-
Call Now
1800-102-2727
Hydrohalic Acids– Definition, Properties, Acidic Strength, Thermal Stability, Practice Problems & FAQs
A strong attraction can always be sensed between the tiny bees and the flowers. It's wonderful how bees collect nectar and pollen from flowers and fulfil their mutual requirements. Nectar as well as pollen are very much essential for the nutrition of bees. While the pollen offers protein and other nutrients, the nectar is used for energy. Bees carry pollen from plant to plant, providing the pollination services required by plants and by nature as a whole. Bees consume the majority of pollen as nourishment for their larvae.

So in a way, they both compliment each other! Just as does the tiny hydrogen atom with various halogen atoms! Hydrogen gives its single electron to halogen atoms and they both form compounds i.e., hydrogen halides. This compound on dissolution in water forms hydrohalic acid.
Let us learn more about them!
Table of Content
- Hydrohalic Acid–Introduction
- Preparation of Hydrohalic Acids
- Physical Properties of Hydrohalic Acids
- Chemical Properties of Hydrohalic Acids
- Thermal Stability & Acidity of Hydrohalic Acids
- Practice Problems
- Frequently Asked Questions-FAQs
Hydrohalic Acids-Introduction
Hydrohalic acids are basically aqueous solutions of hydrogen halides represented by the general formulaHX, where X= F, Cl, Br, I. In chemistry, hydrogen halides, which are called hydrohalic acids in their aqueous solutions, act as Arrhenius acids, and also are diatomic inorganic compounds.
So, hydrohalic acids are Arrhenius acids which are basically defined as hydrogen-containing substances which can produce protons or H+ ions when they dissociate in water. At the Standard Temperature and Pressure, all known hydrogen halides are gases.
Some features of hydrogen halide:
- The diatomic hydrogen halides have little tendency to ionise in the gas phase (although liquified hydrogen fluoride is a polar solvent).
- Hydrochloric acid and hydrogen chloride are distinguished by chemists.
- At room temperature, the hydrogen halides are gas that produces hydrohalic acids when they interact with water.
Natural Occurrence: Hydrogen fluoride, hydrogen chloride, and hydrogen bromide are examples of hydrogen halides that come within the category of volcanic gases. Volcanic gases include those that are dissolved in lava and those that are encased in vesicles in volcanic rocks. These gases are present in the atmosphere, crust, and mantle of the planet.
Hydrochloric acid is present in the stomach. Hydrogen chloride serves as the main building block of stomach acid which is a digesting liquid.
Preparation of Hydrohalic Acids
- Fluorine oxidises water to oxygen, whereas chlorine and bromine react with water to form corresponding hydrohalic and hypohalous acids.
[Where X= Cl, Br, I]
- Hydrogen fluoride and hydrogen chloride are produced by the direct interaction of hydrogen with fluorine and chlorine, respectively. Dissolution of the produced gas into water forms corresponding hydrohalic acid.
- Industrially these gases are, however, created by the treatment of halide salts with sulfuric acid. Example– formation of hydrochloric acid.
- When hydrogen and bromine are mixed at high temperatures using a platinum catalyst, hydrogen bromide is created.
![]()
- Iodine reacts with hydrogen sulphide or hydrazine to form the least stable hydrogen halide, known as HI. Dissolving the same in water form hydroiodic acid.
Physical Properties of Hydrohalic Acids
- At standard temperature and pressure (STP), the hydrogen halides are colourless gases with the exception of hydrogen fluoride, which boils at 19 °C.
- Since hydrogen fluoride is the only hydrogen halide that shows hydrogen bonding, it has the highest melting and boiling temperatures in the HX series. Hydrogen fluoride condenses under cool conditions. They establish the hydrogen bond, which causes them to have an anomalous boiling point
- Rising trend is seen in the boiling point from HCl to HI. Van der Waals forces between molecules are said to be getting stronger from HCl to HI. Boiling point of HF, HCl, HBr, HI are 293 K, 188K, 207 K, 237.5 K respectively.
- Melting point of HF, HCl, HBr, HI are 189 K, 159K, 186 K, 222 K respectively.
- Bond dissociation enthalpy decreases down the group as bond length increases down the group from HF to HI.
|
Bond dissociation enthalpy (kJ mol-1) |
570 |
432 |
366 |
298 |
|
Bond length (pm) |
92 |
127.5 |
141.5 |
161 |
- Solutions of concentrated hydrohalic acid emit audible and visible white vapours. This mist is created when small droplets of the hydrohalic acid's concentrated aqueous solution develop.
Chemical Properties of Hydrohalic Acids
- The corresponding hydrohalic acids are produced when the hydrogen halides dissolve in water, which is strongly exothermic. These acids have a high ionic strength due to their propensity to form hydronium ions (H3O+) in aqueous solutions.
HCl (g)+ H2O (l) H3O+ (aq) + Cl- (aq)
- However, the hydrogen halides are only slightly acidic when they are dissolved in non-aqueous solvents like acetonitrile.
- Being acidic in nature, hydrohalic acids react with bases to form salt and water.
HCl (aq)+NaOH (aq) NaCl (aq)+H2O (l)
- Being acidic in nature, hydrogen halides when reacting with ammonia (and other bases), it generates ammonium halides.
HX + NH3 → NH4X [Where X= Cl, Br, I]
- Halocarbons are made via the hydrohalogenation process in organic chemistry. For instance, the hydrochlorination of ethylene yields chloroethane.
C2H4(g)+ HCl (g)→ CH3CH2Cl (g)
Thermal Stability and Acidic Strength of Hydrohalic Acids
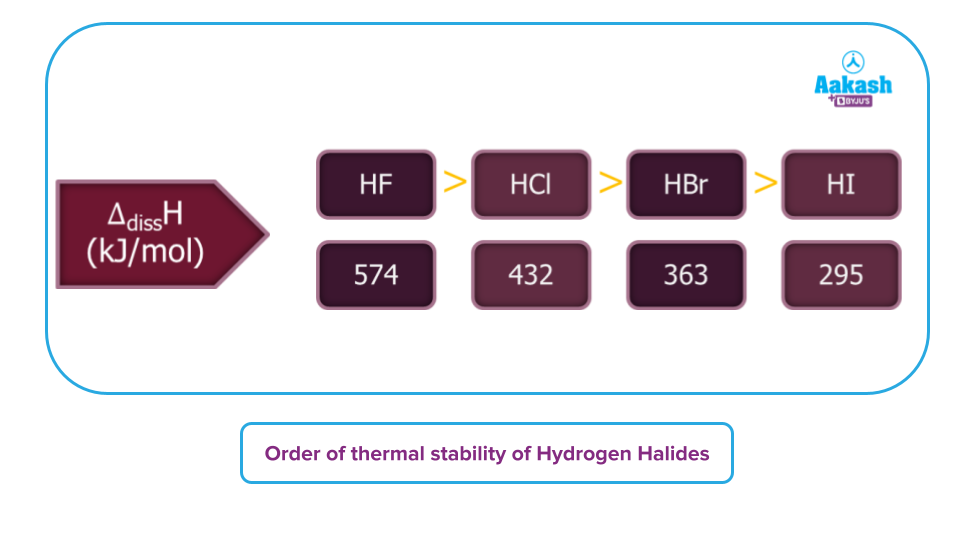
Thermal Stability: As is well-known, the temperature may often have an impact on thermal stability, which demonstrates how an electric current/heating can disrupt the chemical bonds present in any chemical compound.
Combining hydrogen with halogens results in the chemical known as hydrogen halides. And their aqueous solution is termed hydrohalic acids. The thermal stability of the hydrogen halides reduces as we advance down the group in the periodic table because the bond strength weakens. Consequently, the sequence of hydrogen halides' thermal stability is as follows:
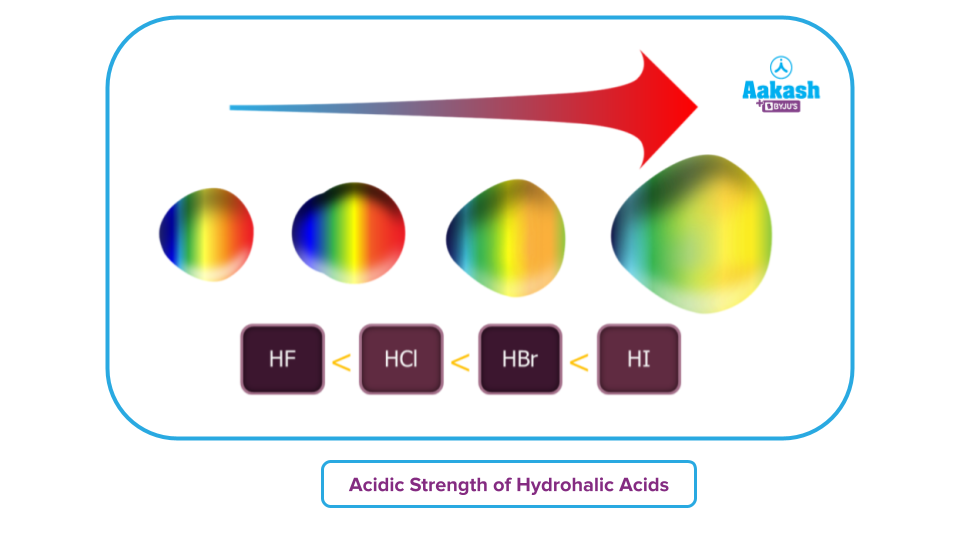
Acidic Strength: It is difficult for a weak acid to contribute its hydrogen ions. The size of the atom that is connected to hydrogen determines how powerful the acid is since a stronger connection results from smaller atoms attaching to hydrogen.
Hydrohalic acids are more acidic in nature if hydrogen-to-other-atom bonds are longer i.e., the weaker the bond between hydrogen and halogen, the more acidic will be the hydrohalic acid. Iodine has the highest acidic strength of all the halogens because of its larger size, which causes its bond length to be longer. Bond strength diminishes across the group as bond (H-X) dissociation enthalpy lowers.
So, the acidic strengths vary in the following order: HF, HCl, HBr, and HI.
Practice Problems
Q.1. Mention the one that has the highest dipole moment?
a. HCl
b. HF
c. HI
d. HBr
Answer: (B)
Solution:
Since Hydrogen and fluorine, have the highest electronegativity difference, so HF has the highest dipole moment. So option B is the correct answer.
Q.2. Which hydrohalic acid is the most reducing in nature?
a. HCl
b. HF
c. HI
d. HBr
Answer: (C)
Solution:
Stability of hydrogen halides decreases down the group due to the increase in the atomic size of the halide ions. And hence hydrogen can be readily removed as we go downwards the group. So the most reducing would be the hydroiodic acid i.e., HI. So option C is the correct answer.
Q.3. Aqueous solution of HF is weakly acidic but the concentrated solution is strongly acidic. Give a reason.
Answer: Aqueous solution of HF is known as hydrofluoric acid. In contrast to the other hydrohalic acids, hydrofluoric acid acts weakly acidic when diluted due to the creation of hydrogen-bonded ion pairs (H3O+).F-.
Concentrated solutions, however, are strong acids because bifluoride anions, rather than ion pairs, predominate. Self-ionisation happens in anhydrous HF in liquid form. This forms an extremely acidic liquid.
3HF ⇌ H2F+ + HF2-
Q.4. Which is the strongest hydrohalic acid?
a. HI
b. HBr
c. HF
d. HCl
Answer: (A)
Solution:
Because the dissociation energy of the H-I bond is the lowest among all the given hydrohalic acid, owing to greater atomic radii of I-. So, it is easier for hydroiodic acid to dissociate in water and furnish H+ ions. Hence, HI is the strongest acid among all the given options. So, option A is the correct answer.
Frequently Asked Questions–FAQs
Q.1. What is the difference between hypohalous acid and hydrohalic acid?
Answer: The inorganic compounds known as hydrogen halides (or hydrohalic acids) have the formula HX, where X is one of the halogens, including fluorine, chlorine, bromine and iodine. Gases called hydrogen halides dissolve in water to produce hydrohalic acids.
The oxyacids of halogens with the general chemical formula HOX, where X is the halogen atom, are known as hypohalous acids. Example- HOF, HOCl, HOBr, HOI.
Fluorine is able to oxidise H2O to oxygen, whereas Cl2 and Br2 react with water to form, corresponding hypohalous acids and hydrohalic acid.
Q.2. Is HF a hydrohalic acid?
Answer: Yes. It is a polar molecule and readily dissolved in water to produce aqueous solutions of hydrogen fluoride, which is thus called hydrofluoric acid (a hydrohalic acid). Although this is a weak acid in an aqueous solution due to its capability to form hydrogen bonding with water, making it reluctant to lose H+ ions.
Q.3. Are hydrogen halides and hydrohalic acids the same?
Answer: The chemical formula representing a hydrogen halide and a hydrohalic acid of a specific halogen, is the same. But the key difference is that hydrohalic acids are the aqueous solutions of hydrogen halides. For example, hydrochloric acid is a hydrohalic acid formed when hydrogen chloride is dissolved in water. Both are expressed with the formula HCl.
Q.4. What is the significance of hydrohalic acids?
Answer: Alkyl iodides are produced when hydrohalic acids undergo an addition reaction with alkenes. So these acids are important for hydrohalogenation or sometimes halogenation reactions. Also, it can be employed as a reducing agent to convert aromatic nitro compounds into anilines.
Other than this, specific hydrohalic acids have several important roles to play. For example- In the stomach, hydrochloric acid is a necessary acid. It aids in the eradication of dangerous bacteria and other species. It also functions as a food preservative. Pepsinogen, which is not active, is changed into HCl into pepsin, which is then used by our stomach to digest proteins.


