-
Call Now
1800-102-2727
Hybridisation of BCl3: Boron Trichloride
Boron Trichloride (BCl₃) is a colourless gas. It has a pungent odour and finds application in various industries, including semiconductor manufacturing. BCl₃ is one of the best examples to understand sp² hybridisation. The molecule has a boron (B) as its central atom and three chlorine (Cl) atoms.
Let’s understand the hybridisation of BCl₃ in the easiest way possible.
What is the Hybridisation of BCl₃?
To begin with, let’s understand the term. Hybridisation is the mixing of atomic orbitals to form new, equivalent orbitals that determine the molecule’s geometry.
Using the Hybridisation Formula
We can determine the hybridisation of BCl₃ using the simple formula:

Step-by-step calculation:
- Valence electrons of central atom (Boron) = 3
- Number of monovalent atoms (Chlorine atoms) = 3
- Negative charge = 0
- Positive charge = 0
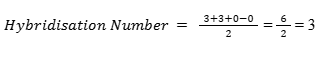
Interpretation: A hybridisation number of 3 corresponds to sp² hybridisation.
In sp² hybridisation, one s and two p orbitals of an atom mix together and redistribute their energies. Due to this redistribution, three hybrid orbitals of equal energy and identical shape are formed.
Here is a step-by-step breakdown of BCl₃ sp² hybridisation:
Electronic Configuration of Boron
Boron’s atomic number = 5
Ground state configuration:
1s² 2s² 2p¹
Only one unpaired electron → not enough for 3 bonds!
So, boron promotes one electron:
Excited state configuration:
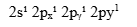
Now, 3 unpaired electrons are available for bonding. The following orbital diagram will help you understand the ground state vs excited state electronic configuration of boron.

Ground state vs excited state of boron
Formation of Hybrid Orbitals
To bond with three chlorine atoms, Boron undergoes sp² hybridisation:
- Orbitals involved: 1 s + 2 p
- Result: 3 half-filled equivalent sp² hybrid orbitals
New electronic configuration:
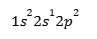
More specifically, the distribution in the p orbit will be:
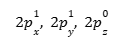

orbital diagram for hybridised state
Each orbital overlaps with the lone pair of Cl to form a sigma (σ) bond, i.e., three σsp-p bonds are formed between B half-filled sp² hybrid orbitals and three Cl atoms.
Formal Charge in Boron Trichloride
Boron is present in Group 13 of the periodic table. Chlorine is present in Group 17 of the periodic table. Thus, boron and chlorine have 3 and 7 electrons in the outermost shell, respectively. The structure of BCl3 comprises boron with three sigma bonds, each with a chlorine atom. It has zero formal charge, indicating the optimal Lewis structure of the compound.
Formal charge = Valence electrons of the atom - (Number of lone pair of electrons + 0.5 Number of bonding electrons )
Formal charge = 3-(0+0.56)
Formal charge = 3-3
Formal charge = 0
Geometry of BCl₃
The 3 sp² orbitals arrange themselves at 120° to minimise repulsion. The shape of the molecule is trigonal planar. The three Cl atoms arrange themselves symmetrically around the boron atom, and the trigonal planar shape is consistent with the VSEPR theory.

Sigma bonds in Boron Trichloride
| Property | Description |
|---|---|
| Shape | Trigonal Planar |
| Bond Angle | 120° |
| Central Atom | Boron |
| Peripheral Atoms | 3 Chlorine atoms |
| Bond Type | σ (sigma) bonds only |
| Octet of Boron | Incomplete (6 electrons) |
| Stability | Stable (electron-deficient compound) |
Summing Up
Boron trichloride is a covalent compound with sp² hybridisation. Boron has three valence electrons. Each chlorine atom provides one electron. In total, the molecule has six valence electrons. BCl₃ forms when boron uses three of its orbitals, one 2s and two 2p to bond with the three singly occupied 3p orbitals of chlorine atoms. The Lewis structure of boron trichloride shows three single bonds between the B and Cl atoms, resulting in a trigonal planar geometry and 120° bond angles.
Frequently Asked Questions
Q1. Why is BCl₃ stable even though Boron has only 6 electrons?
BCl₃ is electron-deficient. It has no lone pairs and no strong repulsion, so the structure is stable. Boron also tends to act as a Lewis acid.
Q2. Does BCl₃ show back bonding like BF₃?
Yes, to a lesser extent. The 3p orbital of Cl overlaps with the empty p orbital of B (pπ–pπ back bonding), but this effect is weaker than in BF₃ due to size mismatch.
Q3. Is BCl₃ polar or non-polar?
The B–Cl bonds are polar due to electronegativity difference, but the molecule is non-polar overall due to symmetrical shape (dipoles cancel out).
Q4. What formal charges are present on B and Cl in BCl₃?
In the Lewis structure of BCl₃, B carries a formal charge of +1. Every chlorine atom carries a formal charge of -1.
Q5. Which type of overlapping occurs in the orbitals of Boron Trichloride?
Head-to-head overlapping leads to sigma bond formation in boron trichloride.



