
Understanding Hybridisation of BrF₃: Bromine Trifluoride
Bromine trifluoride (BrF₃) is a pale yellow liquid used as a powerful fluorinating agent. BrF₃ has 5 electron domains around the central atom Bromine, including lone pairs. Therefore, it’s an excellent example of sp³d hybridisation with a T-shaped molecular geometry.
Let’s go through the hybridisation of BrF₃ in a stepwise manner.
What is the Hybridisation of BrF₃?
Hybridisation is the concept of mixing atomic orbitals to form new hybrid orbitals that help explain the geometry of molecules.
Using the Hybridisation Formula
We can determine the hybridisation using the simple formula:

Step-by-step calculation:
- Valence electrons of central atom (Bromine) = 7
- Number of monovalent atoms (Fluorine atoms) = 3
- Negative charge = 0
- Positive charge = 0
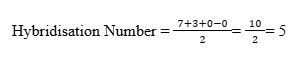
Interpretation:
A hybridisation number of 5 corresponds to sp³d hybridisation.
In sp³d hybridisation, one s, three p, and one d orbital mixup together and redistribute their energies. Thus a set of 5 orbitals of equal energy is obtained.
In BrF₃, bromine forms three bonds with fluorine atoms and holds two lone pairs.
Step-by-Step Understanding of BrF₃ Hybridisation
For a better understanding, let’s start with the electronic configuration and go on to understand the redistribution of electrons.
Ground State Electronic Configuration of Bromine
The Atomic Number of Bromine (Br) of is 35.
It’s ground state configuration:
[Ar] 4s² 3d¹⁰ 4p⁵
In the ground state, bromine has 7 valence electrons.
When bonding with fluorine atoms, bromine excites one electron to a 4d orbital.
Excitation
The excited state configuration:
4s² 4p⁴ 4d¹
Now, five orbitals are available for hybridisation:
- 1 from 4s
- 3 from 4p
- 1 from 4d → sp³d hybridisation

Ground state of bromine
Hybridisation
Bromine undergoes sp³d hybridisation:
- Orbitals involved: 1s + 3p + 1d
- Total hybrid orbitals: 5 sp³d hybrid orbitals
- Electron domains: 5 (3 bond pairs + 2 lone pairs)
So, out of these:
- 3 are used for bonding with fluorine atoms
- 2 remain as lone pairs

Orbital diagram showing sp³d hybridisation
Geometry of BrF₃: T-Shaped
According to VSEPR theory, 5 electron pairs around the central atom would ideally form a trigonal bipyramidal geometry. But due to lone pair–bond pair repulsion, the shape distorts.
- The two lone pairs occupy equatorial positions (to minimise repulsion)
- The three fluorine atoms occupy two axial and one equatorial position
- This leads to a T-shaped molecular geometry
| Property | Description |
|---|---|
| Shape (Geometry) | T-Shaped |
| Hybridisation | sp³d |
| Bond Angle | ~86.2° (slightly < 90°) |
| Central Atom | Bromine |
| Peripheral Atoms | 3 Fluorine atoms |
| Lone Pairs on Br | 2 |
| Bond Type | σ (sigma) bonds |
| Polarity | Polar molecule |

T-shaped molecular geometry of BrF₃ showing lone pairs at equatorial positions
Formal Charges in BrF₃
Let’s check if the molecule is stable by calculating the formal charge.
Bromine:
- Valence electrons = 7
- Bonding electrons = 6 (3 bonds)
- Lone pair electrons = 4
Formal Charge = 7 – (4 + ½ × 6)
= 7 – (4 + 3)
= 0
Each Fluorine:
- Valence electrons = 7
- Bonding = 2 (1 bond)
- Lone pairs = 6
Formal Charge = 7 – (6 + 1) = 0
The formal charge on all atoms is 0, making BrF₃ a stable structure.
Summing Up
BrF₃ undergoes sp³d hybridisation and forms 3 sigma bonds with fluorine atoms. It has 2 lone pairs on bromine, leading to a T-shaped molecular structure. The lone pairs occupy equatorial positions due to VSEPR minimisation of repulsion. The molecule is polar due to its asymmetric shape and polar Br–F bonds.
Frequently Asked Questions
Q1. Why are the lone pairs placed in equatorial positions?
Lone pairs occupy equatorial positions to minimise repulsion, as equatorial–equatorial repulsion is less than axial–equatorial.
Q2. Is BrF₃ polar or non-polar?
Polar, due to the asymmetrical shape and the presence of lone pairs.
Q3. How many sigma bonds and lone pairs are in BrF₃?
3 sigma bonds with fluorine atoms and 2 lone pairs on the central atom.
NEET Related Links
JEE MAIN Related Links
JEE Main marks vs rank vs percentile
JEE Advanced Related Links
JEE Advanced Eligibility Criteria
JEE Advanced Chemistry Syllabus
JEE Advanced Registration Dates

