-
Call Now
1800-102-2727
Halogenation of Benzene - Electrophilic Substitution Reaction, Addition Reactions, Practice Problems and FAQs
Typhoid fever is most common in areas with poor sanitation and limited access to safe drinking water. Chloramphenicol, a chlorine-containing antibiotic produced by soil microorganisms, is very effective in the treatment of typhoid fever. Many halogen-containing organic compounds, such as chlorobenzene derivatives, find widespread use in industry and daily life.
Let us now look at how to make chlorobenzene or another halobenzene!
Table of Contents
- Electrophilic Substitution Reaction of Benzene ring
- Halogenation of aromatic hydrocarbons
- Addition Reaction-Halogenation of aromatic hydrocarbons
- Practice Problems
- Frequently Asked Questions
Electrophilic Substitution Reaction of Benzene ring:
Reactions in which an electrophile replaces one or more H-atoms of aromatic compounds are known as electrophilic substitution reactions.
As benzene is electron-rich, it undergoes electrophilic substitution reactions. It comes under the
category of Lewis bases. It has 3 π bonds, i.e., 6 π electrons.
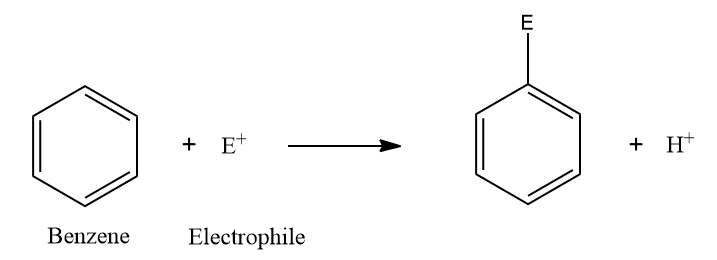
Note: In short, electrophilic substitution reaction is written as SE .
Mechanism of Electrophilic substitution reaction:
SE reaction of benzene occurs in 3 steps.
Step-1: Generation of an electrophile
In the first step, different electrophiles are generated in different electrophilic substitution
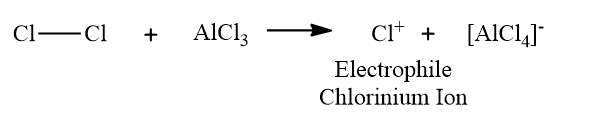
reactions. Electrophiles for Halogenation electrophilic substitution reactions are X+: Cl+, Br+ , I+
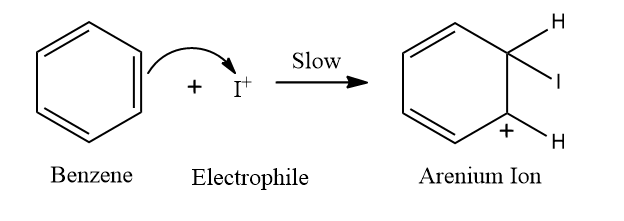
Step 2: Formation of a carbocation (C+) intermediate
Benzene attacks the electrophile resulting in the formation of arenium ion. Here E+ can be Cl+,Br+,I+ in the case of halogen of benzene.
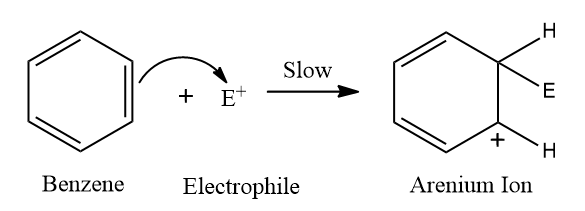
One sp3 Hybridized carbon exists in the arenium ion, commonly known as the sigma (σ) complex. Wheland's intermediate is another name for it. Because one of the C-atom becomes sp3 hybridized, it is resonance stabilized but not aromatic.
Since there is an extended conjugation in arenium ions, resonance stabilization is there.
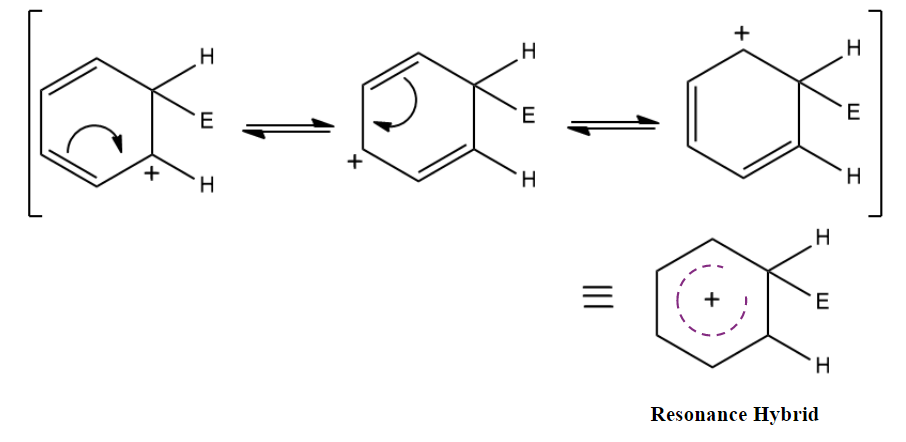
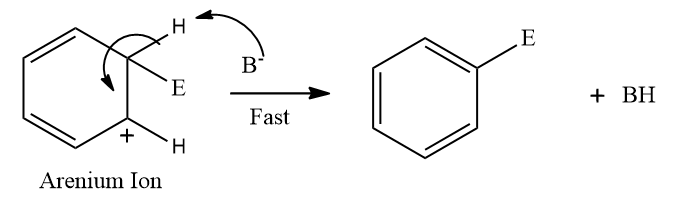
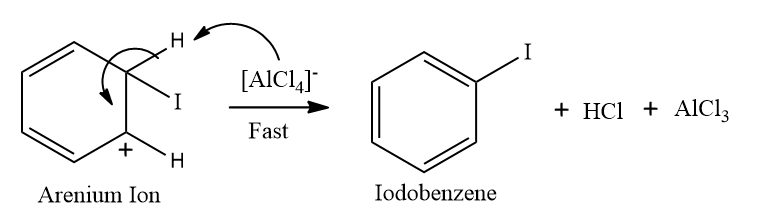
Step 3: Restoration of aromaticity by removal of a proton (H+)
To restore the aromatic character, σ-complex loses H+ from the sp3 hybridised carbon. In this step, the base (B-) attacks the sp3 hybridised carbon to remove H+ and BH is formed.
Halogenation of aromatic hydrocarbons:
Arenes react with halogens in the presence of anhydrous Lewis acid like anhydrous FeCl3, FeBr3, or AlCl3 to yield haloarenes.

Mechanism: Halogenation of benzene takes place in three steps
Step-1: Generation of the electrophile
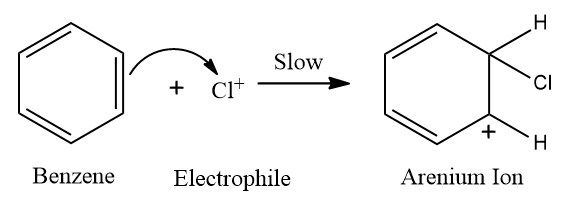
Step-2: Formation of a carbocation intermediate
In this step, the electrons of one of the double bonds of benzene attack the halonium ion,
and a carbocation intermediate is formed. Because of the extended conjugation, the () sigma complex is resonance stabilised but not aromatic.
Step-3: Restoration of aromaticity by removal of a proton (H+)
In this step, loss of proton takes place from sp3 hybridised carbon, and aromaticity is regained and halobenzene is formed.
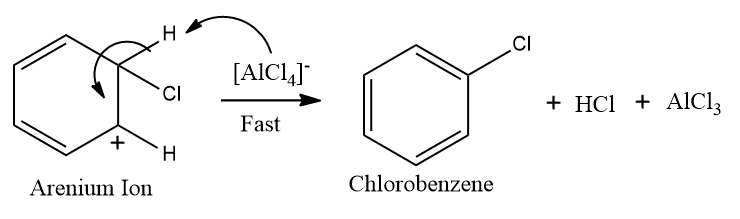
Note: AlCl3 does not get consumed in the reaction, thus, acts as a catalyst.
If an excess electrophilic reagent is used, a further substitution reaction may take place in which
Other H-atoms of the benzene ring may also be successively replaced by the electrophile.
Example:
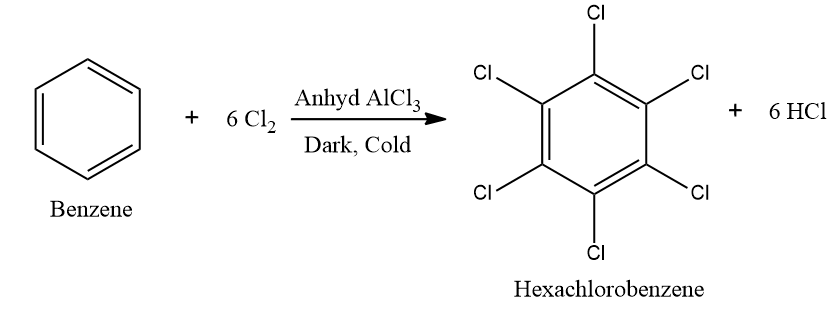
Addition Reaction - Halogenation of aromatic hydrocarbons:
Under ultraviolet light, three molecules of chlorine react with benzene to produce benzene
hexachloride at 500 K.
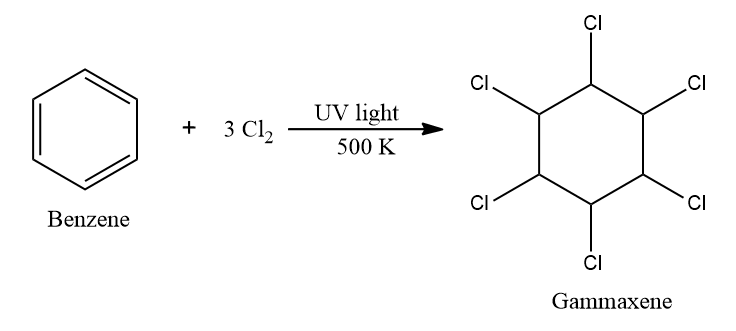
Gammaxene is used both as an agricultural insecticide and as a pharmaceutical treatment for lice and scabies (skin infection). It is banned as it affects the nervous system, liver, kidneys and is a Carcinogen.
Practice Problems:
Q1. Predict the product A for the given reaction:
A)
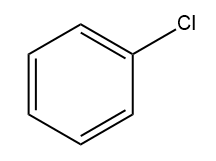
B)
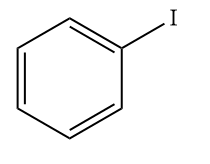
C)
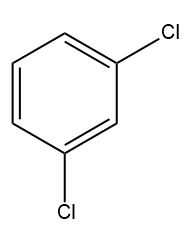
D)
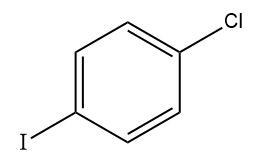
Answer: (B)
Solution: AlCl3 can accept a pair of electrons and thus acts as a Lewis acid. Since iodine is less electronegative than chlorine, AlCl3 reacts with ICl to form iodonium ion (I+) as the electrophile. The electrophile (I+) reacts with the benzene ring to form iodobenzene.
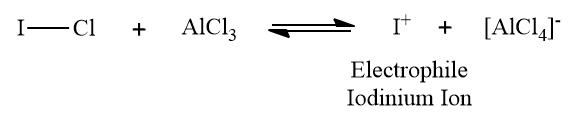
Hence, option (B) is the correct answer.
Q2. Which of the following is correct regarding the presence of a halogen group on a benzene ring?
a. It activates the ring towards electrophilic substitution.
b. It renders the ring basic.
c. It deactivates the ring towards nucleophilic substitution.
d. It deactivates the ring towards electrophilic substitution.
Answer: (D)
Solution: Based on electrophilic substitution reaction, Since halogens exhibit both the –I (strong) and +R effects. Halogens are moderately deactivating groups due to their relatively stronger –I effect in aryl halides. The +R effect, on the other hand, increases electron density at ortho-para positions more than at meta positions.
Therefore, option (D) is the correct answer.
Q3. Why does benzene easily undergo electrophilic substitution reactions but struggle with nucleophilic substitutions?
Solution: Benzene is a planar molecule with electrons delocalized above and below the plane of the ring. As it is electron-rich. It is very appealing to electron-deficient species, such as electrophiles. As a result, it readily undergoes electrophilic substitution reactions.
Nucleophiles are electron-rich species and benzene repels them. As a result, nucleophilic substitutions of benzene are difficult.
Q4. What will be major product A and B in the following reaction?
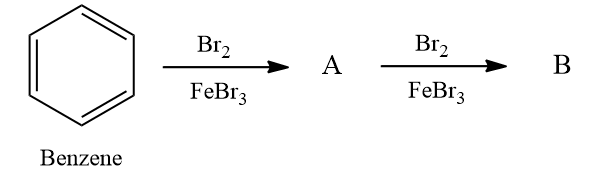
a. Bromobenzene, 1,2-dibromobenzene
b. Bromobenzene, 1,4-dibromobenzene
c. Reaction will not occur
d. Bromobenzene, 1,3-dibromobenzene
Answer: (B)
Solution: Arenes react with halogens in the presence of a Lewis acid like anhydrous FeCl3, FeBr3, or AlCl3 to yield haloarenes. Hence as halogen is bromine, the product A is bromobenzene.
The +R effect of halogens increases the electron density at the ortho-para positions more than the meta position. Hence product B should be ortho or para product with respect to bromine. The para product is more stable than ortho due to less steric hindrance. Hence the major product is 1,4-dibromobenzene.
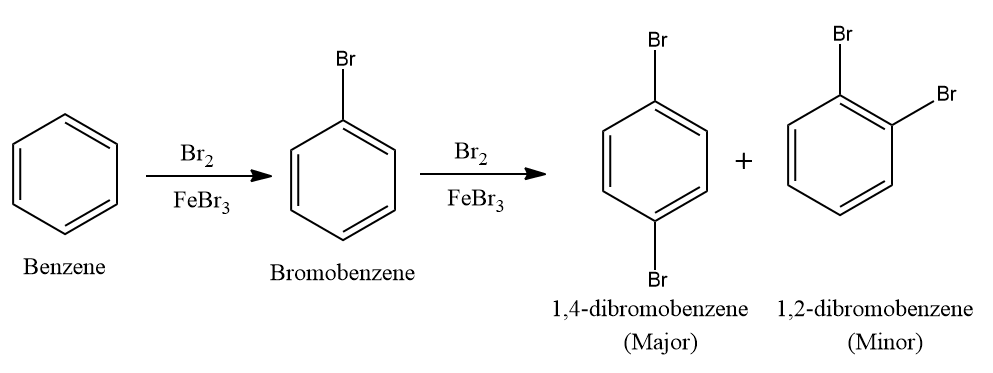
Hence, the correct answer is option (B).
Frequently Asked Questions-FAQs:
1. Why do catalysts often come in powder form?
Answer: Catalysts often come in the form of powders, pellets powders, pellets or fine gauzes, this provides the largest possible surface area for them to work.
2. What role does the catalyst play in electrophilic aromatic substitution?
Answer: The catalyst aids in the formation of an electrophile from the attacking reagent. For example, Because chlorine is a poor electrophile, ferric chloride is utilised as a catalyst in the chlorination of benzene. To increase its electrophilicity and make it reactive, the catalyst is used. There is no reaction possible without the Lewis acid as a catalyst.
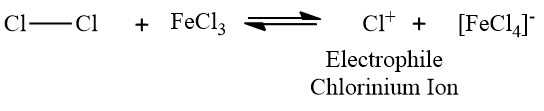
3. Why is a metal catalyst required during benzene halogenation?
Answer: Electrophilic aromatic substitution is demonstrated by halogenation. In electrophilic aromatic substitutions, a benzene is attacked by an electrophile, resulting in hydrogen substitution. However, halogens are insufficiently electrophilic to break the aromaticity of benzenes, which must be activated by a catalyst.
4. Why isn't fluorine used in the electrophilic substitution of benzene via halogenation?
Answer: Because the intermediate formed in the first step is a halonium ion, fluorine is not used in the electrophilic substitution of benzene via halogenation. It is nearly impossible to obtain a positive charge on a fluorine atom.


