-
Call Now
1800-102-2727
Understanding Hybridisation of XeF₂: Xenon Difluoride
Xenon Difluoride (XeF₂) is also known as Difluoroxenon. It has one xenon atom bonded to two fluorine atoms. It’s an excellent example of sp³d hybridisation in inorganic chemistry.
Let us understand how hybridisation happens in XeF₂. Read on to learn how it leads to its bonding and molecular shape.
What is the Hybridisation of XeF₂?
Xenon Difluoride consists of one central xenon atom (a noble gas), which is bonded to fluorine atoms and has three lone pairs left. Since xenon has 5 regions of electron density (2 bonding pairs + 3 lone pairs), it undergoes sp³d hybridisation and follows the principle of expanded octet (as d orbitals are available in the valence shell).
Using the Hybridisation Formula
We can determine the hybridisation of xenon difluoride using a simple formula:

Step-by-step calculation:
- Valence electrons of xenon (Xe) = 8
- Monovalent atoms (fluorine = 2) = 2
- Negative charge = 0
- Positive charge = 0
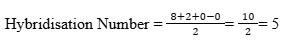
Interpretation:
- Hybridisation number = 5 → sp³d
- Geometry (according to VSEPR) = Trigonal bipyramidal electronic geometry
- Actual shape = Linear (since 3 lone pairs occupy the equatorial positions, forcing the 2 fluorine atoms to remain opposite each other)
- Bond angle = 180°
Breakdown of XeF₂ Hybridisation
XeF₂ has a trigonal bipyramidal electron geometry, but it acquires a linear molecular geometry. The lone pairs take up equatorial positions, leading the fluorine atoms to take the axial positions. The VSEPR Theory explains all this.
Here is a complete understanding of its hybridisation.
Electronic Configuration of Xenon
The atomic number of xenon is 54.
The ground state of xenon :
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶
In the ground state, xenon has a complete octet (all paired electrons in 5p orbitals), so it normally does not form bonds.
Excited state configuration:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁵ 5d¹
Now, five unpaired electrons → enough to form bonds and still have space for lone pair of electrons.

Ground state vs excited state orbital diagram
Formation of Hybrid Orbitals
sp³d hybridisation occurs when 1 s orbital, 3 p orbitals, and 1 d orbital mix to form equivalent hybrid orbitals.
The result:
→ 5 sp³d hybrid orbitals are formed
→ These accommodate lone pairs and are used to form bonds between atoms.
Bond Formation in Xenon Difluoride
The xenon atom uses:
- 2 sp³d hybrid orbitals overlap with the p orbitals in the fluorine atom and form 2 σ bonds.
- The remaining 3 sp³d hybrid orbitals contain lone pairs of electrons.
Result:
- The molecule adopts a linear structure in order to minimise lone pair repulsion.
- Hybridisation type: sp³d
- Bond angle: 180° (F–Xe–F)
- Geometry: Linear

Geometry of Xenon Difluoride
Details At A Glance
| Property | Details |
|---|---|
| Molecule | Xenon Difluoride (XeF₂) |
| Hybridisation | sp³d |
| Geometry | Linear |
| Bond angle | 180° |
| Bonding | 2 σ bonds (Xe–F), 3 lone pairs |
| Unhybridised Orbitals | 0 (all involved in hybridisation) |
| Xenon valency satisfied? | Yes, by forming 2 bonds and holding 3 lone pairs |
Formal Charge in XeF₂
To determine if the Lewis structure of XeF₂ is stable, we calculate the formal charge on each atom using the formula:
Formal charge = Valence electrons – (Lone pair electrons + ½ × Bonding electrons)
Step-by-step for each atom:
Xenon (Xe) :
- Valence electrons: 8
- Lone pairs: 3 (6 electrons)
- Bonding electrons: 4 (2 Xe–F bonds)
Formal charge = 8 – (6 + ½ × 4) = 8 – (6 + 2) = 8 – (8) = 0
Fluorine (F) – each
- Valence electrons: 7
- Lone pairs: 3 (6 electrons)
- Bonding electrons: 2 (1 single bond with Xe)
Formal charge = 7 – (6 + ½ × 2) = 7 – (6 + 1) = 7 – (7) = 0
Thus, all atoms in XeF₂ carry zero formal charge, which confirms that the Lewis structure is stable and correct.
Summing Up
Xenon in XeF₂ forms 2 σ bonds and ends up with 3 lone pairs. The molecule follows the concept of an expanded octet and thus ends up undergoing sp³d hybridisation. XeF₂ has Linear molecular geometry and bond angles of 180° (due to lone pair repulsion).
Frequently Asked Questions
Q1. Why does xenon undergo hybridisation in XeF₂?
To form 2 single bonds with fluorine and also accommodate 3 lone pairs, xenon hybridises into sp³d orbitals.
Q2. How many σ and π bonds are present in XeF₂?
There are 2 σ bonds and 0 π bonds in total.
Q3. What is the shape of Xenon Difluoride?
Linear, due to 2 bonding pairs and 3 lone pairs.
Q4. Is XeF₂ polar or non-polar?
Non-polar, as the molecule has a symmetrical linear shape due to bond dipoles.
Q5. What are some uses of Xenon Difluoride in our daily lives?
Xenon Difluoride is used as a fluorinating agent in the process of chemical synthesis. It is also used in semiconductor etching and as a mild oxidising agent.



