-
Call Now
1800-102-2727
Functional Groups - Functional Groups, Their General IUPAC Nomenclature, Practice Problems and FAQs
Everyone enjoys having a variety of flowers and their pleasant smell. Right???
and we are all aware of how these fruits grow. To produce flowers with a variety of fragrances, colors and appearances, different seeds are buried in the soil.
Just like this, when functional groups are added to hydrocarbons in organic chemistry and H is removed, we obtain new molecules with various physical and chemical characteristics.
A functional group is an atom or group of atoms that determines the characteristics of an organic molecule. Each functional group has a distinct identity that is reflected by a unique prefix provided by IUPAC.

Table of Contents:
- Functional groups
- IUPAC Nomenclature of Common Functional groups
- Aldehydes or Alkanals
- Ketones or Alkanones
- Carboxylic acid or Alkanoic acid
- Acid halide or Alkanoyl Chloride
- Acid anhydrides
- Ester
- Amines or Alkanamines
- Alkylcyanide group or Alkanenitrile
- Acid Amide or alkanamide
- Practice Problems
- Frequently Asked Questions
Functional groups:
In the study of organic chemistry, functional groups are the substituent atoms or groups of atoms that are joined to particular molecules.
Chemical reactions that involve two molecules of different sizes but with the same functional groups will behave in a similar way. The same functional group will behave consistently and undergo similar reactions regardless of the chemical in which it is present.
When a molecule has a functional group, it means that its behaviour and chemical reactions may be predictably expected in a systematic way. A functional group is a collection of atoms or bonds that is crucial for a substance's specific chemical reactions.
Also, by comprehending the characteristics of various functional groups, the purposeful execution of chemical reactions to produce a particular compound can be structured.
IUPAC Nomenclature of Common Functional groups:
The common functional groups are discussed in this subsection that must be used in their nomenclature.
In IUPAC, there are three parts to the name of an organic compound:
(i) Word Root
(ii) Prefix
(iii) Suffix
Word root denotes the number of carbon atoms present in the carbon chain.
|
Chain length |
Word root |
Chain length |
Word root |
|
C1 |
Meth |
C7 |
Hept |
|
C2 |
Eth |
C8 |
Oct |
|
C3 |
Prop |
C9 |
Non |
|
C4 |
But |
C10 |
Dec |
|
C5 |
Pent |
C11 |
Undec |
|
C6 |
Hex |
C12 |
Dodec |
Prefix denotes those groups which are not considered as a functional group but treated as substituents in the naming of the organic compounds.
|
Substituents |
Prefix |
Substituents |
Prefix |
|
-F |
Fluoro |
-N=N- |
Diazo |
|
-Cl |
Chloro |
-OCH3 |
Methoxy |
|
-Br |
Bromo |
-OCH2CH3 |
Ethoxy |
|
-I |
Iodo |
-OH |
Hydroxy |
|
-NO2 |
Nitro |
-NH2 |
Amino |
|
-NO |
Nitroso |
There are basically two types of suffix used in the naming of organic compounds:
(i) Primary Suffix
(ii) Secondary Suffix
Primary suffix denotes the nature of linkage in carbon atoms.
If there are all single bonds present in the parent chain, Then the primary suffix ‘ane’ is used.
If there is a double bond present in the parent chain, Then the primary suffix ‘ene’ is used.
If there is a triple bond present in the parent chain, Then the primary suffix ‘yne’ is used.
Secondary suffix denotes the presence of functional groups in the organic compound.
Let us discuss these functional groups one by one–
(i) Aldehydes or Alkanals:
General formula:
Representation: 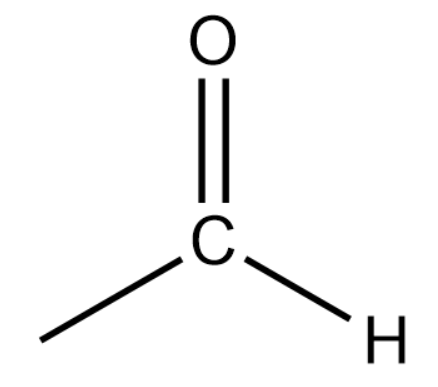
These are derived by replacing the hydrogen atom at the terminal position of a carbon atom with CHO(aldehydic group). The name of alkanals is obtained by using '-al' and replacing 'e' of corresponding alkanes.
Final Name: Alkane -e+ al=Alkanal
Remember one thing while counting the number of carbon atoms in parent chain, carbon of aldehydic group is also included in the numbering.
|
Aldehyde |
IUPAC Name |
|
HCHO |
Methanal |
|
CH3CHO |
Ethanal |
|
CH3CH2CHO |
Propanal |
|
(CH3)2CHCHO |
2-methylpropanal |
(ii) Ketones or Alkanones:
General formula:
Representation: 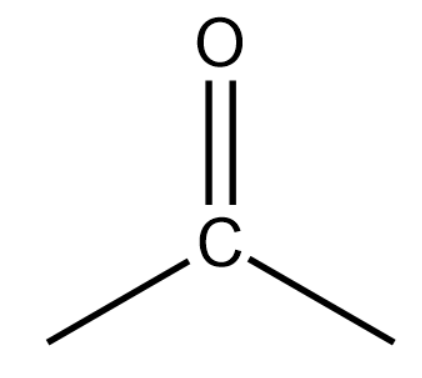
These are derived by replacing the hydrogen atom at the non-terminal position of a carbon atom with CO(ketonic group). The name of alkanones is obtained by using '-one' and replacing 'e' of corresponding alkanes.
Final Name: Alkane -e+ one=Alkanone
Remember one thing while counting the number of carbon atoms in the parent chain, carbon of the ketonic group is also included in the numbering.
|
Ketones |
IUPAC Name |
|
CH3COCH3 |
Propanone |
|
CH2CH2COCH3 |
Butanone |
|
CH3CH2COCH2CH3 |
Pentan-3-one |
|
CH3CH2CH2COCH3 |
Pentan-2-one |
|
(CH3)2CHCOCH2CH3 |
2-methylpentan-3-one |
(iii) Carboxylic acid or Alkanoic acid:
General formula:
Representation: 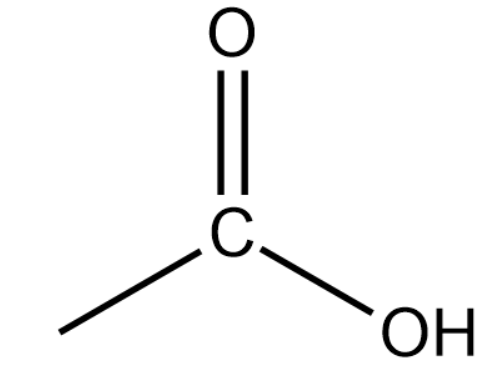
These are derived by replacing the hydrogen atom at the terminal position of a carbon atom with COOH (carboxylic group). The name of alkanoic acid is obtained by using '-oic acid' and replacing 'e' of corresponding alkanes.
Final Name: Alkane -e+ oic acid=Alkanoic acid
Remember one thing while counting the number of carbon atoms in the parent chain, carbon of the carboxylic group is also included in the numbering.
|
Carboxylic acid |
IUPAC Name |
|
HCOOH |
Methanoic Acid |
|
CH3COOH |
Ethanoic Acid |
|
CH2CH2COOH |
Propanoic Acid |
|
CH3CH2CH(CH3)COOH |
2-methylbutanoic Acid |
|
(CH3)2CHCH2COOH |
3-methylbutanoic Acid |
(iv) Acid halide or Alkanoyl Chloride:
General formula:
Representation: 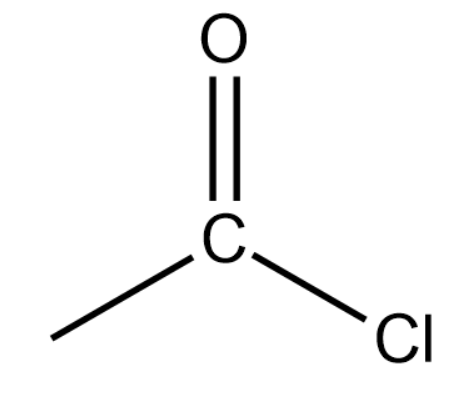
These are derived by replacing the hydrogen atom at the terminal position of a carbon atom with COCl (Acyl chloride group). The name of an acyl chloride is obtained by using '-oyl chloride' and replacing 'e' of corresponding alkanes.
Final Name:
Remember one thing while counting the number of carbon atoms in the parent chain, carbon of acyl chloride group is also included in the numbering.
|
Acyl Chloride |
IUPAC Name |
|
HCOCl |
Methanoyl chloride |
|
CH3COCl |
Ethanoyl chloride |
|
CH2CH2COCl |
Propanoyl chloride |
|
CH3CH2CH(CH3)COCl |
2-methylbutanoyl chloride |
|
(CH3)2CHCH2COCl |
3-methylbutanoyl chloride |
(v) Acid anhydrides:
General formula: R(CO)2O
Representation: 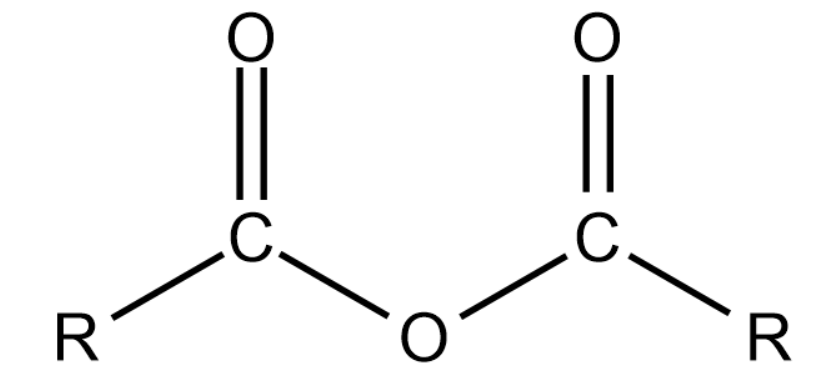
These are derived from a carboxylic acid by removing one molecule of water.
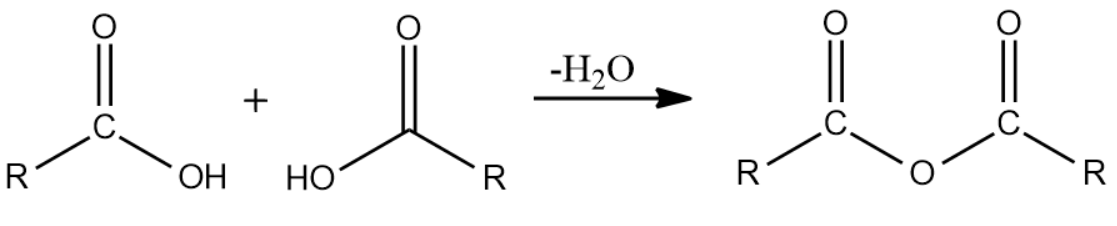
The name of an acid anhydride is obtained by using '- anhydride' and replacing 'acid' of corresponding carboxylic acid.
Final Name:
|
Acyl Chloride |
IUPAC Name |
|
HOCOCOH |
Methanoic Anhydride |
|
CH3OCOCOCH3 |
Ethanoic Anhydride |
|
CH3OCOCOH |
Ethanoic Methanoic Anhydride |
|
CH3OCOCOCH2CH3 |
Ethanoic Propanoic Anhydride |
(vi) Ester:
General formula: RCOOR'
Representation: 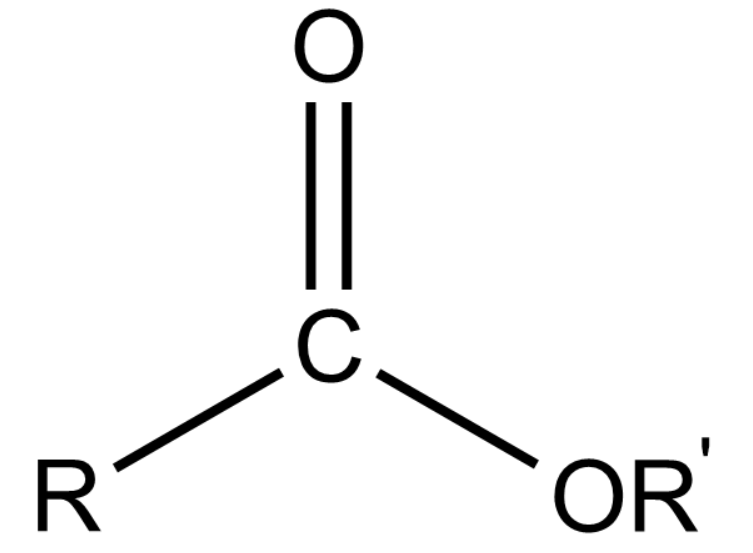
These are derived from a carboxylic acid by removing -OH group with -OR group.
The name of an ester is obtained by prefixing the name of the alkyl group before the parent chain and using '- oate' and replacing 'ic acid' of corresponding carboxylic acid.
Final Name:
|
Ester |
IUPAC Name |
|
HCOOCH3 |
Methyl Methanoate |
|
CH3COOCH3 |
Methyl Ethanoate |
|
HCOOCH2CH3 |
Ethyl Methanoate |
|
CH3COOCH2CH3 |
Ethyl Ethanoate |
(vii) Amines or Alkanamines:
General formula: RNH2
Representation: ![]()
These are derived from ammonia by removing one, two or three hydrogen atoms with alkyl substituents.
The name of an amine is obtained by using '- amine' and replacing 'e' of corresponding alkanes.
Final Name:
|
Amine |
IUPAC Name |
|
CH3NH2 |
Methanamine |
|
CH3CH2NH2 |
Ethanamine |
|
CH3CH2NHCH3 |
N-methylethanamine |
|
N(CH3)3 |
N,N-dimethylmethanamine |
(viii) Alkylcyanide group or Alkanenitrile:
General formula: RCN
Representation: ![]()
These are derived by replacing the hydrogen atom at the terminal position of a carbon atom with CN(cyanide group).
The name of an alkyl cyanide is obtained by adding '- nitrile' to the name of corresponding alkanes.
Final Name:
Remember one thing while counting the number of carbon atoms in the parent chain, carbon of alkyl nitrile group is also included.
|
Amine |
IUPAC Name |
|
CH3CN |
Ethanenitrile |
|
CH3CH2CN |
Propanenitrile |
|
(CH3)2CHCN |
2-methylpropanenitrile |
(ix) Acid Amide or alkanamide:
General formula: RCONH2
Representation: 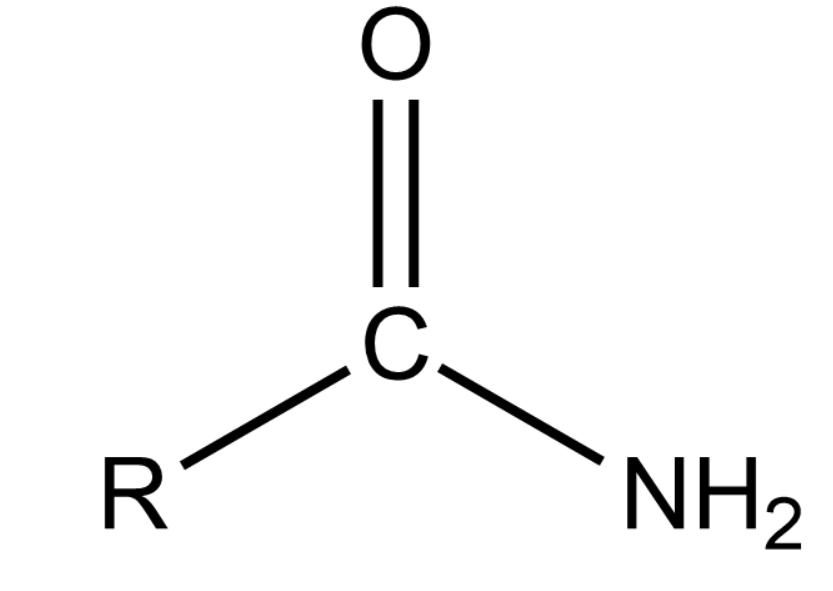
These are derived from a carboxylic acid by removing -OH group with -NH2 group.
The name of an amide is obtained by using '- amide' and replacing 'e' of corresponding alkanes.
Final Name:
|
Ester |
IUPAC Name |
|
HCONH2 |
Methanamide |
|
CH3CONH2 |
Ethanamide |
|
CH3CH2CONH2 |
Propanamide |
|
(CH3)2CHCONH2 |
2-methylpropanamide |
(x) Alcohols or Alkanols:
General formula:
Representation: ![]()
These are derived by replacing the hydrogen atom at the terminal position of a carbon atom with OH(alcohlic group). The name of alkanols is obtained by using '-ol' and replacing 'e' of corresponding alkanes.
Final Name:
|
Alcohols |
IUPAC Name |
|
CH3OH |
Methanol |
|
CH3CH2CHO |
Propanal |
|
(CH3)2CHCHO |
2-methylpropanal |
Related links: Discussion on Organic Compound and Nomenclature & Isomerisms
(23:10 to 29:46)
Practice Problems:
Q1. What of the following functional group is present in the given compound?
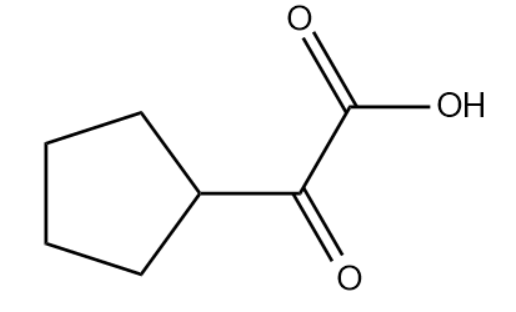
A. Acid Anhydride
B. Ether
C. Alcohol
D. Ketone
Answer: (D)
Solution: The functional group present in the molecule is the ketonic group and carboxylic acid group. Hence, the correct answer is option (D).
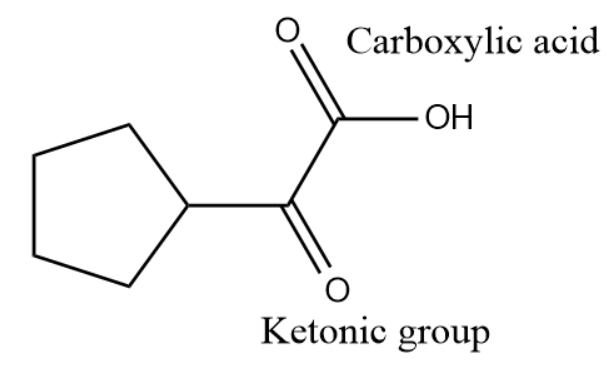
Q2. What will be the correct IUPAC name for the given compound?
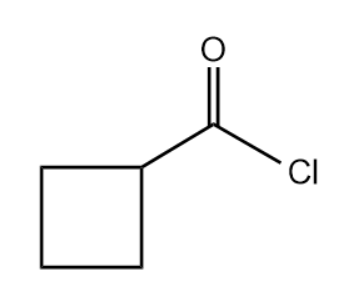
A. cyclobutanoyl chloride
B. cyclopentanoyl chloride
C. cyclobutanecarbonyl chloride
D. cyclobutylmethanoyl chloride
Answer: (C)
Solution: There is acyl chloride present in this compound which is a derivative of carboxylic acid. The substituent present with acyl chloride group is cyclobutyl group.
The correct IUPAC name can be written as cyclobutanecarbonyl chloride.
Hence, the correct answer is option (C).
Q3. What will be the correct IUPAC name for the given compound?
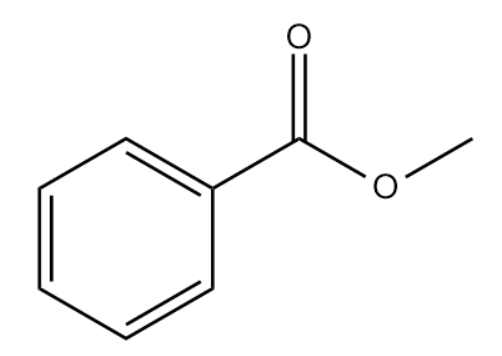
A. Phenyl Ethanoic anhydride
B. Benzyl Ethanone
C. Methyl Benzoate
D. Methyl Phenylmethanoate
Answer: (C)
Solution: The functional group given in this compound is the ester group. The name of an ester is obtained by prefixing the name of alkyl group before the parent chain and using '- oate' and replacing 'oic acid' of corresponding carboxylic acid. So, the IUPAC name can be written as Methyl Benzoate. Hence, the correct answer is option (C).
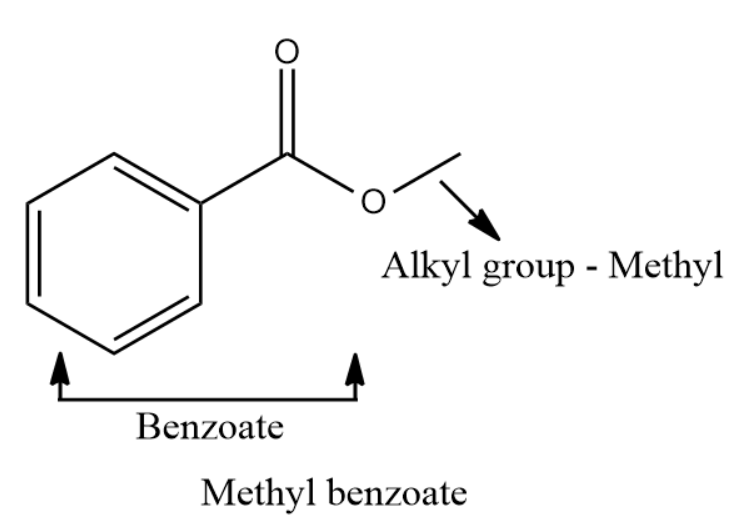
Q4. What will be the correct IUPAC name and functional group present in the given compound?
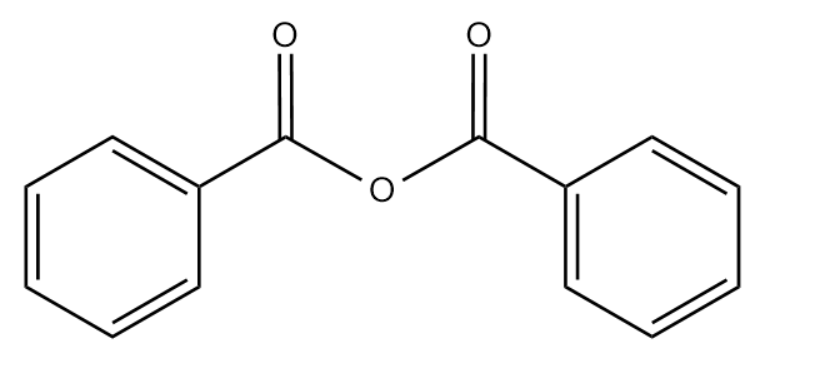
A. Benzyl Carbanoic Anhydride, Acid Anhydride
B. Benzoic Anhydride, Acid Anhydride
C. Benzyl Benzoate, Ester
D. Benzyl Benzoate, Ester
Answer: (B)
Solution: The given functional group is acid anhydride. These are derived from a carboxylic acid by removing one molecule of water.
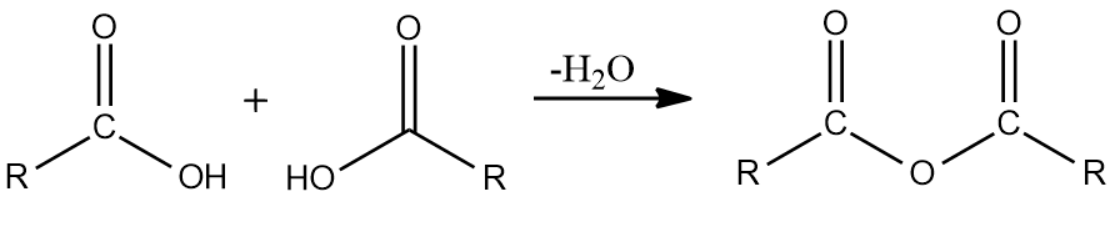
The name of an acid anhydride is obtained by using '- anhydride' and replacing 'acid' of corresponding carboxylic acid.
Final Name:
Hence, the correct IUPAC name for the given compound is Benzoic Anhydride.
The correct answer is option (B).
Frequently Asked Questions-FAQs:
Q1. What makes understanding a functional group crucial?
Answer: Chemistry places a lot of emphasis on functional groups because they are the parts of molecules that can undergo distinctive reactions. As a result, they control the chemistry and characteristics of many organic molecules. Any compound with carbon and another element is said to be organic.
Q2. Can there be more than one functional group?
Answer: Multiple functional groups are frequently present in a single molecule. In such a scenario, one functional group is regarded as the major functional group, and the other as a substitution. The choice of the functional group is made in a priority sequence.
Carboxylic acid > sulphonic acid > acid anyhydride > ester > acid halide > amide > nitrile > aldehyde > ketone > alcohols > amines
Q3. Is a functional group a double or triple bond?
Answer: Functional groups is a phrase for atoms or groups of atoms inside a molecule that are areas of relatively high reactivity including double and triple bonds. So, yes, Double or triple bonds are also functional groups.
Q4. Do functional groups possess unique characteristics?
Answer: A particular set of atoms with unique characteristics and reactivity is called a functional group. These groups will give their parent compounds numerous similarities irrespective of the additional atoms that are involved.


