-
Call Now
1800-102-2727
Atomic and Physical Properties of Group 14 Elements - Introduction, Electronic Configuration, Practice Problems and FAQs
We all know that diamonds are used in jewellery and it's a pretty costly stone to buy. Moreover, it is one of the hardest known substances to humans, hence it has applications in the cutting industry. But do you know what a diamond is made up of? Looking at its demand and cost it looks like it must be made up of some very rare element present in the earth's crust but you will be amazed to know that it is made up of carbon which is one of the essential elements in all living organism. Due to its properties like shiny lustre, durability, non-reactive nature and strength, it is generally costlier than other forms of carbon. There are other elements too which belong to the same family. Let us see the importance of carbon and other elements and their properties to understand those elements.

Table of Contents
- Introduction to Group 14
- Electronic Configuration of Elements of Group 14
- Physical Properties of Group 14 Elements
- Practice Problems
- Frequently Asked Questions-FAQs
Introduction to Group 14
Group 14 elements belong to the IV A group of p-block of the modern periodic table, the elements belonging to this group include carbon, silicon, germanium, tin and lead. These elements together constitute a group and are named as carbon family (the name given in accordance with the first member of the group).
Carbon is found in the free state in the form of diamond, graphite, coal etc. Silicon is the 2nd most abundant element in the earth’s crust after oxygen it also exists in the form of silicates and silicons. Germanium is a rare element. Tin and lead do not generally exist in the free state but exist in the form of ore like cassiterite (SnO2) which is also known as tin stone and galena (PbS) which contain lead.
Electronic configuration of elements of Group 14
All the elements of the group 14 have the same outermost electronic configuration. In general, it can be represented as ns2 np2, where ‘n’ stands for the valence shell number.
|
Element |
Atomic number |
Electronic configuration |
|
Carbon |
6 |
|
|
Silicon |
14 |
|
|
Germanium |
32 |
|
|
Tin |
50 |
|
|
Lead |
82 |
As can be seen from the electronic configuration, the outermost shell of the group-14 elements has 4 electrons; thus, their valency is 4. In order to stabilize their valence p-orbital, they need four more electrons. So, removing and gaining 4 electrons is quite difficult, the elements of this group especially carbon and silicon form covalent bonding by sharing the valence electrons. As the last electron lies in the p-orbital, it also justifies its position in the p-block of the periodic table.
Physical Properties of Group-14 Elements
Physical state of group-14 elements
All the elements belonging to this group are in the solid state but possess different colours. Carbon is black, silicon appears to be light brown, and germanium is greyish white. Whereas tin, and lead are lustrous and silvery-white in appearance.
Oxidation state of group-14 elements
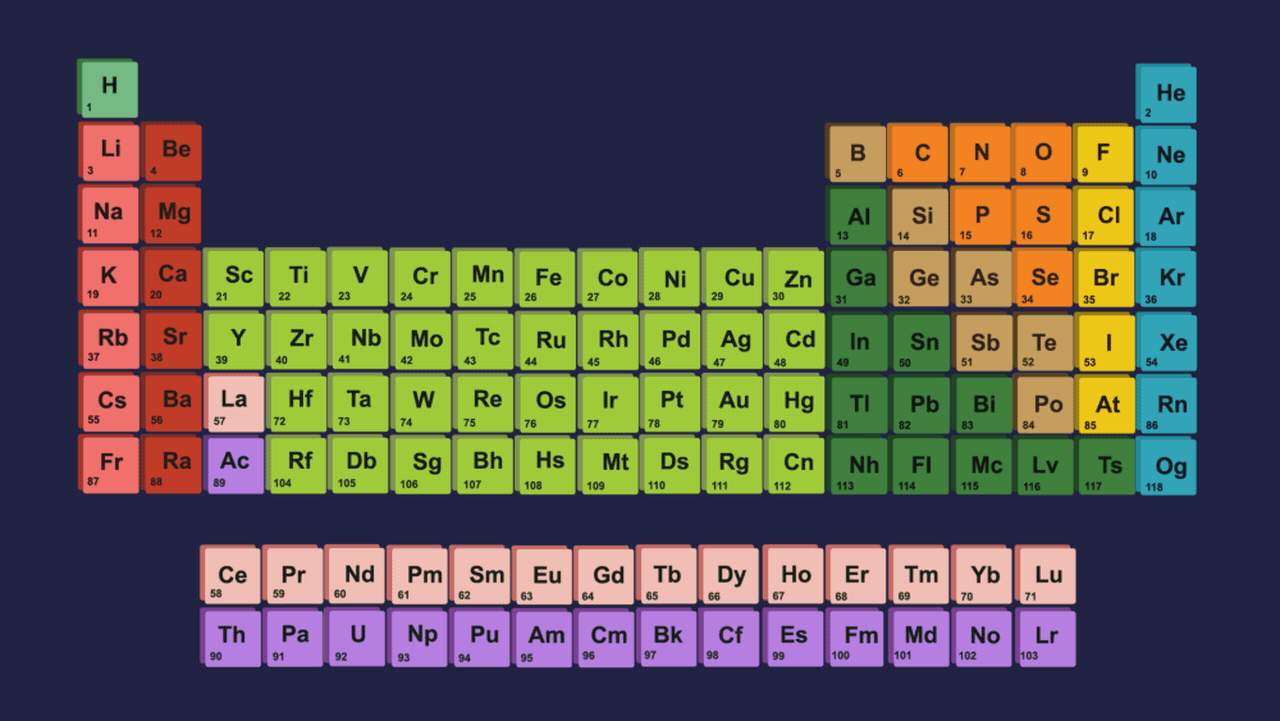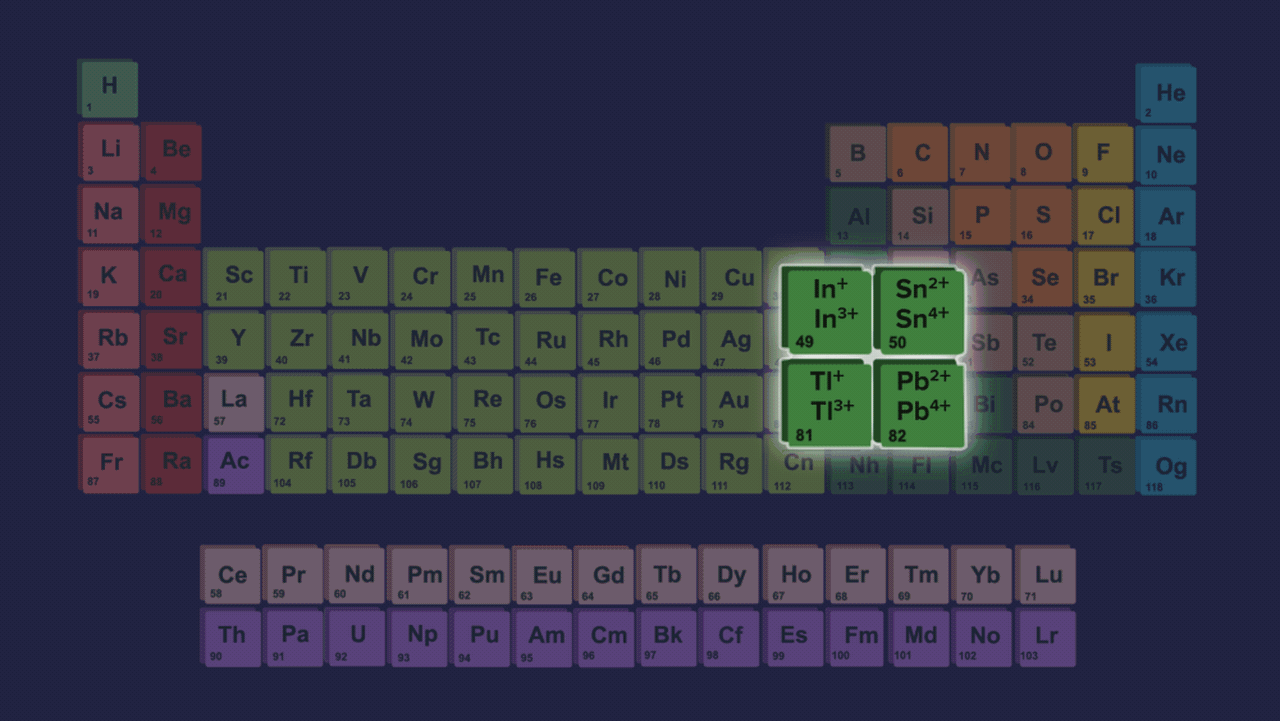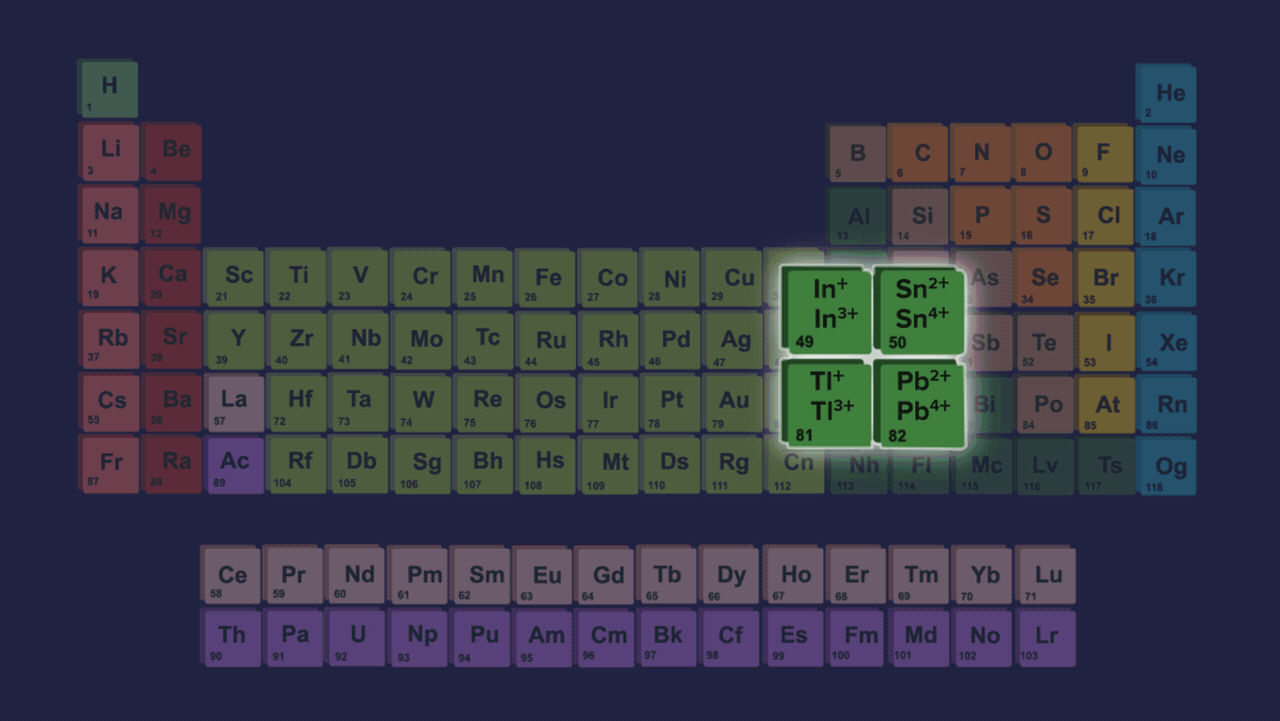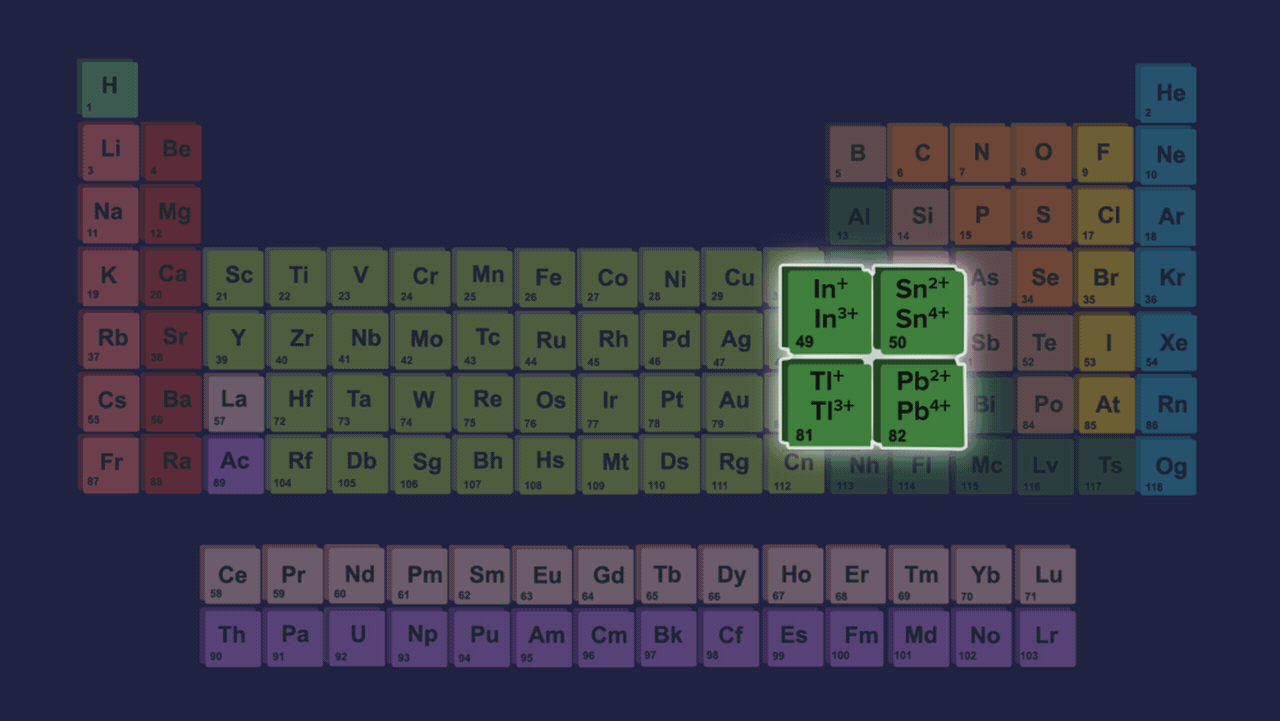
- Element present in this group shows two different types of oxidation states +2 and +4. However the stability of +2 the oxidation state increases on moving down the group and the stability of +4 the oxidation state decreases down the group due to the inert pair effect.
- The inert pair effect describes the unwillingness of s-subshell electrons to participate in chemical bonding. The effective nuclear charge pulls valence electrons present in the s-subshell tightly if we talk about elements down the group in group 14 due to poor shielding of d and f-orbitals, limiting their participation in bonding.
- Inert pair effect starts from (here ‘n’ belongs to period number) due to poor shielding of d-orbital electrons which starts to fill in the 4th period but is significant in the 6th and 7th period of the periodic table as electrons are filled in the f-subshell.
- Both +2 and +4 oxidation states have been seen in the case of germanium. In the case of tin and lead, compounds of Pb2+ & Sn2+ are more stable than Pb4+ & Sn4+ compounds but from carbon to tin, compounds which have +4 oxidation state are more stable than +2 oxidation states compounds.
Order of stability of (+2) oxidation state: C<Si<Ge<Sn<Pb
Order of stability of (+4) oxidation state: C>Si>Ge>Sn>Pb
![]()
Atomic and ionic radius of group-14 elements
- Atomic radius of the group-14 elements increases on moving down the group from carbon to lead because of an increase in the number of shells due to which the distance between the nucleus and valence shell electron increases resulting in an increase in size.
The order of atomic radius of group-14 elements: C<Si<Ge<Sn<Pb
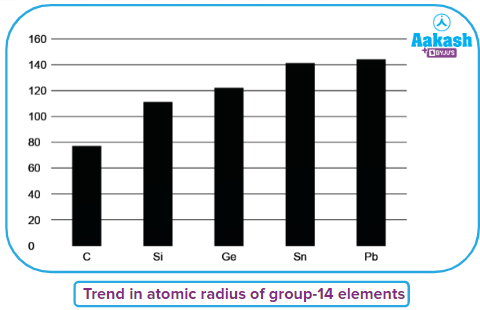
- The difference in the atomic size as we move from silicon to germanium and tin to lead decreases because of poor shielding of d and f-orbital electrons filled in the case of germanium and lead respectively which leads to the stronger attraction of valence shell electron and contraction in the size of atoms happens.
- The same trend is observed in the case of the ionic radius of an element belonging to this group with both +2 and +4 oxidation state.
The order of ionic radius with (+2) oxidation state:
The order of ionic radius with (+4) oxidation state:
Note: It is important to note that the shielding effect of d-orbitals electrons is more than the f-orbitals electron.
The order of screening effect is s > p > d > f
Density of group-14 elements
- Density of the element gradually increases from silicon to lead (except carbon) due to a greater increase in the atomic weight of the element as compared with its atomic volume which also increases due to an increase in atomic size.
The order of density of group-14 elements is Si<Ge<Sn<Pb
- In the case of carbon that exists in the form of diamond has a higher density as compared with the silicon element and graphite has a lesser density than silicon.
Melting point of group-14 elements
- Melting point of group 14 element decreases from carbon to tin but in case of lead melting point slightly increases.
- The melting point of lead is more than tin but less than germanium.
The order of melting point of group-14 elements is C>Si>Ge>Pb>Sn.
- Melting point depends upon the packing and small size of atoms get packed tightly. Therefore, more energy will be required to overcome the attraction force present between the molecules and therefore the melting point increases.
Boiling points of group-14 elements
- Boiling point of group-14 elements present in the group-14 gradually decreases from carbon (C) to lead(Pb)
The order of boiling point of group-14 elements is C>Si>Ge>Sn>Pb.
Electronegativity of group-14 elements
- Electronegativity of the elements present in group-14 decreases from carbon to silicon but electronegativity of other elements present in this group from silicon to lead remain almost constant.
The order electronegativity of group-14 elements is .
- Electronegativity of elements from silicon to lead remains almost constant because 10 electrons are added in the valence d-subshell of germanium & tin and 14 electrons are added in the valence shell of lead (Pb) and due to poor shielding of d and f-orbital electrons, electronegativity does not decrease and remains constant.
Ionisation enthalpy of group-14 elements
- Ionisation energy of group-14 element decreases from carbon to tin but in the case of lead, it is slightly greater because lanthanide contraction takes place in case of lead due to poor shielding of 14 f-subshell electron due to which the valence electrons are strongly attracted towards the nucleus and more energy is needed to remove the valence shell electrons.
The order of ionisation energy of group-14 elements is C>Si>Ge>Pb>Sn
- The difference in the ionisation energy decreases from silicon to tin because in the case of germanium & tin, 10 electrons are filled in the valence shell of an atom and due to poor shielding of d-orbital electrons, attraction force increases.
Catenation power
- It is defined as the combining power of an element to form a long chain, the branched-chain or cyclic compound is known as catenation power.
- The catenation power of elements present in the group decreases from caron to germanium and elements present below germanium have very low catenation power.
The trend in the catenation power of group-14:
- Carbon has very high catenation power in its group because carbon atoms form multiple bonds, non-availability of vacant d-orbital, small size and high C-C bond energy.

![]()
Allotropy
- The occurrence of an element in more than one form having the same chemical properties but different physical properties are known as allotropy. Allotropy exists due to the difference in the arrangement of constituent atoms.
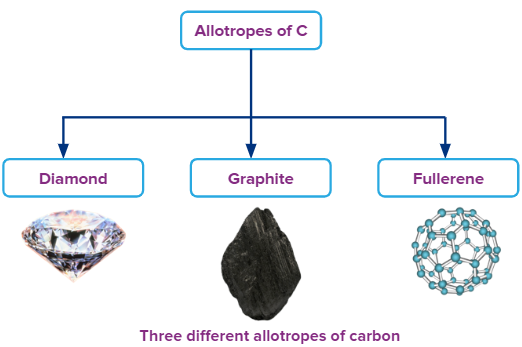
- Except for lead all other elements show allotropy. Carbon forms a large number of allotropes and is broadly classified into amorphous form- coal, charcoal, wooden charcoal, lamp black etc. and crystalline form- diamond, graphite and fullerene.
- Graphite is thermodynamically more stable than diamond. Conversion of diamond into graphite is slow with time as diamond is kinetically more stable than graphite.
Metallic and non-metallic character
- Metallic property of an element is determined by the tendency to loosen electrons and vice-versa for the non-metallic character of an element.
- On moving down the group ionisation energy generally decreases except for some abnormality and therefore the tendency to lose electrons also increases on moving down the group and hence metallic character increases.
- Carbon and silicon are non-metal, whereas germanium is metalloid. But in the case of tin and lead, they exhibit metallic properties.
Practice problems
Q1. Which of the ion among the given options is most stable.
A. Sn2+
B. Pb2+
C. Ge2+
D. Si2+
Answer: (B)
Solution: As we know the element present in group-14 shows two different types of oxidation states +2 and +4. However, the stability of +2 the oxidation state increases on moving down the group and the stability of +4 the oxidation state decreases due to the inert pair effect.
Compounds having oxidation state are more stable than compounds having oxidation state .but from carbon to tin (+4) oxidation state compounds is more stable than (+2) oxidation states compounds.
Q2. Select the correct trend for the mentioned properties of the group-14 element.
A. C>Si>Ge>Pb>Sn (Electronegativity)
B. C<Si<Ge>Sn<Pb (atomic size)
C. C>Si>Ge>Pb>Sn (ionisation enthalpy)
D. C>Si>Ge>Sn>Pb (Melting point)
Answer: (C)
Solution: Electronegativity of the elements present in group-14 decreases from carbon to silicon but electronegativity of other elements present in this group from silicon to lead remain almost constant.
The trend in the electronegativity of group-14:
Atomic radius of the group-14 element increases on moving down the group from carbon to lead because of increase in the number of shells.
The trend in the atomic size of group-14:
Ionisation energy of group-14 elements decreases from carbon to tin but in the case of lead, it slightly increases because of lanthanide contraction.
The trend in the ionisation energy of group-14:
Melting point of group 14 element decreases from carbon to tin but in the case of lead melting point slightly increases. The melting point of lead is more than tin but less than germanium.
The trend in the melting point of group-14:
Q3. Select the correct option to identify the reason for the high catenation power of carbon atom as compared with other elements present in the group.
A. High bond dissociation energy of C-C bond
B. Multiple bond-forming tendency
C. Small size of carbon atom
D. All of the above
Answer: (D)
Solution: Carbon has very high catenation power in its group because carbon atoms form multiple bonds, non-availability of vacant d-orbital, small size and high C-C bond energy.
Q4. Select the correct option for the element that will not exhibit the show allotropy.
A. Carbon
B. Lead
C. Tin
D. Silicon
Answer: (B)
Solution: Except lead all other elements show allotropy. Carbon forms a large number of allotropes and is broadly classified into amorphous form- coal, charcoal, wooden charcoal, lamp black etc. and crystalline form- diamond, graphite and fullerene.
Frequently asked question-FAQ
Q1. Why carbon monoxide is a good reducing agent but PbO does not possess the reducing power?
Answer: Carbon monoxide is a very good reducing agent because in the case of carbon monoxide molecule oxidation state of carbon is (+2) and we know that for carbon atom is more stable in a higher oxidation state (i.e (+4)) therefore it gets oxidise to form carbon dioxide molecule and act as a reducing agent. Whereas, in the case of lead +2 oxidation state is more stable due to the inert pair effect and hence PbO is stable and does not get oxidised.
Q2. What is the effect of the inert pair effect on the properties of an element?
Answer: The inert pair effect describes the unwillingness of s-subshell electrons to participate in chemical bonding. The effective nuclear charge pulls valence electrons present in the s-subshell tightly down the group due to poor shielding of d and f-orbitals, limiting their participation in bonding. Due to the inert pair effect, the stability of the compound also changes.
Q3. List some important uses of the elements of the carbon family.
Answer: Some important uses of the elements of the carbon family are:
- Graphite is used in making pencil lead
- Graphite is used as a lubricant in machinery
- Coal is used as a fuel, for manufacturing coal gas.
- Lead is used as a useful alloy such as solder
- Tin is used for the deposition over iron or steel and is known as tin plating
Q4. Why is the name of the element with the atomic number 114 different from the other elements present in the group?
Answer: The element with an atomic number greater than is an artificial element which is named according to the IUPAC nomenclature. In the case of atomic number =, 114 it is named ununquadium with the symbol of the element as Uuq.


