-
Call Now
1800-102-2727
Fractional Distillation– Definition, Principle, Apparatus, Procedure, Industrial Distillation, Fractional Distillation of Crude Oil
Before getting selected for a mountaineering expedition, a bunch of young enthusiasts were once made to go through a fitness test. They were told to climb a flight of stairs. The ones with the greatest of stamina naturally completed the test at the fastest pace and reached the zenith. In fact, the people were segregated as per their stamina into separate sections of the stairs. And when the timer was made to stop, each participant was found in different regions of the stair. This is so much like separating the bunch of people based on their physical stamina.

The scenario here is basically quite similar to that of the process of fractional distillation, which separates a mixture of liquids based on their volatilities. In fact, the process of fractional distillation is extremely integral in the processing of crude oil and is hence the major factor leading to the industrial advancement of our planet. Let’s explore more about this process.
Table of Contents
- Fractional Distillation– Definition
- Principle of Fractional Distillation
- Apparatus of Fractional Distillation
- Procedure of Fractional Distillation
- Industrial Distillation
- Fractional Distillation of Crude Oil
- Applications of Fractional Distillation
- Practice Problems
- Frequently Asked Questions–FAQs
Fractional Distillation– Definition
Miscible liquids are separated via a type of distillation called fractional distillation. Separating a mixture into its constituent parts, or fractions based on the smaller differences in their boiling points, is known as fractional distillation. By heating chemical mixtures to a certain temperature where one or more fractions of the mixture evaporate and hence chemical compounds are separated. Distillation is used to fractionate.
The component in the mixture that is more volatile separates as vapour first, followed by the next.
Under the pressure of one atmosphere, the component elements often have boiling points that are less than 25 °C (298 K) apart from one another. A straightforward distillation is normally utilised when the difference between the boiling points is higher than 25 °C.
The mixture is typically divided into component parts after a series of distillations and condensations. When the combination is heated to a specific temperature where some of the mixtures begins to evaporate, the separation takes place. If the boiling points of the liquids to be separated are closer to each other, then fractional distillation is carried out using the fractionating column.
Principle of Fractional Distillation
- Any liquid mixture's vapour composition typically fluctuates from its liquid composition. The liquid with the lower boiling point boils and turns into vapours when the mixture is heated.
- Compared to the liquid component, the more volatile component spends more time in the vapour state. The procedure involves several condensations and distillations, and the mixture is typically divided into its component parts.
- After heating, the more volatile components become vapour, and when this vapour is liquefied, the more volatile components become liquid.
- The process of vaporisation followed by condensation is referred to as distillation (liquefaction). Repeating this distillation procedure will cause a more volatile component to stay in the liquid state in its pure form. A pure substance can be isolated from the components of the liquid-liquid combination using the fractional distillation process.
- This form of distillation works on the fundamental principle that different liquids boil and evaporate at various temperatures. The component with the lower boiling point, therefore, begins to boil and turn into vapours sooner when the mixture is heated.
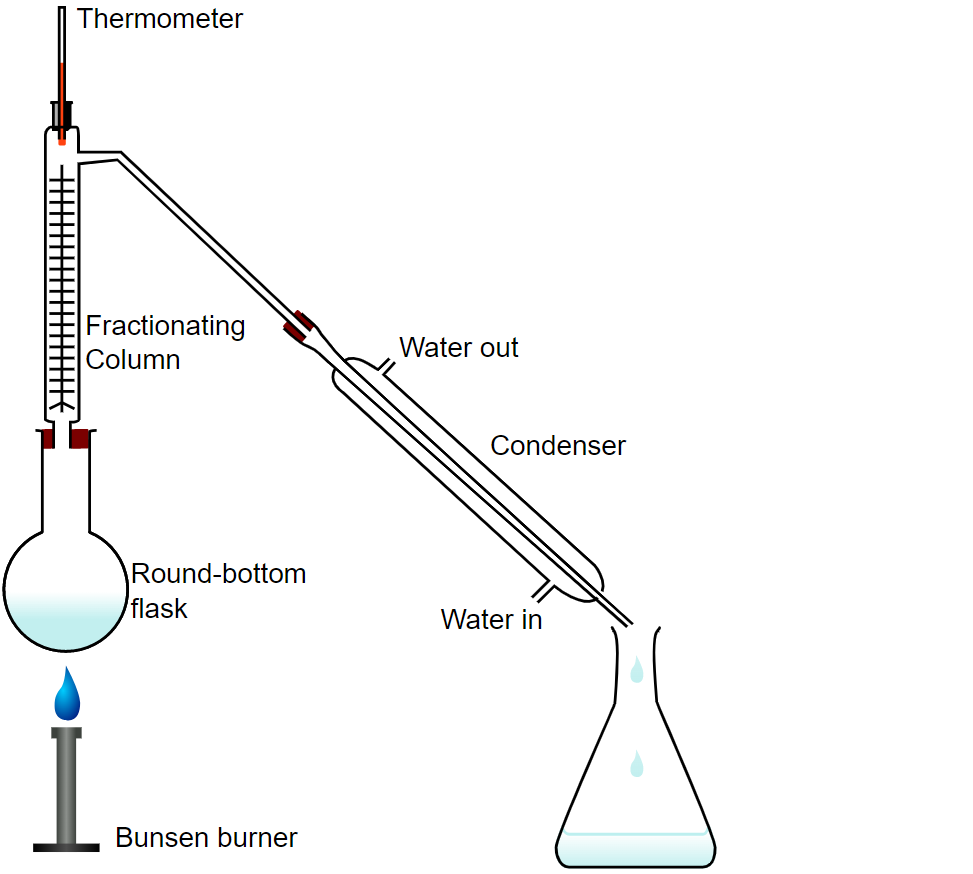
Apparatus of Fractional Distillation
A Bunsen burner, a round-bottomed flask, a condenser, a receiver, as well as a single-purpose fractionating column is generally used in the laboratory for doing fractional distillation. The following are the major apparatus:
- A source of heat (A bunsen burner along with a bath or a hot plate along with a bath)
- A round-bottom distilling flask
- A tripod stand to place a round-bottom distilling flask
- A receiving flask (usually a conical flask or round-bottom flask)
- A fractionating column
- A head used for distillation
- A condenser
- A thermometer
- Few boiling chips (alternatively known as anti-bumping granules)
The mixture is added to the flask with a circular bottom, along with some anti-bumping granules (or, if using magnetic stirring, a Teflon-coated magnetic stirrer bar), and the fractionating column is then screwed onto the top. The heat source for the fractional distillation column is located at the base of the still pot.
A temperature gradient forms in the column as the distance from the still pot grows; it is coldest at the top and warmest at the bottom. Some of the mixed vapour condenses and vaporises along the temperature gradient as it rises the gradient in temperature. The amount of the more volatile component in the vapour changes as it condenses and vaporises more frequently.
As a result, the vapour is finally reduced to just the more volatile component down the length of the column. The distillate is refluxed as the vapour condenses on the glass trays inside the column and drips back into the liquid below.
Procedure of Fractional Distillation
Fractional Distillation apparatus comes with a few equipment such as a fractionating column, condenser, receiver, distillation flask, thermometer, and heat source.
After assembling the device, a solution of two liquids A and B where A (ethanol) is more volatile than liquid B (water). The fractionating column is linked to the distillation flask's tip as the solution is introduced. When heat is supplied, the temperature rises gradually. The mixture then begins to boil, and the flask fills with rising vapours. The volatile component A (ethanol) is what is emitting the vapours. After entering the condenser, where it is cooled to create a liquid that is collected in the receiver, the vapours continue to move through the fractionating column.
Throughout the procedure, condensation and vaporisation occur frequently until the two mixtures are entirely split apart.



The mixture is added to a flask with a circular bottom. The column fills with vapour as the mixture boils. The vapour rises inside the column and falls back down into the liquid below where it condenses on the glass platforms, referred to as trays. The distillate is "refluxed" in this manner.
Only a small portion of the vapours—those that are the most volatile—remain in gaseous form at the top of the column. This fraction enters the condenser, where it is cooled until it liquefies and in the receiving flask is then collected as a liquid.
By insulating the outside of the column with an insulator like wool, aluminium foil, or (ideally) a vacuum jacket, the efficiency in terms of the amount of heating and time needed to achieve fractionation can be increased. The top tray is the coolest, and the bottom tray is the hottest. The vapour and liquid on each tray are in equilibrium under steady-state circumstances. With more trays added, the fractionation is more thorough (up to certain practical limitations).
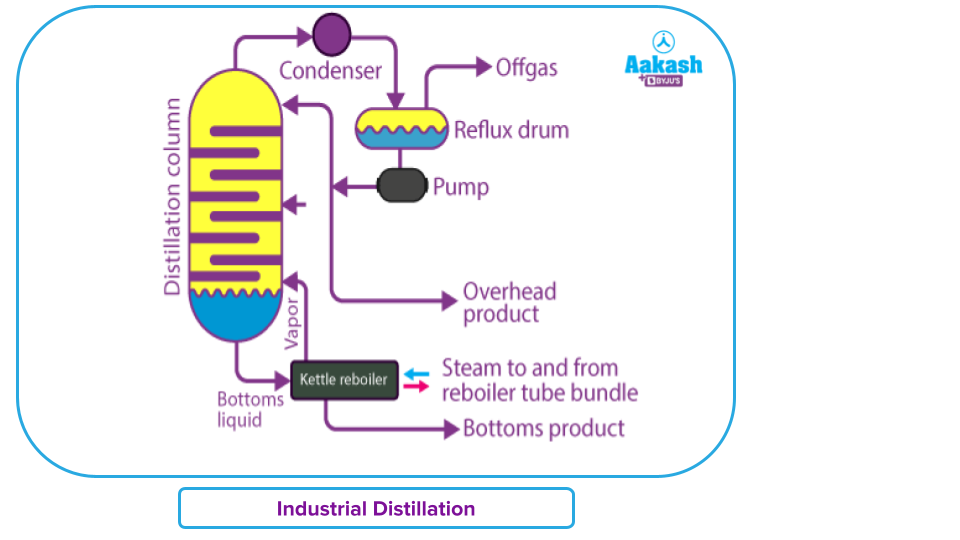
Industrial Distillation
One of the often utilised separation processes in a variety of sectors is fractional distillation. The distillation is done on a greater scale, but the basic idea behind the procedure stays the same. Typically, "distillation columns" or "distillation or fractionation towers"—both terms for enormous vertical cylindrical columns—are utilised. Reflux is used in these industrial towers to ensure that the mixes are completely separated.
The most often utilised separation technique in petrochemical and chemical plants, natural gas processing facilities, and cryogenic air separation facilities is fractional distillation.
Industrial distillation is often carried out in sizable, vertical cylindrical columns called "distillation or fractionation towers" or "distillation columns" that range in size from 0.65 to 6 metres in diameter to 6 to 60 metres or more in height. The liquid outlets in the distillation towers enable the removal of various fractions or products with various boiling point ranges.
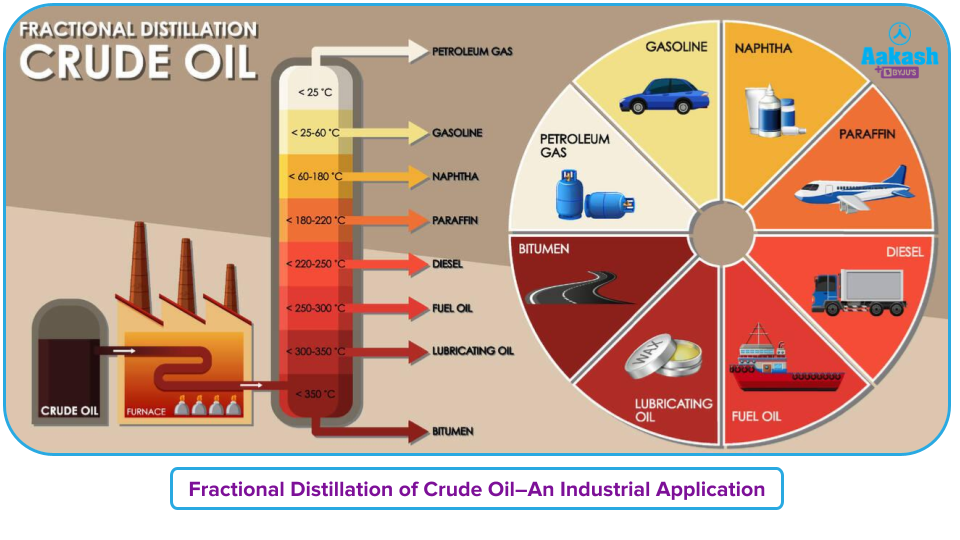
Fractional Distillation of Crude Oil
The process of separating the various components of crude oil is a typical example of fractional distillation in the industrial world. Typical components of crude oil include kerosene, paraffin wax, gasoline, diesel, naphtha, and lubricating oil. Effective separation of these components is made possible by the distillation process.
The chamber is filled with crude oil and heated using high-pressure steam. As the mixture begins to boil, the vapours are produced. Various compounds now transition into the vapour phase. In the fractional distillation column, which is made up of multiple plates, the vapour climbs to the top.
One of the more significant fractions is a light distillate, whose byproducts have boiling temperatures between 70 and 200°C. Petroleum gas, gasoline, naphtha (a chemical feedstock), kerosene, jet fuel, and paraffin are among the useful hydrocarbons in this spectrum. These substances flow readily, burn rapidly, contain tiny molecules, are highly volatile, and have low boiling temperatures.
The products known as medium distillates have boiling temperatures between 200 and 350 °C. Diesel fuel and gas oil, which are used in the production of town gas and for commercial heating, are products in this group.
The products with the lowest volatility have boiling points over 350°C, which are those of heavy distillate. These fractions may need to be heated to flow if they are solid or semi-solid. This percentage produces lubricating oil and bitumen.
Applications of Fractional Distillation
- Chlorosilanes can be converted into high-purity silicon by fractional distillation. Semiconductors make extensive use of silicon.
- It is employed to separate liquefied air. We get substances like oxygen, concentrated argon, and liquid nitrogen. Air is a mixture of several trace gases, such as water vapour and other noble gases, with a composition of roughly 78% nitrogen, 21% oxygen, 0.93% argon, and 0.378% carbon dioxide. The majority of these gases may be recovered straight from the air using liquefaction and fractional distillation since they have a variety of industrial and commercial uses.
- The separation and purification of diverse organic molecules is the principal use of fractional distillation in a variety of sectors, including chemical plants and oil refineries.
- The crude oil, also known as petroleum, that runs the engine in our car is located deep beneath the surface of the planet and the ocean floor. It is a naturally occurring, yellowish-black liquid made mostly of organic matter and hydrocarbon deposits. These components can be used in a variety of applications, but not when combined. By using the fractional distillation method, it is possible to separate the intricate mixture of hydrocarbons that makes up crude oil into fractions.
- It is helpful in the water purification process.
- The alcohol manufacturing industry utilises fractional distillation to a large extent. Fractional distillation is used to raise the liquid's alcohol content even further. Alcohol has a boiling temperature of 78.5 °C (173.3 °F), while water has a boiling point of 100 °C (312 °F). This is the basis for the principle of alcohol distillation. A higher alcohol concentration or strength will be present in the condensate when an ethyl alcohol-containing liquid is heated to a temperature that is above 78.5 °C but below 100 °C. A stronger liquor with up to 80% v/v alcohol can be produced using fractional distillation, which can further concentrate the alcohol.
- Acetone and water are separated by fractional distillation.
- Fractional distillation is also heavily utilised by the pharmaceutical business. Solvent swapping or exchange plays a crucial part in the production of active medicinal compounds. Through fractional distillation, it is accomplished. The original solvent mixture is combined with the swap solvent before being fed into the fractionating column.
- Fractional distillation is employed in pharmacy for a range of tasks, such as the separation of alkanes, the creation of alcoholic solutions fit for pharmaceutical application, and even the disintegration of cannabis to produce its oil and boost THC (tetrahydrocannabinol) levels.
- In the field of perfumery and the fragrance industry, the entire procedure is referred to as fragrance extraction and is done largely by fractional distillation in the process of separating aromatic chemicals from raw materials. Essential oils, absolutes, and other concretes are the end products of the extracts. From a variety of plants, including mint, clove, tea tree, and patchouli, raw essential oils are extracted.
- Fractional distillation is primarily used in the fragrance business to create highly pure isolates and aroma compounds from essential oils. Different distilled fractions from a material can be eliminated using a fractionating column in order to control the aroma of the finished product.
Recommended Video
https://www.youtube.com/watch?v=bw4nqBjsUao
Timestamp: 05:43 - 09:21
Practice problems
Q.1 Which of the following step is not involved in fractional distillation?
A. Vapourisation
B. Condensation
C. Collection
D. Extraction
Answer: (D)
Solution: Fraction distillation involves three major steps: vaporisation or evaporation where the mixture is heated until it evaporates and then vapours are collected in a fractionating column. This is followed by condensation of vapours separately based on volatility followed by the collection of these separate vapours. So, extraction is not involved. Hence, option D is the correct answer.
Q.2. The idea that different liquids boil and evaporate at certain temperatures is the principle behind:
A. Distillation
B. Sublimation
C. Solvent Extraction
D. Chromatography
Answer: (A)
Solution: Miscible liquids are separated via a type of distillation called fractional distillation. Separating a mixture into its constituent parts, or fractions based on the smaller differences in their boiling points is known as fractional distillation. So, option A is the correct answer.
Q.3. The separation of liquid occurs when the mixture to be separated is heated at a specific temperature where fractions of the mixture start to:
A. Boil
B. Solidify
C. Cool
D. Vapourise
Answer: (D)
Solution: Separating a mixture into its constituent parts, or fractions, is known as fractional distillation. By heating chemical mixtures to a temperature where one or more fractions of the mixture evaporate and then chemical compounds are separated. The component in the mixture that is more volatile separates as vapour first, followed by the next. Hence, the correct answer is option D.
Q.4. Explain how crude oil’s fractionation occurs.
Answer: Heat is applied to crude oil until it evaporates. At the bottom, a fractionating column receives crude oil vapour, which rises up the column. The bottom of the column has the warmest temperature. At the bottom, long-chain hydrocarbons condense and accumulate as liquids.
Hydrocarbons with short chains have lower boiling points. Nearer the top of the column, they pass through and condense at lower temperatures. They gather the fractions. They undergo further processing to produce final goods: Fuels (such as gasoline and diesel) are a typical final product.
Some fractions from the petrochemical sector can be utilised as feedstock (a component of a manufacturing process) to create solvents, lubricants, detergents, and other products.
Frequently Asked Questions–FAQs
Q.1. What is the difference between simple distillation and fractional distillation?
Answer: Fractional distillation is used for mixes containing chemicals with comparable boiling points, whereas simple distillation is used to separate components in mixtures with vastly different boiling points.
Simple distillation separates liquids having boiling point gaps of more than 50 degrees, whereas fractional distillation separates liquids with closer boiling points. This is the major distinction between fractional distillation and simple distillation. Moreover, in simple distillation, no fractionating column is used to separate the liquids.
Simple distillation, in contrast to fractional distillation, is only useful for relatively pure liquids, necessitates a significant difference in boiling points between components, and doesn't as thoroughly separate fractions.
Q.2. What are the advantages of fractional distillation?
Answer: Due to a large number of theoretical plates, fractional distillation is more effective than simple distillation in separating. It is a crucial procedure in the fields of industry, food science, and chemistry. Processes like desalination, crude oil refinement, and chemical purification are among the applications of fractional distillation.
To improve the separation of liquids from the mixture in a fractional distillation arrangement, a fractionating column is often positioned between the distilling flask and head. These columns have more theoretical plates because they have a bigger surface area where liquid-vapour equilibria can take place. Vigreux and glass bead columns, which have six to eight theoretical plates, are two types of fractionating columns.
Q.3. Which type of mixtures can be separated and purified using fractional distillation?
Answer: Miscible liquid mixes are separated by fractional distillation, especially whose components’ boiling points differ by less than 25°C. It is used to divide crude oil, or "petroleum," into usable fractions like kerosene, gasoline, or diesel, for example.
Q.4. What are the disadvantages of fractional distillation?
Answer:
These are the major disadvantages listed below:
- It is an expensive procedure. It is slow and complicated when done on a large scale, industrially. Needs very well-versed professionals to carry it out. Fractional distillation is costly on an industrial scale because it needs substantial structures, robust materials, and specialised equipment.
- There are operational risks with it. The explosion is one of the largest risks, which can happen if the system is not set up with the proper safeguards.
- Pollution of the environment may result from it. The ecological effects of fractional distillation depend on the kinds of mixes that are distilled; fractional distillation itself is not environmentally detrimental.
- For instance, the refining of crude oil may result in the release of significant quantities of hazardous chemicals into the environment. When refineries incorrectly dump waste, it may also contribute to the contamination of rivers, streams, and other bodies of water.