-
Call Now
1800-102-2727
Hess Law of Constant Heat Summation: Hess Law Statement with Example, Applications, Practice Problems, FAQs.
Recently India hosted an international Chess tournament. It is an interesting intellectual game, played during early times by Kings to develop and test their skills of war strategies.
The game is played on a board divided into 64 squares. The game is supposed to be played between two opposing kings with their arsenal of queen, bishop, rook, horse and soldiers. Each of them has a unique movement with the ultimate motive of killing or at least immobilizing the opponent king to win the battle.
You are not going to play chess but simple play with the chessboard. You shall take a rook which can move in only a straight line path, front and back. Place it in any corner square of the board.
You have to move it to the opposite corner square. You are free to move in any straight direction and any number of squares you want. But, nullify the squares if you are moving to return.
Can you tell me the minimum number of squares you have to move to reach the end?
Irrespective of your directions and the number of squares you cross, ultimately the minimum number of squares to move will be 14. A parallel exists in chemistry as pointed out by Hess.
Say, you have some reactants and products obtainable from them. The reactants are allowed to react with other materials forming different products. After all these reactions, if the final product is going to be the same as expected of a direct reaction, then the net energy involved in all reactions will be the same as the direct conversion of the reactants to products.
You will know more about Hess Law here.
TABLE OF CONTENTS
- Hess Law of Constant Heat Summation with Example
- Applications of Hess Law of constant Heat Summation
- Practice Problems
- Frequently Asked Questions (FAQs)
Hess Law of Constant Heat Summation with Example
First law of thermodynamics talks about the energy being neither created nor destroyed, but convertible from one form to another of chemical bond energy, heat energy, electrical energy, work etc. Hence it implies in chemical reactions also the energy before and after the reactions should be equal between a given set of reactants and products irrespective of the path followed in the conversion ie direct or involving multiple steps. It also implies that the energies involved in reactions can be considered as either reactant or product and can be subjected to arithmetic laws.
The thermodynamic principle is the basis of Hess's law of heat summation, which states that the overall change in enthalpy for a multi-step direct chemical reaction is equal to the sum of the enthalpy changes for each step, and it is independent of the path taken.
Consider for example a reactant A forming a product B. Let us assume three different pathways with multiple intermediates or product formation.
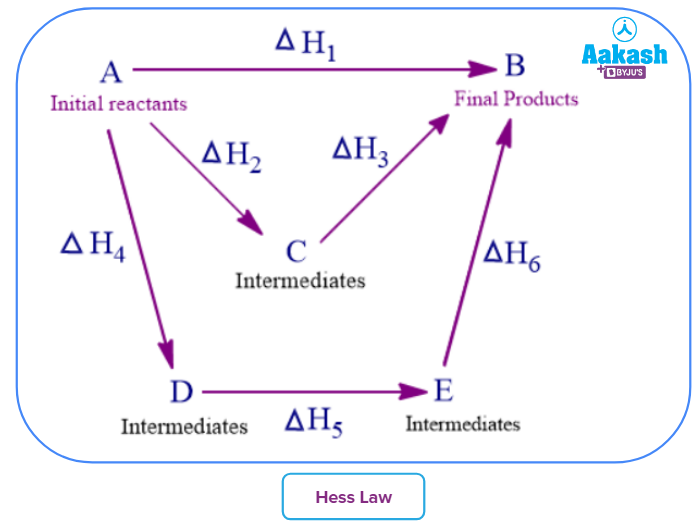
The direct path of A → B involves an energy change of ΔH1
The conversion, through a single intermediate C, involves a formation and dissociation energy of ΔH2 and ΔH3
The conversion through two intermediates, similarly involves energies of ΔH4, ΔH5 and ΔH6
According to Hess’ law of heat summation, summation of energies of all the reactions must be same irrespective of single or multiple steps followed in forming the product.
ie, ΔH1 = ΔH2+ ΔH3 = ΔH4+ΔH5+ΔH6.
Hence, heat energy also can be considered as a reactant or product of the reaction and included in the reaction.
Example 1
Carbon and oxygen react to form carbon dioxide. It is an exothermic reaction with 94.3kcals.
It may proceed in two steps as follows
On summing up the reactions and the energies,, we get C + O2 → CO2 + 94.3kcals
Which is the same as the direct reaction energy release of 94.3kcals, confirming the energy or enthalpy of a formation of carbon dioxide reaction is same for both single and two-step reaction as per the Hess law.
Example 2
Combustion reactions of carbon, sulphur and carbon disulphide are exothermic. Reactions with the enthalpy or energy changes are as follows-
C(s) + O2 (g) → CO2 (g) + 393.5 kJ ……..(i)
S(s) + O2 (g) → SO2 (g) + 296.8 kJ .…….(ii)
CS2 (l) + 3O2(g) → CO2(g) + 2SO2(g) + 1075.0 kJ ..…….(iii)
Let us calculate the enthalpy of formation of carbon di sulphide
.Equation (i): C(s) + O2 (g) → CO2 (g) + 393.5 kJ
2x equation(ii): 2S(s) + 2O2 (g) → 2SO2 (g) + 593.6 kJ
Reverse of equation (iii): CO2(g) + 2SO2(g) → CS2 (l) + 3O2(g) -1075.0 kJ
Adding all the above three reactions: C (s) + 2S (s) → CS2 (l) -87.9 kJ
The heat of formation of carbon disulphide is 87.9kJ
Applications of Hess law of Constant Heat Summation
Hess’ law of heat summation is a useful method for theoretically calculating enthalpy changes of reactions which cannot be experimentally performed.
1. Enthalpy change in allotropic transformations
Graphite and diamond are allotropes of carbon. No method is available to convert graphite to diamond as of now and the enthalpy changes of the conversion of graphite to diamond can only be calculated theoretically and that by Hess law of heat summation only.
Both of them react with oxygen to form carbon dioxide but their enthlphy of reaction is different as -393.4kJ and – 395.4kJ respectively.
C (graphite) + O2 → CO2 ΔHg = -393.4kJ ……(i)
C (diamond) + O2 → CO2 ΔHd = -395.4kJ……(ii)
As heat of reactions is numerically same with reverse sign for reverse reactions
CO2 → C (diamond) + O2 ΔHd = + 395.4kJ….(iii)
Adding (i) and (iii)
C (graphite) → C (diamond) ΔHtr = +2.kJ
Enthalpy change in the allotrope transition of graphite to diamond is endothermic of 2KJ.
2. Exothermicity or endothermicity of a reaction
The energy changes of a reaction can be calculated even before conducting experiments, which will help in designing reactors in large-scale processes.
Hydrogen and iodine combine to form hydrogen iodide.
12H2 (g) + 12I2 (g) → HI (g) + ΔHf
In the reaction, hydrogen and iodine molecules are broken to form an HI bond.
The dissociation energies of hydrogen, iodine and hydrogen iodide molecules are 218, 107kJ and 299kJ respectively.
The heat of formation= Enthalpy of reactants.- Enthalpy of products
The heat of formation of HI reaction = 12 Bond energy of H2 + Bondenergy of I2 - Bond energy of HI
=12 (218 + 107) - 299 = - 162.5kJ
The heat of formation of HI is negative and so the formation of HI is an exothermic reaction..
3. Enthalpy of formation
Enthalpy of formation of compounds which cannot be theoretically determined can be theoretically estimated.
Benzene cannot be synthesised directly by the combination between carbon and hydrogen. Still, the enthalpy of the formation of benzene can be calculated as follows using known heat of reactions.
6C + 3H2 → C6H6 ΔHf C6H6 = ?
Heat of formation of carbon dioxide and water from their elements are availbl eas -393.5kJand -285.8KJ respectively.
C + O2 → CO2 ΔH1 = -393.5kJ…. (i)
Pure benzene can be burned and the heat of combustion of benzene is observed to be -3301kJ.
C6H6 + 9O2 → 6CO2 + 3H2 O ΔH3 = -3301kJ ……(iii)
Reaction (iii) can be reversed and with proper balancing of elements as
6 x Reaction (i): 6C + 6O2 → 6CO2 6ΔH1 = -2361kJ….(iv)
3 x Reaction (ii): 3H2 + 3O2 → 3H2 O 3ΔH2 = -857.4kJ……(v)
Reverse of reaction(iii): 6CO2 + 3H2O → C6H6 + 9O2 -ΔH3 = +3301kJ ……(vi)
Adding(iv), (v) and (vi), we get 6C + 3H2 → C6H6 ΔHf = +82.6kJ
Theoretical heat of formation of benzene is + 82.6kJ.
4. Calculation of Lattice Energy
Ionic solids have specific long-range crystal lattices where the ions are closely packed. The energy used to pack them closely in a crystal lattice structure is called Lattice energy. The lattice energy of a crystal cannot be experimentally determined but can be calculated using Hess's law of constant heat summation.
Practice Problems
Q1. Hess’s law deals with
(A) Heat change in a chemical reaction
(B) the Influence of pressure on the volume of gas
(C ) Equilibrium constant
(D) Rate of reaction
Answer: Option (A)
Solution: Hess’s law deals with the heat changes occurring in a chemical reaction. It states that the heat change is irrespective of the paths the reactants take to form the product.
Q2. Which of the following is not an application of Hess’s law?
(A) determination of heat of formation
(B) determination of heat of transition
(C) determination of Gibb’s energy
(D) determination of heat of hydration
Answer: Option (C)
Solution: Hess’s law deals with the enthalpy changes associated with chemical reactions like the heat of formation, transitions and hydration. Gibb’s energy is not a heat of reaction but the determination of available work during processes and the spontaneity of processes.
Q3. Hess Law of heat summation stems from which law of thermodynamics?
a. Zeroth Law
b. First Law
c. Second Law
d. Third Law
Answer: Option A.
Hess's law of constant heat summation is based on the constancy of the total energy of the system before and after the reactions. It is the same as the zeroth law of thermodynamics which says that energy can neither be created nor destroyed but can be converted from one form to another.
Q4. Calculate the lattice energy of potassium bromide from the following given data.
The heat of Sublimation of potassium = 89.24 kJ mol-1
Ionization energy of potassium = 419 kJ mol-1
Bond dissociation energy of bromine = 193 kJ mol-1
Electron affinity of bromine = - 319 kJ mol-1
Heat of formation of KBr = -394 kJ mol-1
From the given data-
K (s) → K (g) - 89.24kJ mol-1
K (g) → K++ (g) + e-‒ - 419 kJ mol-1
Br22 (g) → 2 Br (g) - 193 kJ mol-1
Br (g) + e- → Br-‒ (g) +319 kJ mol-1
K (s) + ½ Br2 (g) → KBr (s) +394 kJ mol-1
Lattice energy is the energy required to keep the gaseous ions in a crystal lattice. It is determined as the enthalpy required to break one mole of the solid ionic molecule into gaseous ions.
KBr (s) → K++(s) + Br-‒ (g) + X
The formation of solid KBr can be considered to involve the following energy in a multistep process steps-
Sublimation of potassium- K (s) → K (g) - 89.24kJ mol-1 ……..(i)
Ionization of potassium- K (g) → K++ (g) + e-‒ - 419 kJ mol-1 ……..(ii)
Half of dissociation of Br2- 12Br22 (g) → Br (g) - 1932 kJ mol-1 ……..(iii)
Formation of bromide ion- Br (g) + e- → Br-‒ (g) +319 kJ mol-1 ………(iv)
Formation of solid KBr- K++ (g) + Br-‒ (g) → KBr (s) + ΔH ……..(v)
Adding all the five equations, we have,
K (s) + ½ Br2 (g) → KBr (s) + ΔH -285.74 kJ mol-1
The enthalpy of this reaction is given as K (s) + ½ Br2 (g) → KBr (s) +394 kJ mol-1
Comparing the two equations, ΔH -285.74 = 394 or ΔH = 679.74 kJ mol-1
Enthalpy of the reverse reaction,
KBr (s) → K++(s) + Br-‒ (g) + X will be = - ΔH = - 679.74 kJ mol-1 = X
So, the lattice energy = 679.74 kJ mol-1
Frequently Asked Questions(FAQs)
Q1. What is enthalpy?
Answer: Enthalpy is a measure of the total energy content possessed by a system at constant pressure conditions. It is denoted by ΔH
Q2. Which among enthalpy or enthalpy changes is more relevant in thermodynamics?
Answer: Enthalpy, is the absolute energy content of a system under certain conditions of pressure, temperature and volume. It is not measurable.
When a system changes from one state to another, its enthalpy changes. We are interested only in the enthalpy changes between the initial and final state. These enthalpy changes in the system can be measured even without the knowledge of the enthalpy of the initial and final states. Hence enthalpy changes are more useful than enthalpy.
Q3. What is the standard heat of formation?
Answer: Standard heat of formation is enthalpy changes accompanying the formation of one mole of product from reactants in their standard conditions.
Q4. Two people A and B estimate the heat of the formation of carbon dioxide using excess oxygen. A starts with coal and B uses graphite. Whose values can be taken as the standard heat of the formation of carbon dioxide?
Answer: Since graphite is the standard form of carbon, values of the experiment conducted by B can be taken as the standard heat of the formation of carbon dioxide.


