-
Call Now
1800-102-2727
Group Displacement Law and Its Limitations- Alpha Decay, Beta Decay and Gamma Decay, Group Displacement Law, Practice Problem and FAQs
Some of the most useful discoveries are accidental and unexpected. For example, a little amount of uranium compound wrapped in black paper was kept in a drawer that also contained photographic plates. Later examination of these plates revealed that an exposure had occurred, from the uranium. The term "radioactive decay" was used to describe this new phenomenon.
Some elements constantly disintegrate from their parent elements in an attempt to stabilize themselves by releasing a significant amount of energy in the form of radiation and changing into a different element.
Now, You understand what radioactivity is.
But, what are these Radioactivity and Radioactive decay?
Let's clarify what it is. An atomic nucleus is made of protons and neutrons. Nuclear instability causes the atom's nuclei to break or decay and release particles with energy during radioactivity.
Let us understand the decay particles and their effect on atomic number and atomic weight leading to what is called as group displacement law in the periodic table.
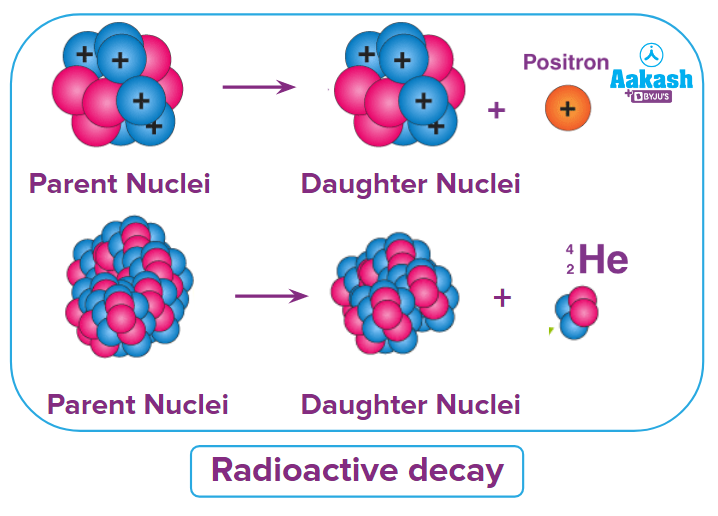
Table of Contents
- Alpha decay
- Beta decay
- Gamma-Decay
- Group Displacement Law
- Limitations of Group Displacement Law
- Practice Problems
- Frequently Asked Questions
During radioactive decay, the radioactive atom releases particles like helium, electrons and electromagnetic radiations. Let us understand what different types of decays are there.
Alpha decay
- An energetic He-nucleus with mass number 4 and atomic number 2 is known as an alpha particle. It has a +2e positive charge.
- When heavy materials like uranium U-235 experience radioactive decay, alpha particles are released.
- The atomic number falls by 2 while the mass number drops by 4 during decay.
Beta decay
- When a positron or electron is emitted from the radioactive nucleus, beta () decay is said to have taken place.
- The electron's antimatter, the positron , has the same mass as it does but the opposite charge.
- The electrons and positrons released during the - decay do not leave the nucleus directly.
- Similar to how photons are released when an electron changes from a higher energy level to a lower energy level, they are produced at the moment of emission.
Gamma-Decay
A radioactive atom's -ray emission has no impact on its atomic weight or number. Its place in the periodic table remains unaffected as a result. In natural radioactivity, radiation is followed by either emission or absorption.
According to Soddy, Fajans, and Russell's (1911–1913) observations, when a -particle is lost, a new element is created that has an atomic number of 2 and a mass number that are reduced by 4. Similar to this, a new element with an atomic number larger than 1 is obtained when the - particle is lost. The element that emits the particle is referred to as the parent element, and the newly produced element is referred to as the daughter element. This is a brief introduction to Group displacement law
Group Displacement Law
This law informs us about a radioactive element's weight reduction and atomic number:
The resulting new nuclei, which are produced from previous radioactive elements, have a smaller atomic number and atomic weight.
The atomic number and atomic weight of the newly generated element are reduced by 2 and 4 units, respectively, if -particle is produced by the nucleus of a radioactive element.
The chemical elements are organised in order of increasing atomic number, or the number of protons in a nucleus of the atom, which typically corresponds with atomic weight, from left to right and top to bottom in the periodic table.
So, based on this, we can conclude that the newly produced element will be two groups to the parent element’s left on the periodic table on emitting an alpha particle.
A radioactive element that releases -particle forms an element with one atomic number higher than the parent element. The new nuclei that are created from previously radioactive elements have an increased atomic number but a constant atomic weight. So, based on this, we can conclude that the newly produced element will move one group to the parent element’s rightside on the periodic table.
According to the group displacement law, the daughter element occupies a position in the periodic table that is either to the left or right of that of the parent element.
For alpha particle
Consider the element , whose mass number is A and atomic number Z. It changes into a daughter nucleus Y after decaying.
Mass of the parent nucleus,
Mass of the daughter nucleus,
Mass of the alpha particle , me- the mass of the electron in amu.
Example of a - decay: decays to Thorium emitting an particle.
So, the daughter element ( ) occupies a position in the periodic table that is two positions left of that of the parent element ( ) in this case.
For beta particles
Consider the element , whose mass number is A and atomic number Z. It changes into a daughter nucleus Y after decaying.
Mass of the parent nucleus,
Mass of the daughter nucleus,
Mass of the beta particle , me- the mass of the electron in amu.
Example of a decay: decays to emitting an - particle.
92U238 93Np238++1e0
So, the daughter element ( ) occupies a position in the periodic table that is one position right from that of the parent element ( ) in this case.
Limitations of Group Displacement Law
Group displacement Law explains the changes in the atomic nuclei on alpha and beta particle emissions. But there are also other decay processes possible for radioactive nuclei. For example, positron emission and K or L-electron capture are two different decay processes that occur in radioactive isotopes. But both of them result in a daughter nuclei having one atomic number less than the parent nuclei. Can the two daughter elements be the same or different is not considered by the group displacement law.
Practice Problems
Q1. Which of the following options provides the daughter element's proper position using Group displacement law?
- 2 position right in case of decay and 1 position left in case of decay
- 2 position left in case of decay and 1 position left in case of decay
- 2 position right in case of decay and 1 position right in case of decay
- 2 position left in case of decay and 1 position right in case of decay
Solution: The atomic number is reduced by two as a result of alpha emission. This results in shifting the position to the left upon the emission of the daughter element of the - particle.
The atomic number rises by one due to beta emission. This results in shifting the position to the right in response to the emission of the daughter element of the - particle.
Hence, the correct answer should be the option (D).
Q2. If the radioactive nucleus releases -particle followed by two -particles in the subsequent two changes, what will be the new element?
A. An element one step right to the parent element
B. An element one step left to the parent element
C. An element two-step left to the parent element
D. Isotope of parent element
Solution: If the radioactive nucleus emits a -particle followed by two -particles in the following two changes, the resultant element will be an isotope of the parent element. One alpha particle pushes two positions to the left and two beta particle emission pushes two positions to the right. These two movements nullify each other. The new element will have the same atomic number but less in mass by four units. Hence the new element will be an isotope of the parent element. The atomic number of the first and new elements will match.
For example: If the parent nuclei is
Hence, the atomic number of the first and new elements will remain the same.
Hence, the correct answer should be the option (D).
Q3. What will be the mass of the beta particle if undergoes beta decay?
A. 2me
B. -me
C. me
D. 3me
Solution: Given the element , whose mass number is 238 and atomic number 92. It changes into a daughter nucleus Np after decaying.
Mass of the parent nucleus,
Mass of the daughter nucleus,
Mass of the beta particle =2Me
Hence, the correct answer is the option (A).
Q4. What will be the mass of the alpha particle if undergoes alpha decay?
Solution: Given about the element , whose mass number is A and atomic number Z. It changes into a daughter nucleus Y after decaying.
Mass of the parent nucleus,
Mass of the daughter nucleus,
Mass of the alpha particle
Hence, the correct answer is the option (C).
Frequently Asked Questions
Q1. Who discovered radioactivity?
Answer: Henry Becquerel and Marie Curie conducted the first radiation experiments in 1896 while experimenting with a mineral called pitchblende. Madam Curie received the Nobel Prize in 1903 for her exceptional work in the field of radioactivity.
Q2. Name a few applications for radioactivity.
Here are a few applications for radioactivity:
A. It is applied to home smoke detectors
B. It is used to disinfect medical equipment
C. It is employed to detect and cure diseases
D. It is used to generate electricity
Q3. Is radioactive water safe to drink?
Answer: Radiation exposure at various doses has various health impacts. You are exposed to daily very low levels of radiation if you drink water that contains radionuclides. If you regularly consume radioactive water for many years, your risk of developing cancer increases.
Q4. What radioactive decay is the worst?
Answer: Compared to beta and gamma particles, alpha particles are the most dangerous internal threat. The most hazardous methods of exposure to radioactive elements that release alpha and beta particles are ingestion, inhalation, absorption, and injection. The most damaging external threat is gamma radiation.


