-
Call Now
1800-102-2727
Factors Affecting Adsorption of Gases on Solids - Adsorption of Gases on Solids, Practice Problems and FAQs
I mean, who doesn't adore gold? Even the most dormant of hearts may become excited at the sight of gold jewellery. Women, in particular, take tremendous pride and style in wearing gold jewellery. The gold jewellery may eventually stop shining and won't be as good as it once did, though. It implies that a complete cleaning and polishing treatment is required for the gold ornaments.

But what causes gold to lose its glossy surface?
Essentially, this is the adsorption of gases onto the surface of gold, toning down its lustre.
Let's talk about the factors that affect the adsorption of gases on solids.
Table of Contents
- What is an Adsorption?
- Adsorption of Gases on Solids
- Factors Affecting Adsorption of Gases on Solids
- Practice Problems
- Frequently Asked Questions
What is an Adsorption?
There are numerous instances that show how a solid's surface has a tendency to attract in and hold onto the molecules of the phase it comes in contact with. These molecules don't penetrate further into the bulk; they simply stay on the surface.
Adsorption is the concentration of atoms or molecules at the surface of a solid or liquid instead of within its bulk. Adsorption is a process in which a substance builds up on a surface in increasing concentrations of specific molecular species.
There are two components necessary for the adsorption process:
Adsorbate: A material that adheres to another substance's surface.
Adsorbent: Adsorbate adsorbs on the surface of a substance known as an adsorbent.
Adsorption of Gases on Solids
When the adsorbate is gas and the adsorbent is solid, this kind of adsorption is known as the adsorption of gas on solids. Adsorption of gases on solids largely occurs in two different ways.
Adsorption is known as physical adsorption or physisorption when gas builds up on the surface of a solid as a result of weak Van der Waals forces. Chemical adsorption, also known as chemisorption, is the process whereby gas molecules or atoms are adhered to a solid surface by chemical bonding.
Factors Affecting Adsorption of Gases on Solids
To some degree, almost all solids absorb gases. The following factors have an impact on how much a gas adsorbs on a solid surface:
- Nature of the gas
- Nature of adsorbent
- Surface area of the adsorbent
- Effect of temperature
- Effect of pressure
- Activation of adsorbent
Nature of the gas:
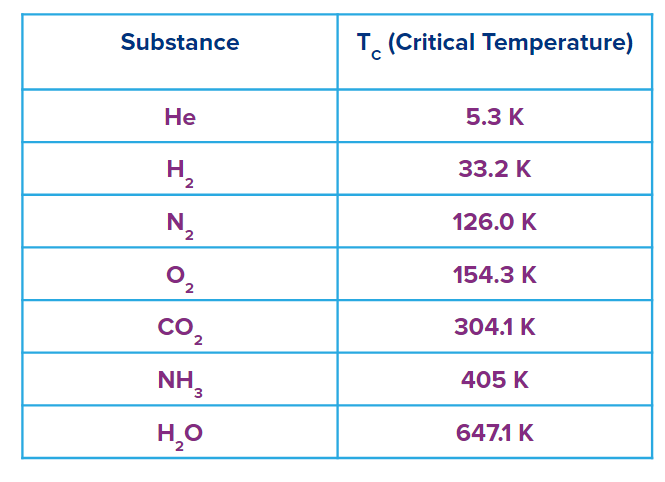
The type of gas will determine how much gas a solid can absorb. In general, the easily liquefiable gases like CO2, NH3 & H2O are adsorbed more than the permanent gases like H2, N2 & O2 under the specified temperature and pressure conditions.
A gas's critical temperature (Critical temperature is the lowest temperature above which gas cannot liquefy, regardless of how much pressure is applied) determines how easily it can liquefy and ultimately can adsorb on solids.
Nature of adsorbent:
The type of adsorbent will determine how much of the gas will be absorbed on its surface. Gases that are easily liquefied can be adsorbed by activated charcoal. Charcoal is able to adsorb a variety of toxic gases. As a result, it is employed in gas masks to absorb these toxic gases.
Typically, gases like H2, N2 & O2 can be adsorbed on transition metals with finely split particles, such as Ni and Co. Activated charcoal, finely divided metals, metal oxides (silica gel, aluminium oxide, etc.), and clay are the most often utilised adsorbents. Each of these has unique adsorption characteristics.
Surface area of the adsorbent:
With an increase in the surface area of the respective adsorbents, the extent of adsorption also increases. This clearly means the capability of a solid to adsorb would increase with increasing surface area.
As a result, the porous and finely divided adsorbent forms may adsorb a lot of adsorbates. Because porous surfaces can provide more binding sites for gas as compared to plane surfaces. To allow the gas molecules to pass through, the pore size of the adsorbent must be sufficiently large.
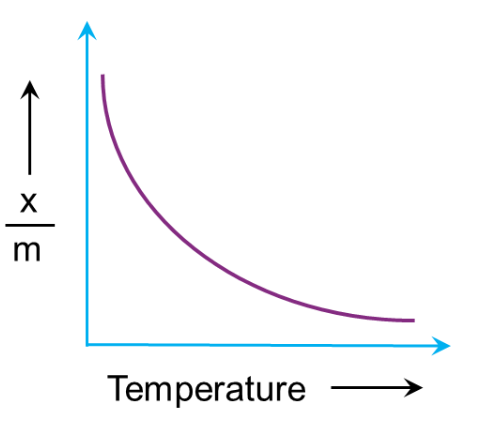
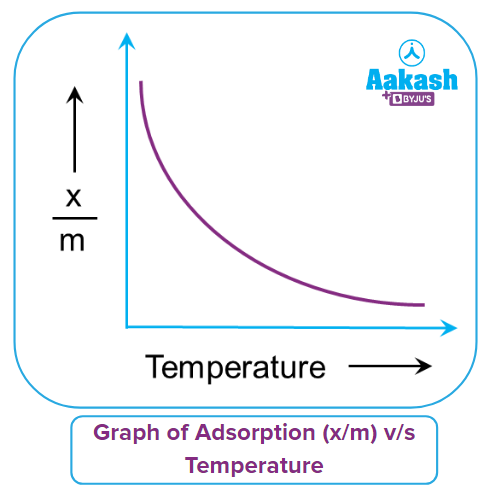
Effect of temperature:
Adsorption is an exothermic process that almost always results in heat emission. Adsorption is a process that involves a real equilibrium, just like any other equilibrium. Condensation (or adsorption) of the gas molecules on the surface of the adsorbent material and evaporation of the adsorbed gas molecules from the solid surface into the gaseous phase are the two opposing processes at play. Condensation is an exothermic process, whereas evaporation is an endothermic process in reverse.

The le-Chatelier principle states that a decrease in temperature will favour adsorption as the forward reaction is exothermic in nature and vice-versa. Hence, adsorption decreases with an increase in temperature and can be represented in a graph. Here, adsorption extent is typically expressed as x/m, where x is the mass of the adsorbate and m is the mass of the adsorbent.
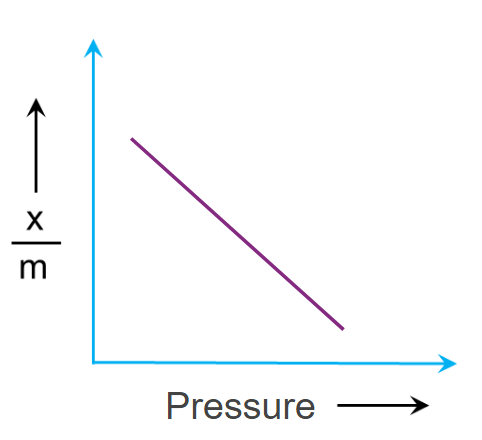
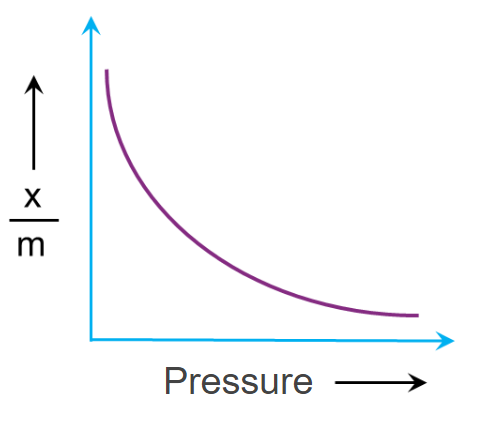
Effect of pressure:
An adsorption isotherm is a relationship between the amount of substance adsorbed by the adsorbent and the equilibrium gas pressure at a constant temperature.
Adsorption extent is typically expressed as x/m, where x is the mass of the adsorbate and m is the mass of the adsorbent. Freundlich calculated the variation of adsorption extent (x/m) with pressure (p) at a given temperature and known as Freundlich Adsorption Isotherm.
Here k & n are constants and value of 1n is generally less than one and hence the value of n should be greater than one.
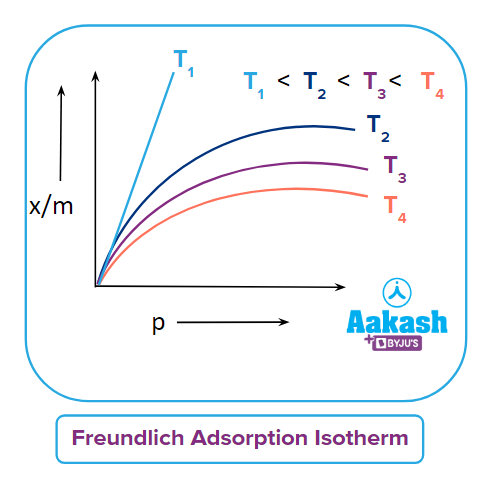
The adsorption of a gas increases with pressure when the temperature remains constant. On the basis of Le-Chatelier's principle, the adsorption of gas causes a decrease in pressure. Hence, when pressure increases, in order to counteract, adsorption also increases. As a result, a drop in temperature and a rise in pressure tends to increase the amount of gas adsorbing on a solid.
Activation of adsorbent:
Adsorbent activation refers to an increase in the adsorbing capacity of the adsorbent. Increasing the rate of adsorption is crucial and some techniques can be used to do this.
(i) Roughening the surface of the adsorbent activates metallic adsorbents. This is accomplished through mechanical rubbing, chemical interactions, or electroplating, which deposits finely scattered metals on the surface of the adsorbent.
(ii) Adsorbents are broken into tiny bits to boost their absorption capacity. As a result, the surface area grows, which also enhances the adsorbing power.
(iii) Some adsorbents are heated vigorously to 623 to 1273 K in a vacuum or in contact with superheated steam to activate them and allow gases to adhere to them.
Recommended video link: 20:54 to 41:36 Surface Chemistry Class 12 | JEE Chemistry Important Topics | JEE 2022 Exam Preparations
Practice Problems
Q1. What among the following influences the amount of adsorption of a gas on a solid surface?
- Nature of the gas and adsorbent
- Effect of temperature
- Effect of pressure
- All of these
Answer: (D)
Solution:
All given factors affect the adsorption of gases on solids and are explained as:
The amount of the gas that a solid can adsorb will depend on the type of gas. It also depends on the type of adsorbent. Activated charcoal has the ability to adsorb gases that are easily liquefied. On the basis of Le-Chatelier's principle, the amount of a gas that adsorbs on a solid tends to increase as there is a decrease in temperature. On the basis of Le-Chatelier's principle, the amount of a gas that adsorbs on a solid tends to increase as there is an increase in pressure. Hence, all the given options are correct as all the given factors affect the adsorption of gases on solids. So, the correct answer is option (D).
Q2. Which of the following conditions favours the decrease in the adsorption of gases on solids?
- Low pressure and high temperature favour the decrease in the adsorption of gases on solids.
- Low pressure and low temperature favour the decrease in the adsorption of gases on solids.
- High pressure and high temperature favour the decrease in the adsorption of gases on solids.
- High pressure and low temperature favour the decrease in the adsorption of gases on solids.
Answer: (A)
Solution: The le-Chatelier principle states that a decrease in temperature will favour adsorption as the forward reaction is exothermic in nature and vice-versa. Hence, adsorption decreases with an increase in temperature and can be represented in a graph. Here, adsorption extent is typically expressed as x/m, where x is the mass of the adsorbate and m is the mass of the adsorbent.

The adsorption of a gas increases with pressure when the temperature remains constant. On the basis of Le-Chatelier's principle, the adsorption of gas causes a decrease in pressure. Hence, when pressure increases, in order to counteract, adsorption also increases. As a result, a drop in temperature and a rise in pressure tends to increase the amount of gas adsorbing on a solid.
Hence, the correct answer is option (A).
Q3. Which of the following statements is/are incorrect with respect to the adsorption of a gas on solids?
- Adsorbents are fragmented into tiny pieces to reduce their ability for absorption.
- The amount of a gas adsorbing on a material tends to decrease as temperature and pressure increase.
- It is possible to adsorb more adsorbates using flat adsorbents than with porous and finely split forms.
- All of these
Answer: (D)
Solution: All the given statements are wrong as adsorbents are divided into minute pieces to increase their potential for absorption. The amount of a gas adhering to a solid tends to increase as temperature decreases and increases with the increase in pressure. Fewer adsorbates may be adsorbed by planar adsorbents as opposed to porous and finely split types.
Hence, the correct answer is option (D).
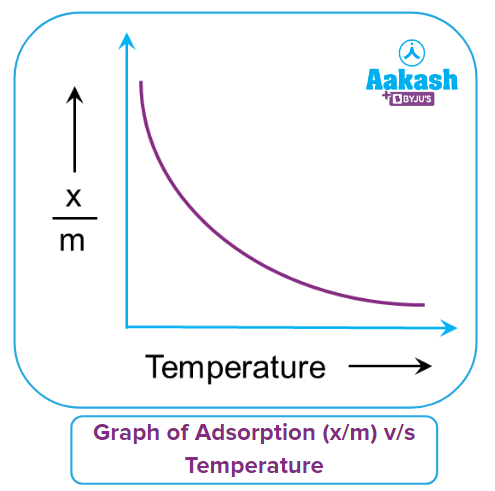
Q4. What should be the correct graphical representation of the effect of temperature on the adsorption of gases on solids?
Answer: (C)
Solution:
Adsorption is an exothermic process that almost always results in heat emission. Adsorption is a process that involves a real equilibrium, just like any other equilibrium. Condensation (or adsorption) of the gas molecules on the surface of the adsorbent material and evaporation of the adsorbed gas molecules from the solid surface into the gaseous phase are the two opposing processes at play. Condensation is an exothermic process, whereas evaporation is an endothermic process in reverse.

The le-Chatelier principle states that a decrease in temperature will favour adsorption as the forward reaction is exothermic in nature and vice-versa. Hence, adsorption decreases with an increase in temperature and can be represented in a graph. Here, adsorption extent is typically expressed as x/m, where x is the mass of the adsorbate and m is the mass of the adsorbent.
Q5. What should be the correct graphical representation of the effect of pressure on the adsorption of gases on solids?
Answer: (D)
Solution: Adsorption extent is typically expressed as x/m, where x is the mass of the adsorbate and m is the mass of the adsorbent. Freundlich calculated the variation of adsorption extent (x/m) with pressure (p) at a given temperature and known as Freundlich Adsorption Isotherm.
Here k & n are constants and the value of 1n is generally less than one and hence the value of n should be greater than one.
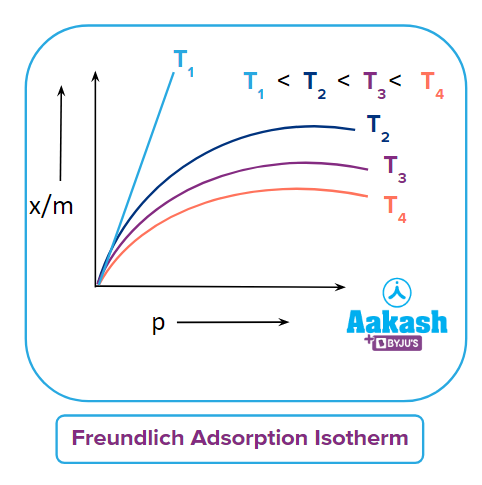
The adsorption of a gas increases with pressure when the temperature remains constant. On the basis of Le-Chatelier's principle, the adsorption of gas causes a decrease in pressure. Hence, when pressure increases, in order to counteract, adsorption also increases.
Only option (D) represents the increase in adsorption with the increase in pressure.
Hence, the correct answer is option (D).
Frequently Asked Questions
Q. What distinguishes absorption from adsorption?
Answer: We should know first what is adsorbate and adsorbent before discussing adsorption and absorption. The substance that has molecules adsorbed onto the surface is known as the adsorbate in the process of adsorption. Adsorbent refers to the substance on whose surface the process occurs. Absorption comprises the entire volume of the absorbing agent, whereas adsorption is based on the surface where a film of adsorbate has formed.
Q. Describe occlusion.
Answer: The adsorption of gases on the surface of metals is referred to as occlusion. We may therefore state that it is a sort of adsorption in which the adsorbent is a metal (solid) and the adsorbate is a gas.
Q. What in chemistry is Le Chatelier's principle?
Answer: Le Chateleir's principle states that if one or more of the determining factors (such as temperature, pressure, or concentration) change, a system's equilibrium will be disturbed. In this case, the system will adjust to a new equilibrium by limiting the effect of the change.
Q. Why is the process of adsorption exothermic?
Answer: Adsorption is an exothermic process because the adsorbent's surface particles are unstable, and as the adsorbate adheres to the surface, the adsorbent's energy reduces, leading to the evolution of heat. In light of this, adsorption is always exothermic.