FAQs)
Detergents, preservatives and fragrances are generally composed of these three basic compounds: alcohol, phenol and ether. We learned that replacing one or more hydrogen atoms in a hydrocarbon with another atom or group of atoms creates a whole new chemical compound with completely different properties and uses. When the hydrogen atom of an aliphatic or aromatic hydrocarbon is replaced with a -OH group, alcohol and phenol are formed. These chemicals have a wide range of uses in business and everyday life.
Compounds with -OH groups are found in the sugar we eat, the cotton we wear, and the paper we write. Think of a paperless life. There are no notebooks, books, newspapers, invoices, checks, or certificates, to name just a few. Like publications, beautiful photographs and compelling stories will disappear from our lives. It would have been a completely different planet.
Table of content:
- Methanol
- Ethanol
- Phenol
- Anisole
- Practice problems
- Frequently asked questions(FAQs)
Methanol:
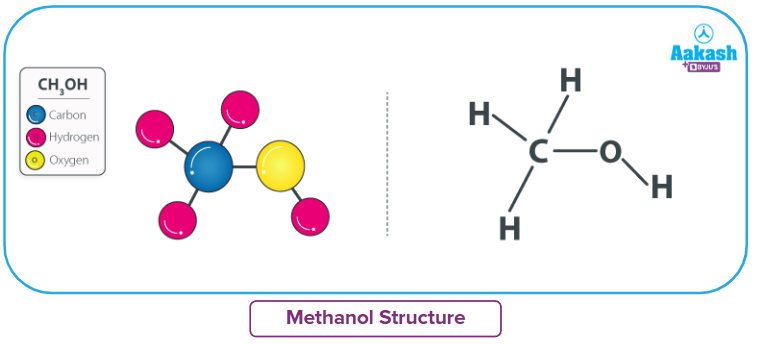
Structure of methanol
Methanol (CH3OH) is composed of one carbon, one oxygen and four hydrogen atoms. Carbon (6) has a valency of four, which means it needs four electrons to achieve stability. In methanol, carbon achieves this stability by forming covalent bonds with three hydrogens and one oxygen atom of the hydroxyl group.
In the molecule of methanol, both carbon and oxygen are sp3 hybridised. Because of sp3hybridization, the molecule has a tetrahedral structure, but due to the lone pair of electrons on oxygen, the bond angle H-O-C is less than 109.5o
 Production of Methanol
Production of Methanol
Conventional Method of Production
The reaction between carbon monoxide and hydrogen is the oldest and most widely used method for producing methanol. This carbon monoxide was originally derived from fossil fuels, but agricultural waste is now used to produce methanol in a much more environmentally friendly manner.
The catalyst zinc or cupric oxide, which is coated on the surface of alumina, facilitates the direct combustion reaction between carbon monoxide and hydrogen. Because of its high selectivity, the reaction is also used in the commercial production of methanol.
Biosynthetic Production of Methanol
With the help of the enzyme methane monooxygenase, methane can be converted to methanol. However, because this zinc or copper-dependent enzyme is not selective, the amount of methanol produced at the end of the process is minimal. Many investigations are being conducted to overcome this limitation.
Properties of Methanol:
Physical Properties:
- Methanol has a molecular weight of 32.04 g mol-1.
- Methanol has a boiling and melting point of 64.7oC and -97.6oC respectively.
- Methanol has a density of 0.792 g cm-3, which is less than that of water.
- Methanol has a hydroxyl group in its structure, which makes it both a polar molecule and water soluble.
Chemical Properties:
Oxidation reaction
The oxidation reaction of methanol produces formaldehyde, an industrially and commercially important chemical. The reaction is as follows. When methanol reacts with oxygen in the presence of potassium dichromate in an acidic medium, formaldehyde and water are produced. The reaction is depicted below.
Ester formation
Alcohol transforms into esters, a group of organic compounds that are found in organic chemistry, when it reacts with carboxylic acids. Below is an illustration of how methanol and acetic acid react to form an ester (methyl ethanoate).
This process occurs in an acidic medium (preferably a dilute sulfuric acid solution is used). Because methanol is a simple alcohol, it can easily combine with any carboxylic acid to create an ester with a fruity odour. Esters are extremely useful in a variety of commercial and industrial contexts, including the food and fragrance industries.
Ethanol:
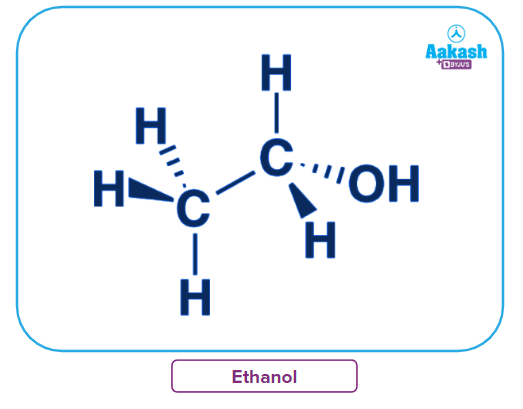
Structural of ethanol
Ethanol has two carbon atoms, six hydrogen atoms and one oxygen atom in its chemical formula C2H6O. The structural formula for ethanol C2H5OH, adds a little more detail, indicating that the 2-carbon chain ends in a hydroxyl group (-OH).
Preparations of ethanol
1. By fermentation of carbohydrates in presence of anaerobic bacteria:
Ethanol is produced on a large scale by fermenting molasses. Molasses is the mother liquor that remains after sugarcane juice crystallises, and it contains approximately 60% fermentable sugar. The procedure is as follows:
(a) Molasses dilution: Molasses is first diluted in a 1:5 (molasses: water) volume ratio with water.
(b) Ammonium sulphate fortification: Ammonium sulphate fortification supplies yeast with an appropriate source of nitrogen.
(c) Sulphuric acid addition: A little amount of sulphuric acid is added to the fortified molasses solution. This promotes yeast development while inhibiting the growth of harmful bacteria.
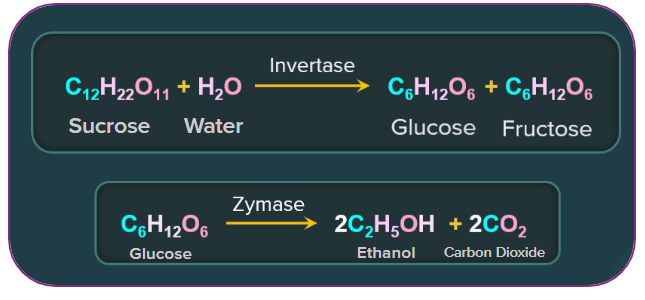
(d) Fractional distillation: The alcohol obtained during fermentation is known as "wash," and it is around 15% to 18% pure. It is turned into 92 percent pure alcohol known as rectified spirit or commercial alcohol via fractional distillation.
2. From Ethylene:
The hydration of ethylene is accomplished by passing a combination of ethylene and a substantial surplus of steam (H2O) over an acidic catalyst, such as a phosphoric acid catalyst, at high temperature (300oC) and pressure (60 atm).

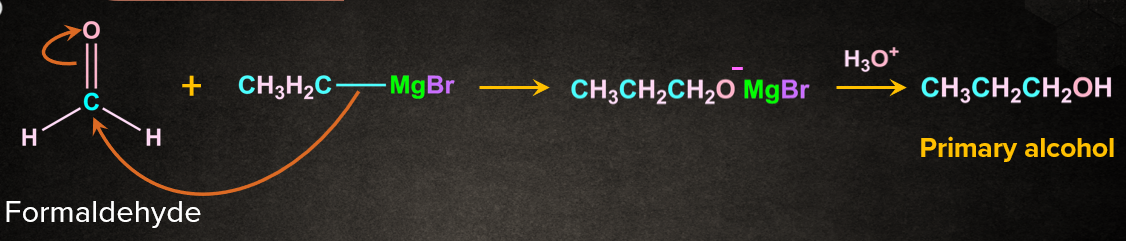
3. Reaction of formaldehyde with grignard reagent:
Formaldehyde on reacting with ethyl magnesium bromide followed by hydrolysis gives primary alcohol propanol.
Physical properties of ethanol
- Ethanol is a colourless liquid that is clear.
- Ethanol has a strong odour and a burning taste.
- Ethanol has a higher boiling point than ethane at 351 K.
- It is soluble in water, which means it can be mixed with any amount of water.
- It is highly flammable and explosive.
- Because of its low melting point of -114.5oC., it can be used in antifreeze products.
- It has a density of 789 Kg/m3, which is nearly 20 %less than that of water.
Chemical properties of ethanol
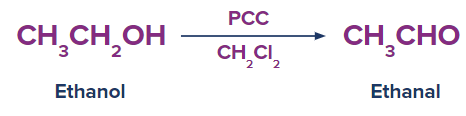
1. Conversion of Ethanol to Ethanal:
A molecule of hydrogen is removed during the conversion (oxidation) of ethanol to ethanal, and a C-O bond is transformed to a C=O bond. The hydroxyl group of alcohol is transformed to the carbonyl group of aldehyde during this reaction. Ethanal is transformed to ethanoic acid if it is oxidised further.
2. Reaction with sodium:
Ethyl alcohol on reaction with sodium metal will give sodium ethoxide and hydrogen gas.
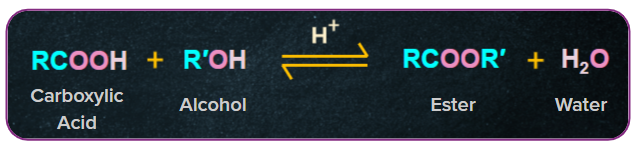
3. Esterification:
Esterification is a chemical reaction in which two reactants (alcohol and an acid) react to produce an ester as the reaction product. And they have the formula RCOOR' (where R and R' are any organic groups), and they are made by reacting alcohols with carboxylic acids in the presence of hydrochloric acid or sulphuric acid.
Phenol:
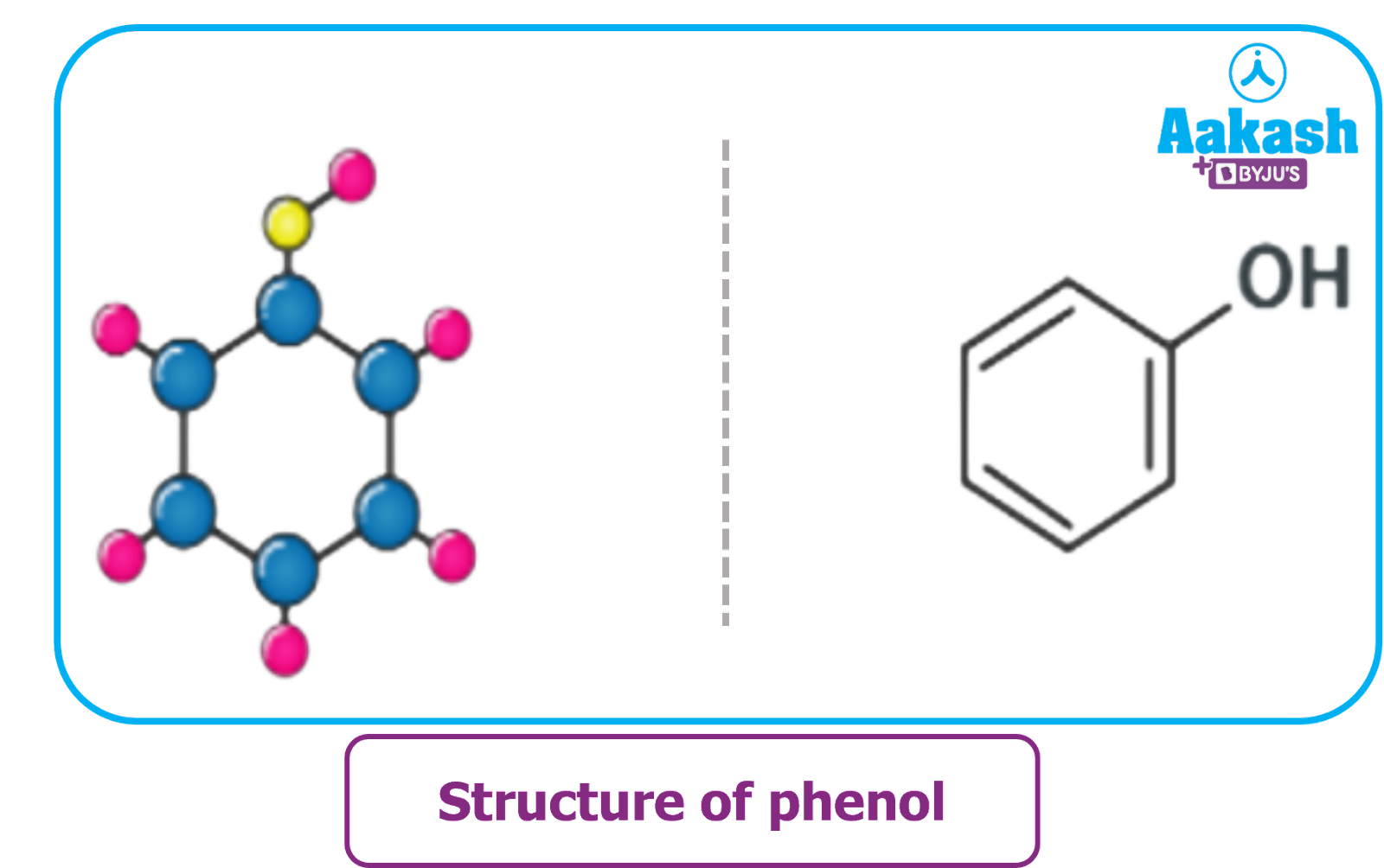
Structure of phenol
The organic compound phenol is formed when an aromatic benzene ring and a -OH group, also known as a hydroxyl group, are joined. It has the chemical formula C6H5OH or PhOH and is an aromatic organic molecule.
All the carbon atoms in phenol's aromatic ring are sp2 hybridised. Therefore, phenyl has a 120obond angle hexagonal planar structure with delocalized pi-electrons distributed throughout the ring.
Preparations of phenols:
Phenol can be synthesised by the following methods.
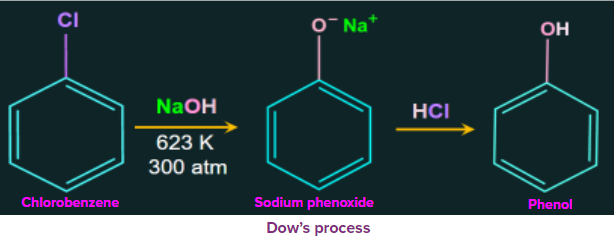
1. Dow’s process:
Dow’s process is a method to prepare phenol. The reactant chlorobenzene is heated with aqueous sodium hydroxide at temperatures of 623 K and 300 atm to get sodium phenoxide ion. Then in the next step sodium phenoxide ion is treated with dil. HCl which gives phenol as the final product.
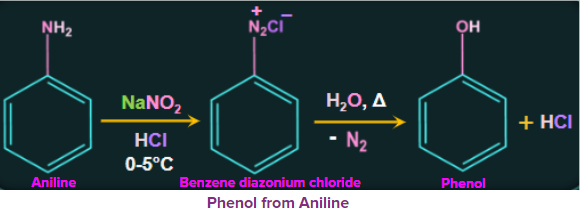
2. From Aromatic primary amines:
Treatment of aromatic primary amines at 273-278 K with freshly prepared nitrous acid
(NaNO2+HCl) gives the diazonium salt. These diazonium salts are inherently very reactive. When heated with water, these diazonium salts eventually hydrolyze to phenol. Phenol can also be obtained from diazonium salts by treatment with dilute acids.
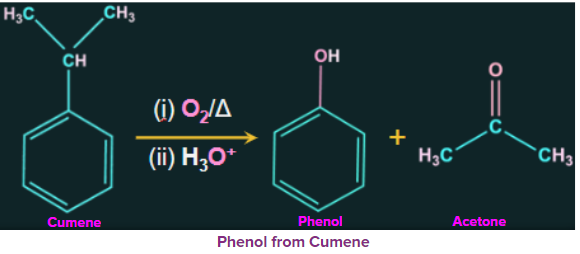
3. Phenol production from cumene:
Cumene is an organic compound obtained by Friedel-Crafts alkylation of benzene with propyl chloride. Cumene hydroperoxide is obtained from the oxidation of cumene (isopropylbenzene) in the presence of air. Further treatment of cumene hydroperoxide with dilute acid gives phenol. Acetone is also produced in large quantities as one of the by-products of this reaction. Therefore, the phenol produced by these processes needs to be purified.
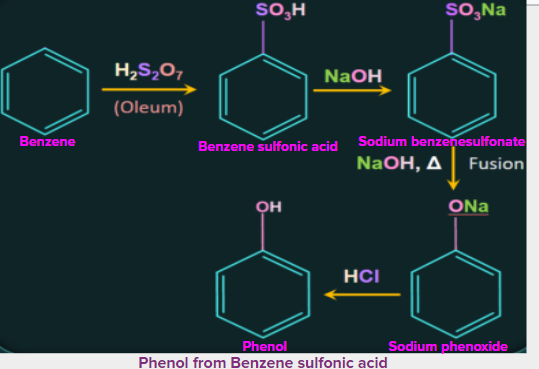
4. Production of phenol from benzenesulfonic acid:
Benzenesulfonic acid can be obtained from benzene by reaction with oleum. This benzenesulfonic acid can be treated with molten sodium hydroxide at high temperatures to promote the formation of sodium phenoxide. Finally, sodium phenoxide produces phenol during acidification.
Properties of phenol:
|
Phenol |
C6H5OH |
|
Molecular Weight/ Molar Mass |
94.11 g mol-1 |
|
Density |
1.07 g cm-3 |
|
Melting Point |
40.5 oC |
|
Boiling Point |
181.7 oC |
1. Boiling point of phenol
Phenol generally has a higher boiling point compared to other hydrocarbons of the same molecular weight. This is because there are intermolecular hydrogen bonds between the hydroxyl groups of the phenol molecule.
2. Solubility of phenol
The solubility of phenol in water depends on the hydroxyl groups present. The hydroxyl groups of phenol are involved in the formation of intermolecular hydrogen bonds. Hydrogen bonds are formed between water and phenol molecules, making phenol water-soluble. However, the aryl group attached to the hydroxyl group is hydrophobic in nature. Therefore, as the size of the aryl group increases, the solubility of phenol decreases.
3. Acidity of phenol
Phenol reacts with active metals such as sodium and potassium to form the corresponding phenoxide. These reactions of phenol show its acidic nature. In phenol, the sp2 hybrid carbon of the benzene ring is directly attached to the hydroxyl group and functions as an electron-withdrawing group. Therefore, the electron density of oxygen decreases.
Phenoxide ions are more stable than alkoxide ions because the negative charge of the oxygen is delocalized over the benzene ring. As a result, phenol is more acidic than alcohol. In substituted phenols, the acidity decreases when an electron-donating group is attached to the ring, and increases when an electron-withdrawing group is attached.
Chemical reactions of phenol:
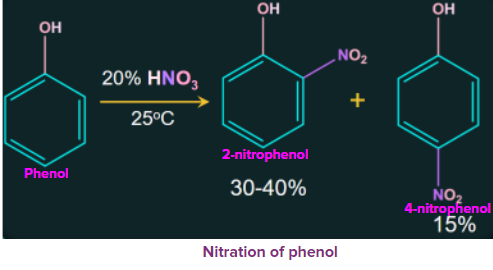
1. Nitration of phenol
Phenol is nitrated when treated with dilute nitric acid at a low temperature (298 K) to give a mixture of ortho-nitrophenol and para-nitrophenol. Due to its volatility, the resulting mixture is further separated into ortho-nitrophenol and para-nitrophenol by steam distillation.
Due to the absence of intermolecular hydrogen bonds, ortho nitrophenols are more volatile than para nitrophenol, which contain only intermolecular hydrogen bonds, while ortho nitrophenols contain intramolecular hydrogen bonds.
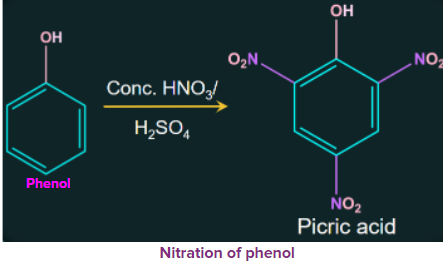
When phenol is treated with concentrated nitric acid, the nitration results in the formation of 2, 4, 6-trinitrophenol (commonly called picric acid) along with many oxidised products.
2. Halogenation of Phenols
Due to the strong activating effect of the hydroxyl groups in phenol, they are halogenated in the absence of Lewis acid. Treatment of phenol with bromine at low temperatures in the presence of low polar solvents such as CS2 or CHCl3 results in the formation of mono bromophenol.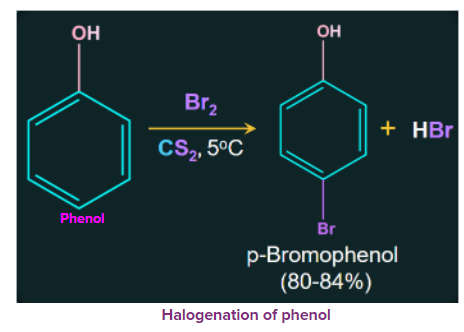
A white precipitate of 2, 4, 6-tribromophenol is formed when phenol is treated with bromine water.
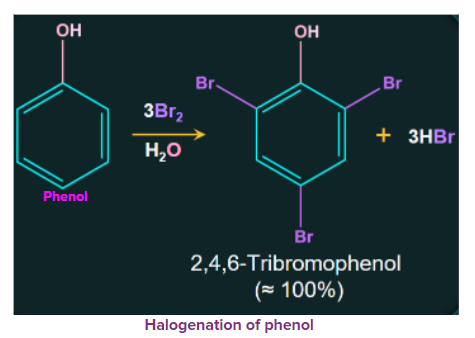
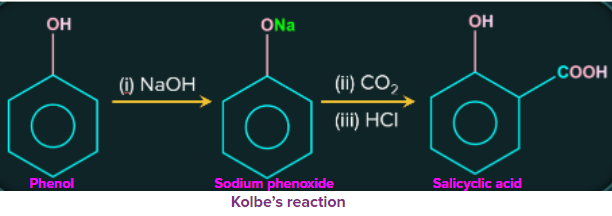
3. Kolbe's reaction
Treatment of phenol with sodium hydroxide produces phenoxide ions. The formed phenoxide ions are highly reactive to electrophilic substitution reactions and then treatment with a weak electrophile (carbon dioxide) causes an electrophilic substitution reaction to form ortho hydroxybenzoic acid followed by acidification. This reaction is commonly known as the Kolbe reaction.
Anisole:
Anisole, also known as methoxybenzene, is a chemical compound with the formula C6H5OCH3. It is a colourless liquid with an anise seed-like odour, and many of its derivatives can be found in natural and synthetic fragrances. The compound is primarily synthesised and serves as a precursor to other synthetic compounds. It comes under the family of ether.

Preparations:
1. Williamson ether synthesis:
Phenol on reaction with sodium hydroxide gives phenoxide ion and that phenoxide ion reacts with methyl iodide via williamson ether synthesis to give anisole. The reaction is as follows:
2. By methylation of sodium phenoxide:
Sodium phenoxide on reaction with dimethyl sulphate gives anisole.
Chemical reactions of anisole:
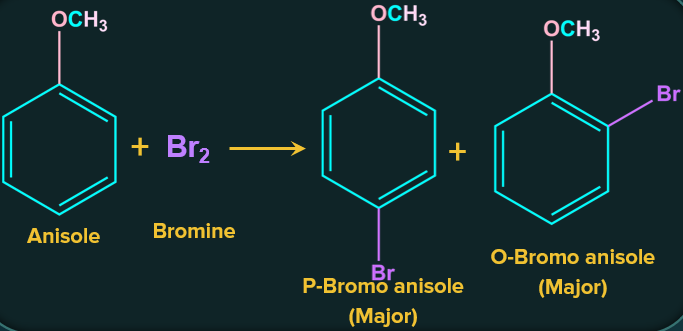
1. Halogenation reaction of anisole:
Bromination of anisole with bromine results in a mixture of ortho and para bromo anisole. It exemplifies the electrophilic aromatic substitution reaction. The bromine atom replaces the H atom attached to the benzene ring. An ortho para directing group is the methoxy group.
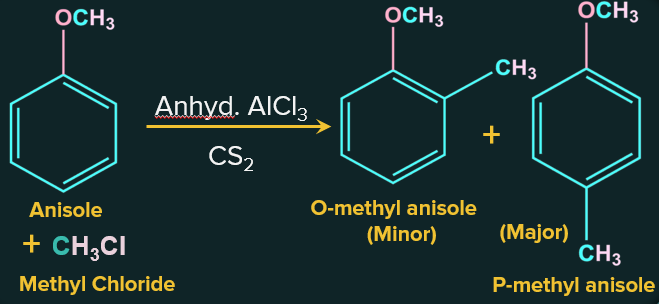
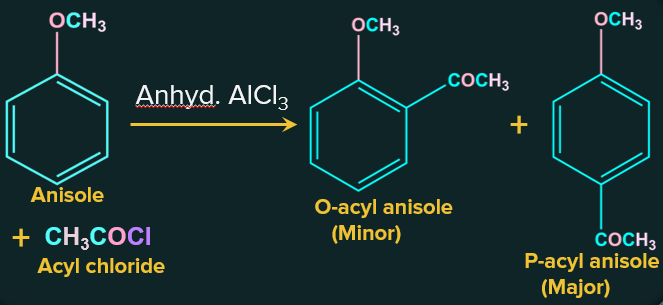
2. Friedel-Crafts Reaction of anisole:
The Friedel-Crafts reaction includes adding alkyl and acyl groups at ortho and para positions in anisole via reactions with an alkyl halide and an acyl halide in the presence of catalyst as anhydrous aluminium chloride (a Lewis acid).
Practice problems:
Q.1. The boiling points of four compounds with similar molecular masses, A, B, C, and D, were determined. Compound C was discovered to have the highest boiling point of the four. What is the most likely compound C?
(A) Hydrocarbon
(B) Haloalkane
(C) Alcohol
(D) Ether
Answer: (C)
Solution: Alcohols will almost certainly have the highest boiling point of compounds with similar molecular masses due to the presence of intermolecular hydrogen bonding, which is absent in case of hydrocarbons, haloalkanes and ethers.
Q.2. Which of the following oxidising agents is used to directly produce carboxylic acids from alcohols?
(A) Acidified KMnO4
(B) Aqueous KMnO4
(C) Alkaline KMnO4
(D) Anhydrous CrO3
Answer: (A)
Solution: Strong oxidising agents, such as acidified potassium permanganate or acidified potassium dichromate, directly convert alcohol to carboxylic acid. However, using CrO3 as the oxidising agent in an anhydrous medium yields only aldehyde.
Q.3. The distillation process used to separate ortho and para isomers of nitrophenol is
(A) fractional
(B) steam
(C) vacuum
(D) zone
Answer: (B)
Solution: o-Nitrophenol is steam volatile due to intramolecular hydrogen bonding between O of nitro group and H hydroxyl group. p-Nitrophenol, on the other hand, is present as associated molecules due to intermolecular hydrogen bonding, making it less volatile.Consequently, steam distillation can be used to separate a mixture of both.
Q.4.Which of the following has the lowest solubility in water?
(A) n-Butyl alcohol
(B) n-Pentyl alcohol
(C) n-Hexyl alcohol
(D) n-Heptyl alcohol
Answer: (D)
Solution: As the size of the alkyl group increases, it prevents the formation of hydrogen bonds with water molecules, and thus the solubility decreases as the compound's size or mass increases.
Frequently asked questions:
1. Explain why nucleophilic substitution reactions are not very common in phenols?
Answer: The -OH group in phenols is a powerful electron donor. As a result, the electron density on the benzene ring is quite high, repelling nucleophiles. In other words, because nucleophiles cannot attack the benzene ring, phenols rarely undergo nucleophilic substitution reactions.
2. Alcohols react with active metals such as Na,K etc to form alkoxides. Write down the decreasing order of sodium metal's reactivity towards primary, secondary, and tertiary alcohols.
Answer: An electron releasing group causes the +I effect by increasing the electron density on oxygen while decreasing the polarity of the O-H bond. As the number of alkyl groups increases from primary to tertiary alcohols, the electron density on the O-H bond increases.
3. When transitioning from primary to tertiary alcohols, the polarity decreases and the strength of the O-H bond increases.
Answer: Sodium metal, as we all know, is basic in nature, whereas alcohols are acidic. As a result, as the acidic strength decreases, so does the reactivity of alcohol with sodium metal. As a result, 1>2 >3 is the correct sequence.
4. Which of the two nitrophenols, o-nitrophenol or p-nitrophenol, is more volatile? Explain.
Answer: o-Nitrophenol molecules form intramolecular H-bonds, whereas p-Nitrophenol molecules form intermolecular H-bonds. The strong intermolecular H-bonding increases the boiling point during boiling, but intramolecular H-bonding cannot. As a result, o-Nitrophenol is a higher steam volatile than p-Nitrophenol.
5. Which is more acidic, 2-chloroethanol or ethanol, and why?
Answer: 2-chloroethanol has a higher acidity than ethanol. The electron density in the O-H bond decreases due to the -I effect (electron withdrawing group) of the Cl-atom. As a result, the O-H bond of 2-chloroethanol becomes weaker than the O-H bond of ethanol. As a result, 2-chloroethanol has a higher acidity than ethanol.


