-
Call Now
1800-102-2727
Huckel's Rule of Aromaticity - Aromaticity, Huckel’s Rule of Aromaticity, Practice Problems and FAQs
Do you have chess-playing skills?
If so, could you please fill in the blanks?
As long as no piece is in his way and he moves one tile in any direction, then the player is.............
If one can move any number of tiles either straight or diagonally in any direction, then the player is.............

King and queen are the first and second, respectively.
Ah, the tendency is always to favor the queen. Right???
LOL, jokes aside. You can portray a player's behavior by having a basic understanding of the chess rules.
Huckel's rule of Aromaticity is a term for similar rules used in organic chemistry to describe the nature of cyclic molecules.
Let’s explore it!
TABLE OF CONTENT
- Aromaticity
- Aromatic, Non-Aromatic and Anti-Aromatic Compounds
- Huckel’s rules of aromaticity
- Practice Problem
- Frequently Asked Questions
Aromaticity:
Unsaturated double or triple bonds could be a sign of a molecule's weak point. However, the molecule acquires better stability and unique features when these multiple bonds take place in conjugation. Aromaticity is the name for such an array of -bonded electrons.
Aromaticity is a feature of cyclic compounds that results in increased molecule stability by having delocalized electrons in the orbitals and a planar structure.

Aromatic, Non-Aromatic and Anti-Aromatic Compounds:
Aromatic Compounds:
Aromatic compounds are defined as organic substances that exhibit -electron delocalization and follow Huckel’s rule of aromaticity. Non-Aromatic Compounds:
Non-Aromatic Compounds:
On the other hand, non-aromatic or aliphatic compounds are organic substances that do not show -electron delocalization or in cyclic form. In the given example, the delocalisation is not possible because the last transfer of electrons is not possible as carbon is sp3 hydribised and has a complete octet.

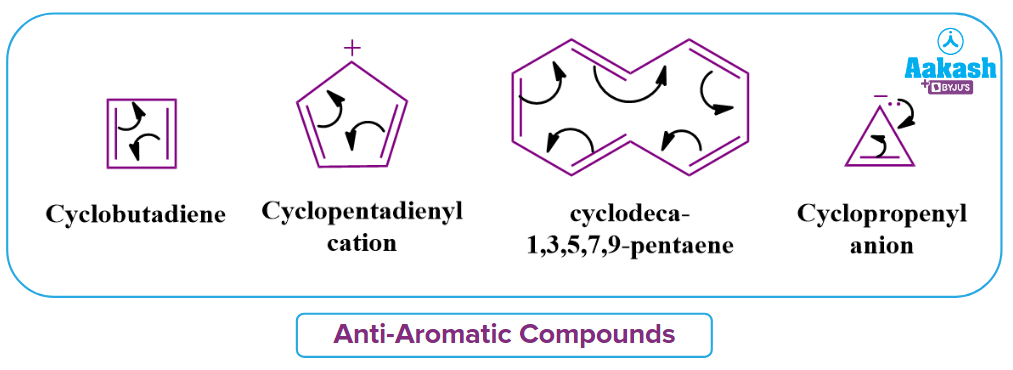
Anti-Aromatic Compounds:
Anti-Aromatic compounds are defined as organic substances that exhibit -electron delocalization but do not follow Huckel’s rule of aromaticity.
Huckel’s rules of aromaticity:
Let's examine the requirements that a compound must satisfy in order to show aromaticity.
The characteristics that decide whether a substance is aromatic are listed in the four following rules.
Rule 1: There must be a cyclic structure in the organic compound. Organic molecules that are linear and branching are considered acyclic, and they lack aromaticity.
Rule 2: The cyclic ring has to be planar. A p-orbital perpendicular to the ring should be present in every member of the aromatic ring.
Rule 3: All the -electrons must be in conjugation.
Rule 4: Huckel's rule must be followed by the organic compound. According to Huckel's rule of aromaticity, only the compound with (4n+2) electrons participating in conjugation exhibit aromaticity; the other compounds do not.
According to Huckel's rule of aromaticity, If the total number of (4n+2) electrons participating in conjugations is present in the given ring structure, they are aromatic compounds; where n is any natural number (0, 1, 2, 3, 4, and so on).
If all the three rules are followed by given compound but exhibit (4n) electrons participating in conjugation in the given ring structure rather than (4n+2) electrons, then the given compound is Anti-Aromatic in nature.
If any of the three rules are not followed by the given compound then the given compound is Non-Aromatic in nature.
Example:
1. Pyridine:
Consider the example of Pyridine. Let us discuss whether pyridine satisfies all the given four rules of aromaticity or not.
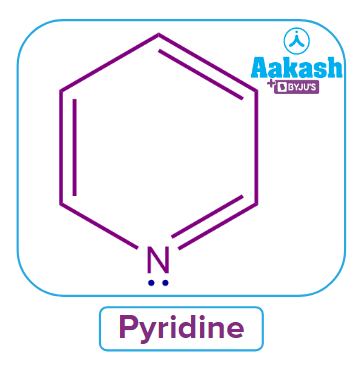
Yes, Pyridine has a cyclic structure and a planar molecule with sp2 hybridized atoms and has a hexagon-like planar shape. Hence, it follows Rule-1 and Rule-2.
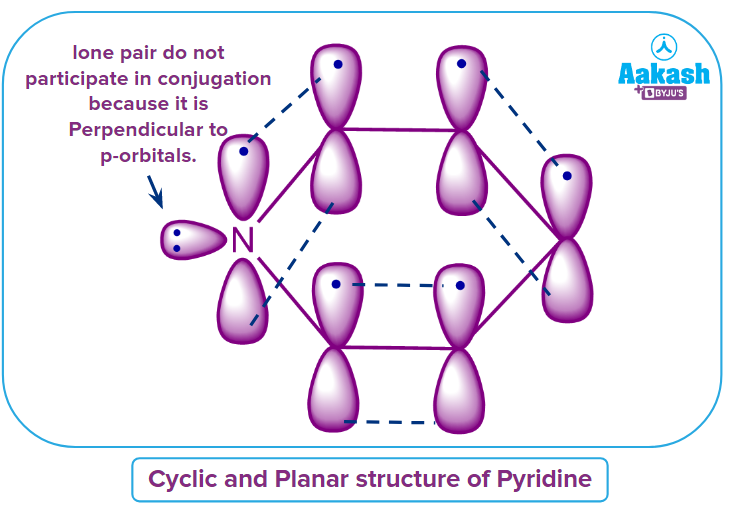
All 6 electrons present in the p-orbitals lobes which are present above and below the plane of pyridine are the delocalised electrons. Thus, it may be said that Rule-3 is also obeyed.
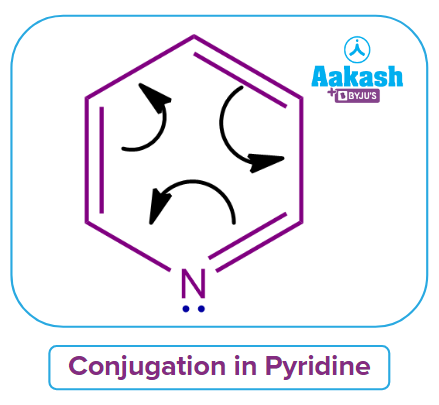
Let's check to see if pyridine agrees with Huckel's rule of aromaticity. In Pyridine, there are 6 electrons in total participating in conjugation.
If n = 1,4n+2 → 4(1) + 2 = 4+2 = 6.
Hence, Pyridine satisfies Huckel’s rule of aromaticity and aromatic in nature.
2. Cyclobutadiene:
Consider the example of Cyclobutadiene. Let us discuss whether Cyclobutadiene satisfies all the given four rules of aromaticity or not.
Yes, Cyclobutadiene has a cyclic structure and a planar molecule with sp2 hybridized atoms and has a planar shape. Hence, it follows Rule-1 and Rule-2.
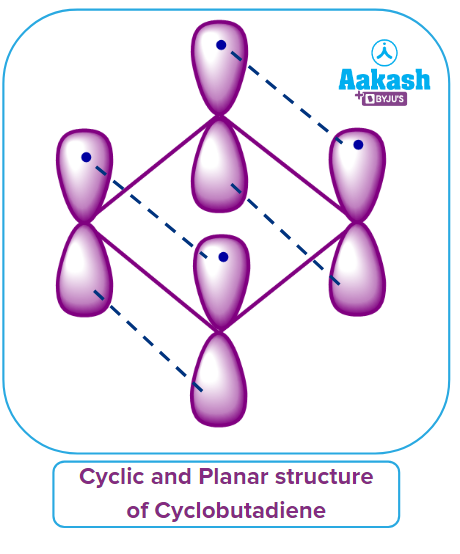
All 4 electrons present in the p-orbitals lobes which are present above and below the plane of cyclobutadiene are the delocalised electrons. Thus, it may be said that Rule - 3 is also obeyed.
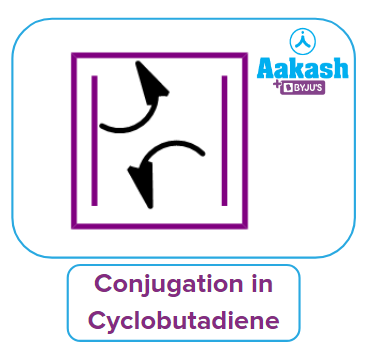
Let's check to see if pyridine agrees with Huckel's rule of aromaticity. In Cyclobutadiene, there are 4 electrons in total participating in conjugation.
If n = 1,4n→ 4(1) = 4
Hence, Cyclobutadiene satisfies three rules but exhibits (4n) electrons rather than (4n+2) electrons, then the given compound is Anti-Aromatic in nature.
3. Cyclopentadiene:
Consider the example of Cyclopentadiene. Let us discuss whether Cyclopentadiene satisfies all the given four rules of aromaticity or not.
Yes, Cyclopentadiene has a cyclic structure but not a planar molecule because it contains one sp3 hybridized carbon atom. Hence, it does not follow Rule 2.
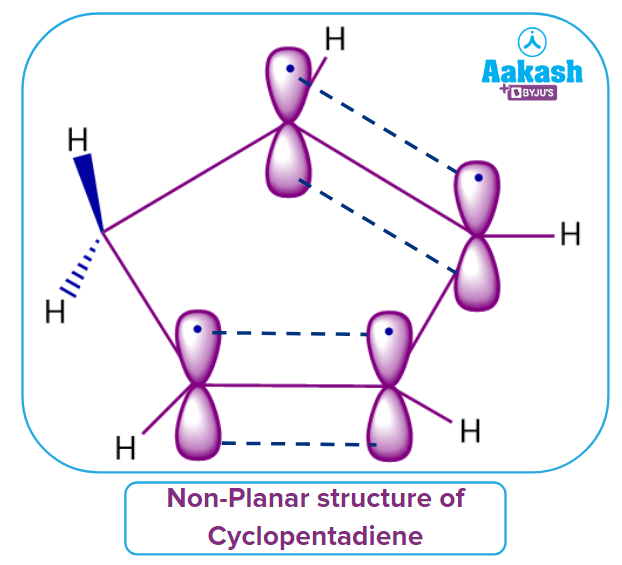
Cyclopentadiene does not satisfy one of the rules of aromaticity. Hence, the given compound is Non-Aromatic in nature.
Practice Problems:
Q1. Predict the nature of cyclic compound Imidazole on the basis of Huckel’s rule of aromaticity?
- Aromatic Compound
- Non-Aromatic Compound
- Anti-Aromatic compound
- None of these
Answer: (A)
Solution: Let's talk about whether imidazole meets all aromaticity requirements or not.
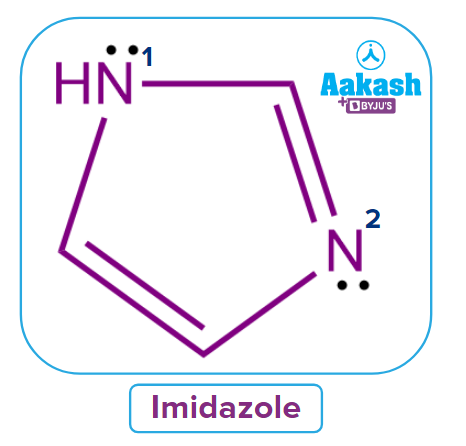
The molecule imidazole has a cyclic form, sp2 hybridized atoms, and a planar structure. The delocalized electrons are one single pair of nitrogen N1 and 4 electrons.

The total number of electrons involved in conjugation in imidazole is 6.
If n = 1,4n+2 → 4(1) + 2 = 4+2 = 6. Hence, Imidazole Huckel’s rule of aromaticity and aromatic in nature.
Hence, the correct answer is an option (A).
Q2. Predict the nature of the compound formed by the following reaction?
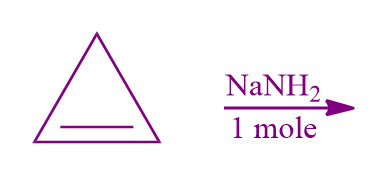
- Aromatic Compound
- Non-Aromatic Compound
- Anti-Aromatic compound
- None of these
Answer: (C)
Solution: This molecule with six e-atoms is cyclic, planar, conjugated, and contains sp3 hybridized atoms, but after reaction with sodium amide, it abstracts one H+ ion and gives a compound with 4 electrons.
The cyclic and planar compounds that must have a total of 4n conjugated electrons are classified as Anti-aromatic compounds.
Hence, the correct answer is option (C).
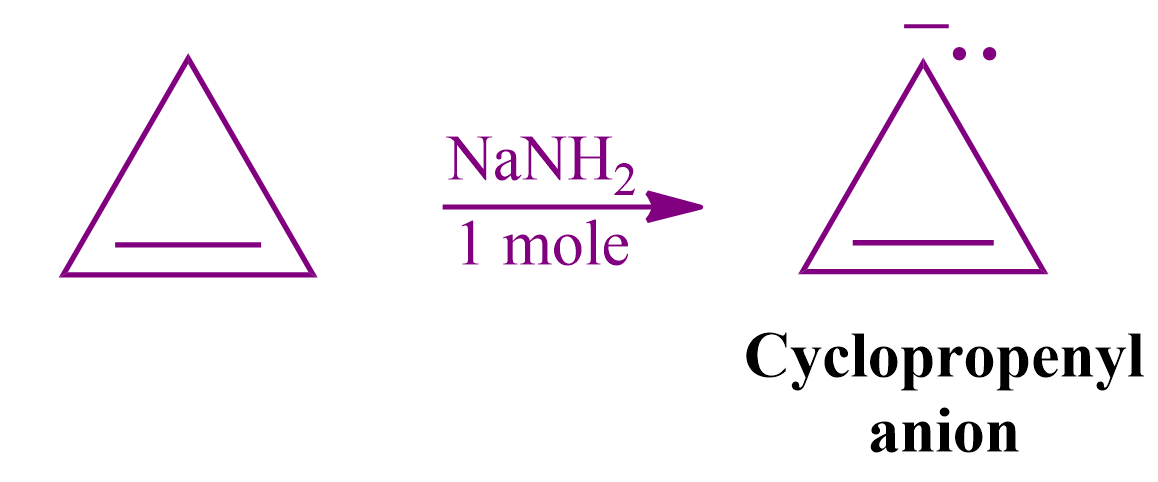
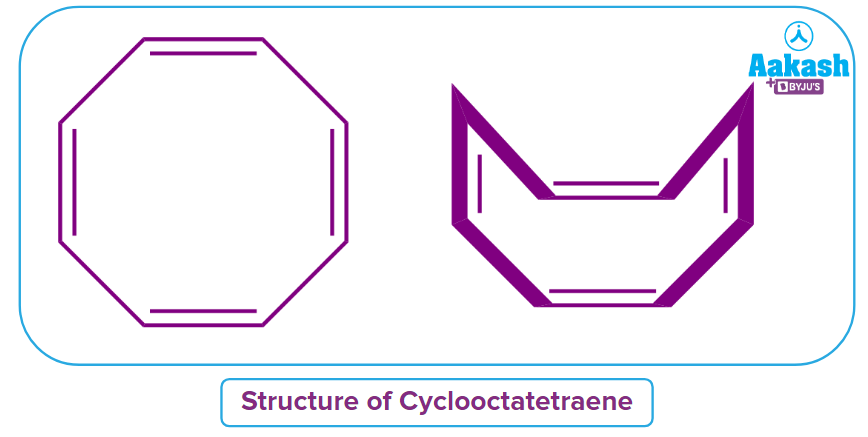
Q3. Using Huckel's rule of aromaticity, determine the nature of the cyclic compound Cyclooctatetraene?
- Aromatic Compound
- Non-Aromatic Compound
- Anti-Aromatic compound
- None of these
Answer: (B)
Solution: The given compound is Anti-Aromatic in nature if it is cyclic, planar, conjugated, and shows 4n electrons participating in conjugation in the given ring structure rather than 4n+2 electrons. The given compound cyclooctatetraene appears to be Anti-aromatic in nature but it is actually a Non-aromatic compound. Because of the large steric hindrance, the compound loses its planarity and looks like a tub as shown in the image and is also referred to as a tub-shaped structure.
Hence, the correct answer is option (B).
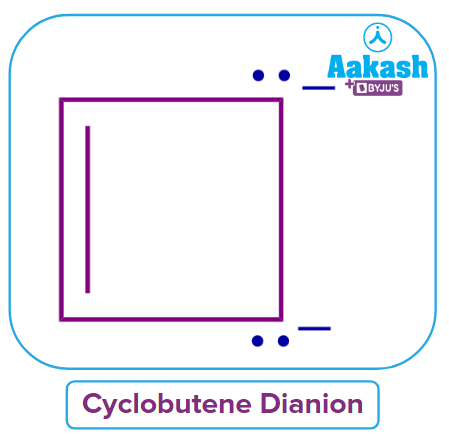
Q4. Predict the nature of cyclic compound cyclobutene dianion on the basis of Huckel’s rule of aromaticity?
- Aromatic Compound
- Non-Aromatic Compound
- Anti-Aromatic compound
- None of these
Answer: (A)
Solution: The cyclic and planar compounds that must have a total of (4n+2) conjugated electrons are classified as Aromatic compounds.
The given cyclobutene dianion is having 6 e- and has a cyclic, planar and conjugated system. It has two sp3 hybridized carbon atoms but the lone pair of sp3 hybridized atoms are participating in the system. If one lone pair of sp3 hybridized atoms will participate in the conjugation, then we can withdraw that condition.
Hence, the cyclobutene dianion is aromatic in nature and the correct answer is option (A).
Related Video Link: Aromaticity | CHEMISTRY | JEE | Concept of the Day | SM Sir
Frequently Asked Questions-FAQs:
1. Comment on the heat of hydrogenation of benzene.
Answer: Because benzene comprises three double bonds, it was predicted that the heat was significantly lower.
Because of its aromaticity, the heat of hydrogenation in the benzene molecule has decreased. The molecule becomes substantially more stable with the presence of 6 delocalizing electrons, which hydrogenation (ΔH) would be higher than 54.9 kcal mol-1. But at 49.3 kcal mol-1, it is lowers the ΔH value.
2. Comment on the aromaticity of (Z)-buta-1,3-dien-1-ylbenzene.
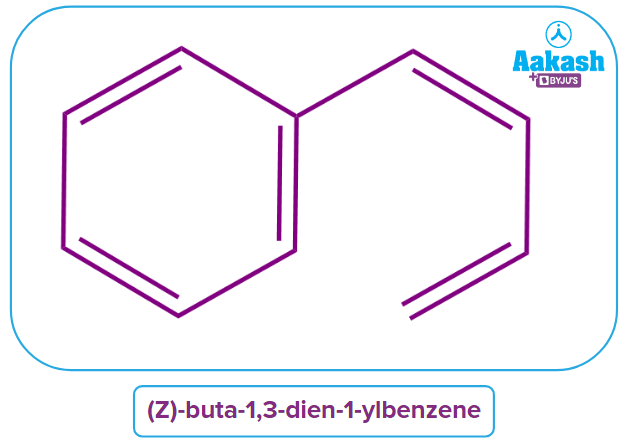
Answer: The aromaticity of any compound depends upon the cyclic and planar structure and the conjugation present within the ring system. As the ring system is benzene which has a cyclic planar structure and has 6 electrons which obey (4n+2) electrons system rule or Huckel’s rule. Hence, the given compound is Aromatic in nature.
3. What is the origin of the Word ‘Aromaticity’?
Answer: Aromaticity is linked to the aroma word and derived from the Greek aromatikos, which means "sweet spice". But there is one key distinction between the two words. Aroma simply means "to smell," which can be either pleasant or unpleasant, as opposed to aromatic, which implies "having a nice smell."
4. What is the basic difference between Aromatic Homocyclic or heterocyclic compounds?
Answer: Aromatic homocyclic compounds have sigma bonds and delocalized electrons between the same type of atoms (carbon) is present in the ring system whereas Aromatic heterocyclic compounds are those have sigma bonds and delocalized electrons between carbon atoms and an additional element, such as oxygen, nitrogen, phosphorus or sulfur present in a ring system.


