-
Call Now
1800-102-2727
Heat and Work - Definition of Heat and Work, Work Done by the System, IUPAC Signs for Heat and Work & P-V graphs

They say that work makes the world go around. No doubt about that. We all need work to live, survive and pay our bills for our daily needs. But too much of it also makes us tired. Work is something that we cannot live without but at the same time, we will be exhausted if we do too much of it. We all need a break sometimes, isn’t it? But what kind of work we are talking about? Well, for us it's the work which we do by using our brain or some physical activity. What about the mechanical work? Mechanical work is a type of work where we can directly see the impact of the expenditure of energy. When we lift and shift heavy objects, we expend energy, we become tired. In other words, we make a conscious effort. But how do we make this effort?
We use force to move the object. In terms of basic physics, the work done is the product of the force we apply by the distance the object is moved. If we push a box by some distance ‘d’ by applying force ‘F’, then work done is given by W=F d . Now, there are certain machines which use the heat energy to convert it partially into useful work. These machines work on certain thermodynamics principles. But to understand this first we need to know what are these terms, Work and Heat?
TABLE OF CONTENTS
- What is a work?
- What is a Heat?
- Work done by the system
- IUPAC signs for Heat and Work
- Practice Problems
- Frequently Asked Questions - FAQs
What is a work?
The energy exchanged between the system and the surroundings due to any factor other than the difference in the temperature is known as work.
• It is denoted by the symbol, W.
• Formula for work,
Where, F is the force and dx is a small displacement.
What is a Heat?
Thermodynamics has existed ever since the existence of fire, where energy is produced by burning wood.

A long back, humans were just another ‘animal’, they used to hunt and eat without even cooking it. But one spark changed it all. Once we had control of fire, nature was suddenly controllable. We could protect ourselves from wild animals and from a harsh cold climate. But that’s not all. Fire helped humans cook! Now, for the first time in history, the complex molecules in food could get broken down outside the body. This meant consuming far less energy to digest it which in turn meant more energy available to us for other uses. One of the early chemical reactions carried out by our forefathers is burning wood to produce fire.

Fire is a simple chemical reaction that releases light and heat. The burning of natural fuels like wood was our first attempt at converting energy from one form to another.
Energy transfers across a boundary as a result of the temperature difference between the system & the surroundings, this energy is known as heat. Heat always flows from high temperature to low temperature.
Let us assume Earth to be the system at a lower temperature and the Sun to be the surrounding with a higher temperature. The exchange of energy from the Sun to the Earth is due to the difference in temperature. This energy is known as heat energy. Heat energy is denoted by the symbol, q.
Both heat and work are the modes of transfer of energy between the system and the surroundings. Both are path functions with the units, J or cal.
Work Done by a System
As discussed earlier work is the motion against an opposing force so work means the energy it takes to move an object against a force.
More specifically, in this case, we are interested in the work done by expansion and work done on the system during the contraction of gases against constant external pressure.
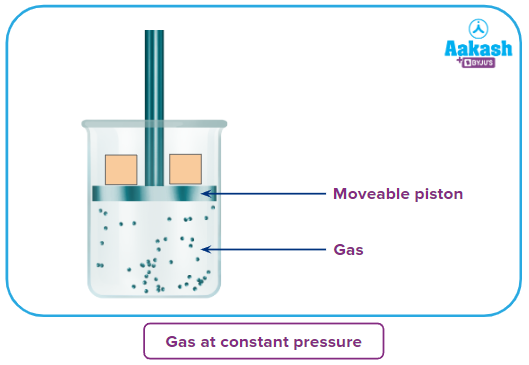
Now how does a gas do work? If the gas can move an object, then surely it can produce work.
Now how can a gas move an object? If it is able to exert some force on the object. Now, the question arises how does any gas exert a force?
Let’s imagine a gas in a cylinder with a movable piston-like lid. The gaseous molecules inside the cylinder and the ones outside it in the atmosphere will be in random motion. They are continuously colliding with the piston both from the inside and the outside. These collisions are the genesis of what we call pressure. Due to collisions inside the cylinder, there will be pressure inside called internal gas pressure (Pin). The collisions from the outside result from the atmospheric pressure called external pressure Assuming wights are placed on the top of the piston, which exerts a force, F over the cross-section A, adding to the external pressure. Let’s consider this external pressure due to atmospheric pressure and these weights is Pext. Now, suppose the gas is slightly heated and according to Charle’s law, with the increase in the temperature the gas will expand. So, the piston will be moved slightly upwards by a distance of dl.
We already know the relationship between force and pressure:
Where F is the force exerted by the gas, P is the pressure and A is the area on which the force is applied.
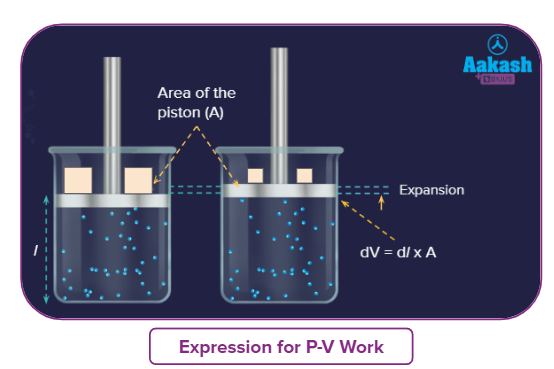
By using the convention that work done by the system on the surroundings is negative, work required to move the piston to a distance dl against an opposing force of magnitude F can be represented as
Hence,
Now ,
So,
For the purpose of this example, let us assume that the piston lid is weightless and frictionless. Now let us start with the piston in equilibrium. There are two opposing forces acting on the piston. One is due to the pressure of the gas from the inside as we have already discussed, that acts upwards on the piston and the atmospheric pressure from the outside that acts downwards on the piston.
If both these opposing forces are equal, then the piston doesn’t move. Now, what if we change the external pressure, thereby the external force on the piston - reduce or increase it.
Let us see what happens when we increase the external pressure by adding weight to the piston. This weight will exert an additional force in the downward direction on the piston. Now, the force acting on the piston in the downward direction is greater than the force acting on the piston in the upward direction.
So the piston shifts downwards, resulting in a decrease in the gas volume. In other words, the gas has contracted. In the case of the expansion, we can see that it is the gas that is moving the piston, while in the case of the contraction, we can see that it is the external pressure that is moving the piston.
Let us take the case of when we reduce the external pressure. This would mean that the upward force on the piston is greater than the downward force. Hence, the piston will move upwards and when the piston moves upwards, we can see that the volume of the gas increases. In other words, the gas expands.
This upward and downward movement of the piston is nothing but mechanical work. Therefore this work done by gas is also sometimes called pressure-volume or P-V work.
P-V Graphs
We now know we can use the equation to find work done by gas. Also, the graphical approach suggests that work done on the gas and by the gas is the area under the P – V graph.
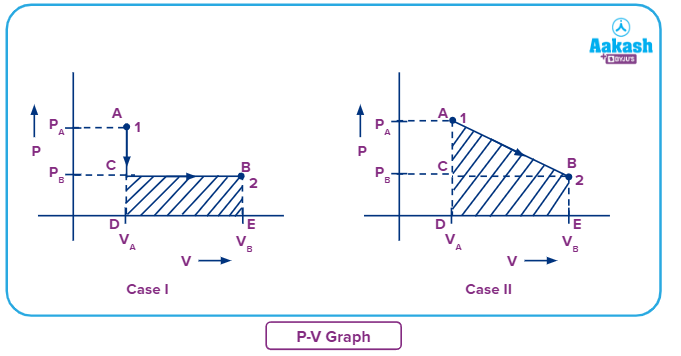
Considering Case I: Work done from state 1 to state 2.
Work done can be calculated by finding the area under the P-V curve (i.e Under AC curve and CB curve).
In AC curve, Volume is constant, so area under AC curve will be 0.
Considering Case II: Work done from State 1 to State 2
Work done can be calculated by finding the area under the P-V curve (i.e., AB curve)
Note: W12 in case I is less than W12 in case II as the area under the curve is more in Case II in comparison to Case I.
So for the above-described processes, we see that the area is different in both the cases as both have followed different paths to reach state 1 from state 2 (as shown in the figure below), and hence work is also not a state function and depends on the path. So, work is a path function.
IUPAC signs for Heat and Work
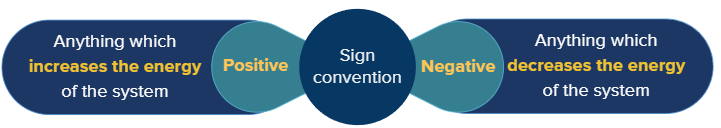
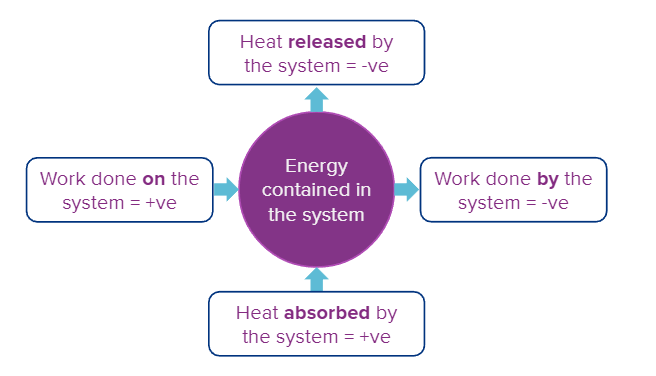
The signs of 'q' and 'w' are:
- Heat absorbed by the system (q) i.e., increase in the energy of the system = +ve
- Heat evolved by the system (q) i.e., loss in the energy of the system = -ve
- Work done on the system (W) = +ve
- Work done by the system (W) = -ve
Related Topic Videos : https://www.youtube.com/watch?v=1qmTgKzTVps&t=1307s
Practice Problems
Q. 1. A system absorbs 20 kJ of heat and does 8 kJ of work. What will be the signs of heat transfer and work done respectively?
a. +ve , -ve
b. -ve , +ve
c. +ve , +ve
d. -ve , -ve
Answer: (A)
Solution:
As per IUPAC sign conventions:
Heat absorbed by the system (q) = +ve = 20kJ
Work done by the system (W) = -ve = -8kJ
Q. 2. Choose the correct statement(s).
a. During expansion, work is done by the system
b. Heat absorbed by the system is taken as positive
c. Work done by the system is taken as negative
d. All of these
Answer: (D)
Solution: During expansion system actually does work to increase the volume, hence work is done by the system. As per IUPAC sign conventions work done by the system is always considered as negative and heat absorbed by the system is taken as positive.
Q. 3. What will be the work done in this AB process?
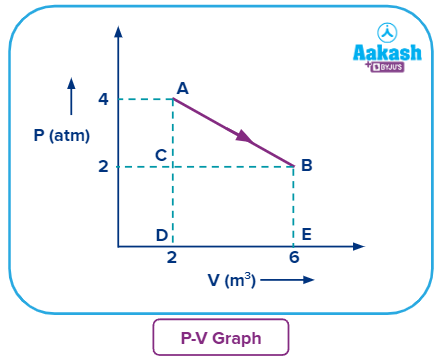
a. -12 atm-m3
b. 14 atm-m3
c. 10 atm-m3
d. 16 atm-m3
Answer: (A)
Solution: Work done can be calculated by finding the area under the P-V curve.
Now during process AB, we can see that volume is increasing i.e., expansion is happening and work done during expansion is always negative.
So,
Q. 4. The work done during the expansion of a gas from a volume of 8 m3 to 16 m3 against a constant external pressure of 5 atm is:
a. - 40 atm m3
b. - 24 atm m3
c. - 80 atm m3
d. 40 atm m3
Answer: (A)
Solution: Using this formula,
Frequently Asked Questions – FAQ
Q. What is the difference between heat and work?
Answer: Heat and work both are forms of energy. Actually, heat is the energy transferred across a boundary as a result of the temperature difference between the system & the surroundings but work is the flow of energy without temperature difference.
Q. Is work a path function or a state function?
Answer: A quantity is said to be a state function only when its value depends on the initial and final state, regardless of the path it has followed.
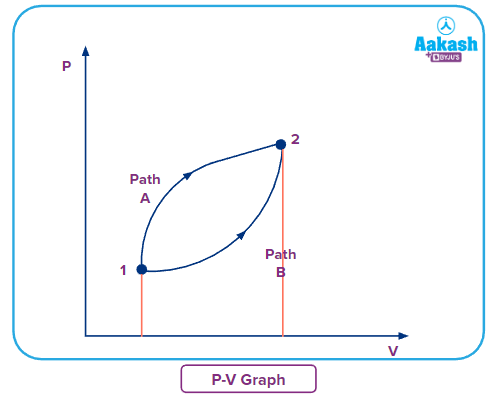
In this P-V diagram, there is two paths A and B to reach from 1st state to 2nd state and we know that work is the area under the curve. So just by observing the area under the curve, we can say that path B has less area under the curve than path A but the state 1 and 2 remain the same, which means work is path-dependent.
Q. Why work done in compressing a gas is positive?
Answer: During compression volume of the gas decreases as the gas is compressed by external pressure which means that work is done on the gas. As the volume decreases so the change in volume is negative, the work will be positive because work is done by the surroundings on the system.
Q. Why is the work given a negative sign convention in the formula W=-PV ?
Answer: The negative sign associated with P-V work done indicates that the system loses energy. If the volume increases at constant pressure (ΔV > 0), the work done by the system is negative, indicating that a system has lost energy by performing work on the surroundings.
In this formula, the internal pressure of the gas that is moving the piston upwards and expanding the gas is considered. In other words, it is the work done by our system which is the gas and in Chemistry, any work done by a system is given a negative sign.


