-
Call Now
1800-102-2727
Fries Rearrangement: Reaction, Mechanism, Applications and Limitations of Fries Rearrangement, Practice problems & Frequently Asked Questions
Have you ever seen anyone playing with a deck of cards?

Or at least have you ever held it in your hands or seen someone doing so? If yes, then you probably know how they are initially packed inside the box. But once you start shuffling them, the order how the spades,hearts, diamonds and clubs were placed, gets changed. And now, although there still remains exactly the same number of cards (52) in your hand, the order of arrangement has definitely been altered!
Similar is the case with the bunch of rearrangement reactions in organic chemistry that we are about to study now.
Although the carbon skeleton is rearranged, the final product obtained after a rearrangement reaction has the same chemical formula. Only structural changes are seen and hence we obtain structural isomers through such reactions.
Table of content:
- Reaction of Fries rearrangement
- Mechanism of Fries rearrangement
- Applications of Fries rearrangement
- Limitations of Fries rearrangement
- Practice problems
- Frequently asked questions(FAQs)
Reaction of Fries rearrangement:
Fries rearrangement is an exciting organic chemistry reaction. The Fries Rearrangement is an organic chemistry rearrangement reaction in which an aryl ester is transformed to a hydroxy aryl ketone with the help of aqueous acid and AlCl3 catalyst.
An acyl group of the phenolic ester is transferred to the aryl ring during the Fries Rearrangement reaction. It's also worth noting that the Fries rearrangement is selective to ortho and para positions, implying that the acyl group is attached to the ortho or para position of the aryl ring. This reaction's selectivity is controlled by making specific modifications to the reaction circumstances (the reaction conditions include the temperature at which the response is produced or the solvent used for the reaction).
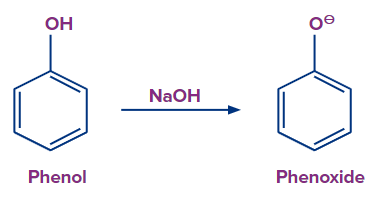
For example, phenol is first converted to phenylacetate.
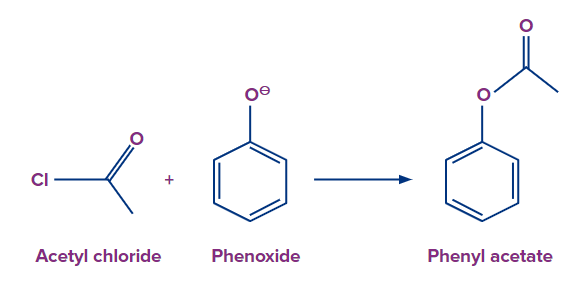
To start this reaction, phenol is treated with a base (NaOH) in the presence of pyridine to generate a phenoxide ion. And later sodium phenoxide is reacted with acetyl chloride to form an ester. NaCl is released during this process and phenylacetate is the ester which is formed in the reaction.
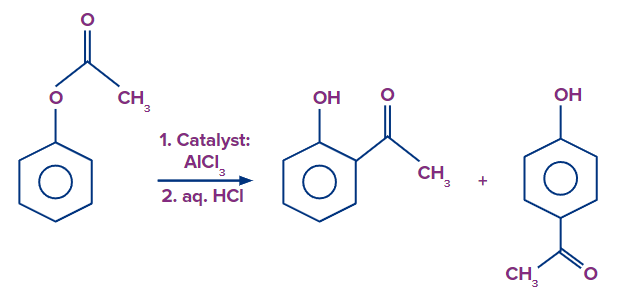
When phenylacetate is exposed to a catalyst aluminium chloride (AlCl3), it undergoes rearrangement, yielding ortho hydroxy acetophenone and para hydroxy acetophenone. This type of rearrangement is known as the fries rearrangement.
Mechanism of Fries rearrangement:
Step 1: Initially, the phenolic oxygen forms a compound with a Lewis acid catalyst (Aluminium chloride). Because phenolic oxygen has more electrons, it is a better Lewis base. As a result, the synthesis of this complex with phenolic oxygen is favoured over the formation of the complex with carbonyl oxygen. As a result, the bond between the acyl complex and the phenolic oxygen becomes polarised, causing the AlCl3 bonding to the phenolic oxygen to rearrange. This results in the creation of the acylium carbocation.
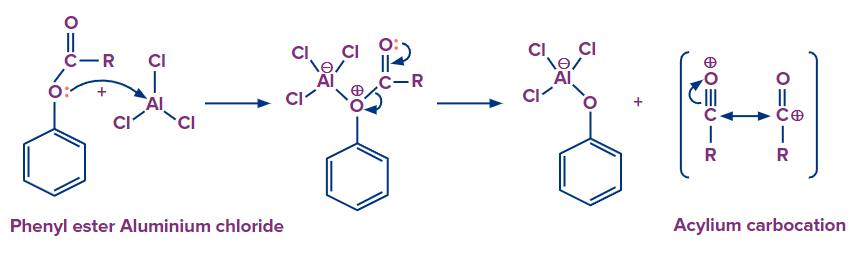
Step 2: Attack of acylium cation via electrophilic aromatic substitution followed by deprotonation to restore aromaticity.
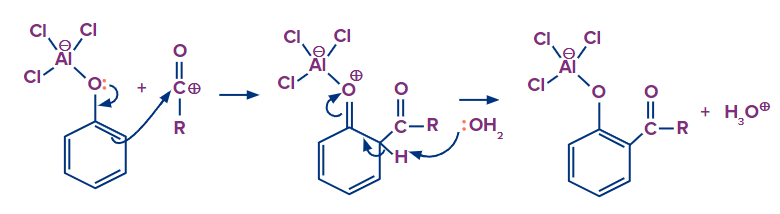
Step 3: The acid work up regenerates the Lewis acid, yielding the end product ortho hydroxy acyl ketone.
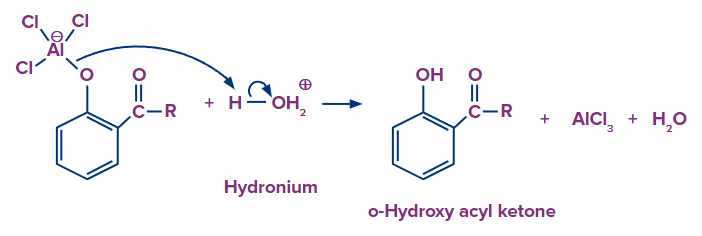
It is also critical to note that the orientation of this electrophilic aromatic substitution is strongly temperature dependent. Lower reaction temperatures make substitution at the para position easier. Temperatures that are relatively high cause ortho substitution.
The use of non-polar solvents in this rearrangement reaction further favours ortho position substitution. In this rearrangement reaction, highly polar solvents allow for para-substituted products. The fries rearrangement reaction is the name given to this particular rearrangement reaction and its mechanism.
Applications of Fries rearrangement:
The Fries rearrangement reaction has numerous applications. They are as follows:
- The Fries rearrangement reaction is required for the synthesis of o- and p-hydroxy acetophenone. Both of these chemicals are essential intermediates in the manufacturing of medicines.
- This reaction is useful in the manufacture of Tocopherol or Vitamin E.
- The Fries rearrangement reaction is required for the manufacture of ortho-acyl hydroxy [2.2] paracyclophanes, which has major industrial value.
- This reaction is used in the pharmaceutical industry to produce various thermographic materials, intermediates, and antiviral drugs.
Limitations of Fries rearrangement:
The following are the main limitations of Fries rearrangement:
- Only esters with slightly less reactive acyl components can be employed in this reaction due to the relatively severe reaction conditions.
- When highly substituted acyl components are used, yields are decreased.
- When deactivating or meta-directing groups are present on the aromatic ring, yields are similarly reduced.
Practice problems:
Q.1. In the Fries rearrangement reaction, what type of catalyst is used?
(A) Lewis acid
(B) Lewis base
(C) Halogens
(D) Acid chlorides
Answer: (A)
Solution: Aluminium chloride (AlCl3) is a Lewis acid that has empty orbitals in the central atom and can accept a lone pair of electrons and is used as a catalyst in the Fries rearrangement.
Q.2. Which isomers are formed during rearrangement reactions?
(A) Structural isomers
(B) Optical isomers
(C) Conformational isomers
(D) Geometrical isomers
Answer: (A)
Solution: Whenever rearrangement reactions occur, the molecular formula remains the same. Bond connectivity changes during rearrangement. When the molecular formula remains the same, but the bond connectivity is different. They are known as structural isomers.
Q.3. Among the following, which is a meta directing group?
(A) -NHCOCH3
(B) -OH
(C) -OCH3
(D) -COOH
Answer: (D)
Solution: The most important thing to remember is that carboxylic acid (-COOH) is an electron withdrawing group (EWG) that is more electronegative than carbon, When attached to a benzene molecule, it removes a significant quantity of electrons from the pi system benzene ring, making electrophilic substitution processes slow and difficult. As a result, -COOH is a strong meta director.
Q.4. Fries Rearrangement Reaction is selective to
(A) Ortho position
(B) meta position
(C) Para position
(D) Ortho and para positions
Answer: (D)
Solution: The acetoxy group is ortho and para directing due to the presence of lone pairs of electrons on oxygen and it will direct the incoming electrophile to ortho and para positions. So fries rearrangement is selective to ortho and para positions.
Frequently asked questions(FAQs):
Q1. What is photo-Fries rearrangement?
Answer: Some Fries rearrangement reaction variations are extremely important in chemistry. The photo-Fries rearrangement reaction is the first variation of this process. Without the use of a catalyst, phenolic esters are converted into hydroxyl aryl ketones in this organic process. UV rays act as a catalyst in this reaction.
Q2. What exactly is intramolecular rearrangement?
Answer: A rearrangement reaction is a large family of chemical reactions that involve rearranging the carbon skeleton of a molecule to produce a structural isomer of the original molecule. When a substituent moves from one atom to another inside the same molecule, these reactions are usually intramolecular rearrangement reactions.
Q3. In electrophilic aromatic substitution, how is the acetoxy group ortho and para-directing?
Answer: When it comes to electrophilic aromatic substitution, phenoxide is more reactive than phenol. However, the number of lone pairs is not a controlling factor because both lone pairs cannot be in conjugation with the benzene ring at the same time. If one lone pair is in conjugation with the benzene ring, the other lone pairs will be incorrectly orientated to overlap with the ring's pi system. So you can have one, two, or three lone pairs - it makes no difference because any lone pairs beyond the first are useless.
Only the availability of lone pairs is important. This must be explained in connection with inductive or resonance effects.There would be +I effect of - O- group which is present over the benzene ring.
Moving on to the acetoxy group -O(CO)CH3, The lone pair on oxygen is delocalised via resonance into the electron-drawing carbonyl group. Consider the -C=O and benzene rings competing for the oxygen’s lone pair, making it less available for donation to the pi system. As a result, the acetoxy group is still o/p-directing (due to the existence of at least one lone pair), but not as powerfully activating as the hydroxy group (due to the presence of at least one lone pair).
Q4. What is the effect of temperature on Fries rearrangement?
Answer: It is important to note that the orientation of this electrophilic aromatic substitution in Fries rearrangement varies with temperature. Low reaction temperatures favours para substitution, while comparatively high temperatures favours ortho substitution.


