-
Call Now
1800-102-2727
General Features of f-block Elements - Introduction, Position in the Periodic Table, Practice problems & FAQs
Have you heard about nuclear weapons in the news? In your history book, you must have studied the nuclear bomb attack on Hiroshima and Nagasaki during world war II. The United States of America dropped an atomic bomb on Hiroshima and Nagasaki, Japan. The city was utterly destroyed when the nuclear bomb exploded above it.
Has this ever come to your mind, what type of chemicals do nuclear weapons consist of?
The answer is Plutonium, a highly reactive element, was used to make the nuclear bombs that were dropped on Hiroshima and Nagasaki. Because these elements are dangerous, governments regulate their manufacture and use.
Let's know more about elements like Plutonium, these elements are called inner transition elements.
While reading history, you must be surprised how one can remember the old dates. Whenever you went to a museum you must have seen things around a thousand of years ago. Do you find these interesting?
Events, discoveries, and innovations are a part of human history. To make it easier to remember, we label events with the dates they occurred and how it is possible for historians to find dates related to pasts. Inner transition metals are used by scientists and archaeologists to assess the age of fossils and other materials such as rocks.

Let’s begin with the study of some interesting facts about inner transition metals. These metals are remarkable because they were among the latest to be identified in the periodic table.
TABLE OF CONTENT
- What are f-block elements?
- Position of f-block in the periodic table
- General features of f-block
- Practice Problems
- Frequently Asked Questions-FAQ
What are f-block elements?
f-block elements are those in which an additional electron enters the (n-2)f orbitals. The electronic configuration of these elements' valence shells is Because the extra electron travel to f-orbitals in the (n-2)th main shell, these are known as f-block elements.
Lanthanides, or rare-earth, are elements in the 4f-block. 5f-block elements are also known as actinides or actinones. Because of their strong resemblance to Lanthanum and Actinium, the names Lanthanides and Actinides were given. The first inner transition series is lanthanides, while the second inner transition series is actinides.
The lanthanides and actinides are frequently represented as a distinct tiny "island" beneath the main body of the periodic table in order to condense it.
Position of f- block elements in the periodic table:
The filling up of the 4f and 5f orbitals corresponds to two series in the f-block. The elements are in the 4f and 5f series of Ce to Lu and Th to Lw, respectively. Each series has 14 elements that occupy the 'f' orbital.
General features of f-block:
For Lanthanides:
From lanthanum to lutetium, the lanthanide series of chemical elements consist of fifteen metallic chemical elements with atomic numbers 57 to 71. The rare earth elements are made up of these fifteen lanthanide elements, as well as the chemically related elements scandium and yttrium.
It should be noted that the atoms of these elements have a common electronic structure of 6s2 but varied occupancy of the 4f level. All tri-positive ions (the most stable oxidation state of all lanthanides) have electronic structures of the form 4fn (n = 1 to 14 with increasing atomic number).
Let us cover some important trends of f-block elements in the periodic table:
Electronic configuration:
For lanthanide starting with cerium and finishing with lutetium (Z = 71), the electronic configuration of the inner transition elements is .
Example: Electronic configuration of Lanthanoids are mentioned below:
|
Atomic Number |
Element |
Symbol |
Configuration |
|
57 |
Lanthanum |
La |
|
|
58 |
Cerium |
Ce |
|
|
59 |
Praseodymium |
Pr |
|
|
60 |
Neodymium |
Nd |
|
|
61 |
Promethium |
Pm |
|
|
62 |
Samarium |
Sm |
|
|
63 |
Europium |
Eu |
|
|
64 |
Gadolinium |
Gd |
|
|
65 |
Terbium |
Tb |
|
|
66 |
Dysprosium |
Dy |
|
|
67 |
Holmium |
Ho |
|
|
68 |
Erbium |
Er |
|
|
69 |
Thulium |
Tm |
|
|
70 |
Ytterbium |
Yb |
|
|
71 |
Lutetium |
Lu |
Atomic size and Ionic size:
- The additional electron reaches the 4f-subshell in lanthanides but not the valence-shell or sixth shell.
- Because the shape of the f-sub shell is very diffused, there is no similar rise in the mutual shielding effect of 4f-electrons, the shielding effect of one electron in the same subshell by another in the same subshell is very modest, even smaller than that of d-electrons.
- The outermost shell electrons in the nucleus suffer more significant nuclear attraction as a result of this. As a result, as we progress from La (57) to Lu (71), the atomic and ionic radii continue to decrease.
Below is the graph which shows the atomic radii of Lanthanides.
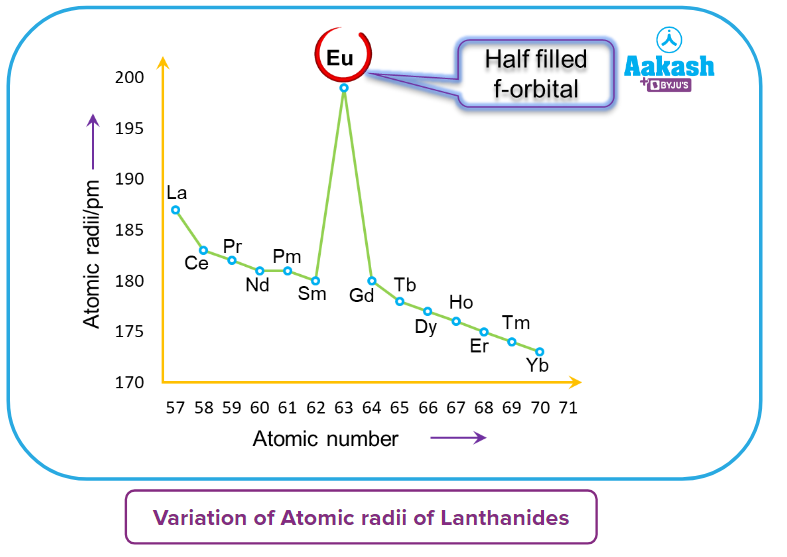
- The lanthanoid contraction (a decrease in atomic and ionic radii from lanthanum to lutetium) is a distinctive aspect of the lanthanide's chemistry.
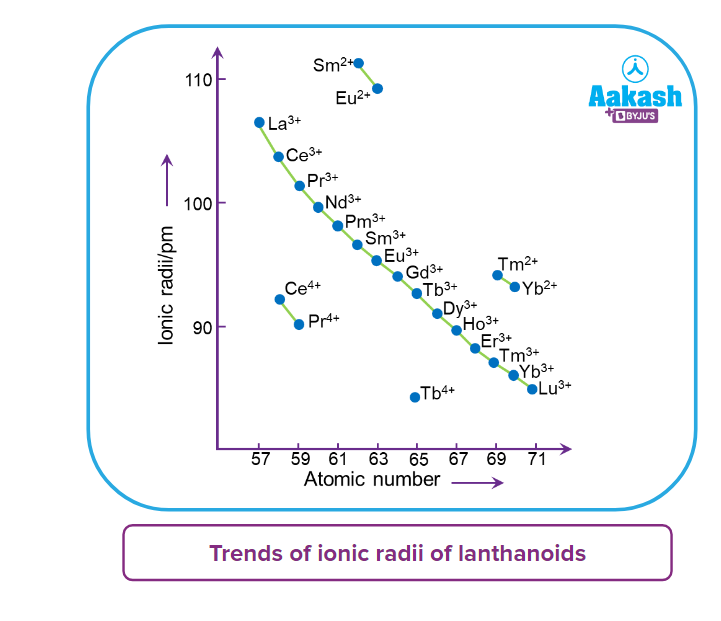
- It has far-reaching implications in the chemistry of the elements' third transition series. The drop in atomic radii (derived from metal structures) is not as consistent as it is in M+3 ions.
- Of course, this contraction is comparable to that seen in a regular transition series and is due to the same cause i.e; improper shielding of one electron by another in the same subshell. With the increase in nuclear charge down the series, however, the shielding of one 4f electron by another is smaller than that of one d electron by another. With rising atomic numbers, the diameters shrink in a pretty regular pattern.
- The radii of the members of the third transition series are remarkably similar to those of the corresponding members of the second series due to the cumulative effect of the lanthanoid series contraction, known as lanthanoid contraction.
- The nearly identical radii of Zr (160 pm) and Hf (159 pm), which result from the lanthanoid contraction, account for their coexistence in nature and the difficulties in separating them.
Oxidation state:
- The +II and +III chemicals are the most common lanthanide. However, +2 and +4 ions are occasionally found in solution or in solid compounds.
- The increased stability of empty, half-filled, or filled f subshells causes this irregularity in ionization enthalpies. The noble gas structure favors the creation of CeIV, although it is a strong oxidant that reverts to the common +3 state. Ce+4/ Ce+3 has a E0 value of + 1.74 V, indicating that it may oxidize water. However, because the reaction rate is slow, Ce(IV) is an excellent analytical reagent.
- Pr, Nd, Tb, and Dy have a +4 state as well, but only in oxides, MO2. Eu+2 is created by losing two s electrons, and its f7 configuration is responsible for its production. Yb+2, which has the f14 configuration, is also a reductant. Tb+4 is an oxidant with half-filled f-orbitals. Samarium behaves similarly to europium in that it has both +2 and +3 oxidation states.
Coloured ions:
Many lanthanide ions are coloured both in solid form and in solutions. Partially filled f-orbitals allow for f-f transitions, hence the color. Colorless M3+ ions with 4f0, 4f7, or 4f14configurations. La3+,Gd3+,Lu3+ are colorless in nature.
The color of pairs of M3+ ions with the same amount of unpaired electrons in 4f-orbitals are the same.
Colors of Lanthanides ions are mentioned in the below table:
|
M3+(M = lanthanide ion) |
Number of 4f electrons |
Color |
|
La3+ |
0 |
colorless |
|
Ce3+ |
1 |
colorless |
|
Pr3+ |
2 |
Green |
|
Nd3+ |
3 |
Lilac |
|
Pm3+ |
4 |
Pink |
|
Sm3+ |
5 |
Yellow |
|
Eu3+ |
6 |
Pale pink |
|
Gd3+ |
7 |
Colorless |
|
Tb3+ |
8 |
Pale pink |
|
Dy3+ |
9 |
Yellow |
|
Ho3+ |
10 |
Pale yellow |
|
Er3+ |
11 |
Pink |
|
Tm3+ |
12 |
Pale pink |
|
Yb3+ |
13 |
Colorless |
|
Lu3+ |
14 |
Colorless |
Ionization energies:
Ionization energies for lanthanides are generally low. The levels of alkaline earth metals, notably calcium, and the values of IE1 and IE2 are extremely comparable. Each element's total first three ionization energies in kJ mol-1 are listed below. Low values are present.
|
Element |
Ionization energy |
|
Ce |
3512 kJ mol-1 |
|
Pr |
3623 kJ mol-1 |
|
Nd |
3705kJ mol-1 |
|
Pm |
– |
|
Sm |
3898 kJ mol-1 |
|
Eu |
4033 kJ mol-1 |
|
Gd |
3744 kJ mol-1 |
|
Tb |
3792 kJ mol-1 |
|
Dy |
3898 kJ mol-1 |
|
Ho |
3937 kJ mol-1 |
|
Er |
3908 kJ mol-1 |
|
Tm |
4038 kJ mol-1 |
|
Yb |
4197 kJ mol-1 |
|
Lu |
3898 kJ mol-1 |
Lanthanides are extremely electropositive elements in nature because of their low ionization energy values. Hydrogen is produced when these elements react with both cold and hot water. However, with cold water, the reactions are gradual whereas, with hot water, they are quick. Thus, all lanthanides are potent reducing agents. From La to Lu, the lowering power gets weaker.
Magnetic properties:
Other than the f0 type (La+3 and Ce+4) and the f14 type (Yb+2 and Lu+3), all lanthanide ions are paramagnetic.
Complex formation:
Due to their huge size and low charge density, lanthanides do not have a strong tendency to form complexes. However, as the atomic number rises, the tendency to form complexes and their stability increases
For Actinides:
Electrons obtained by sequentially filling 5f orbitals are known as actinides or actinides. They get their name from the fact that they are the next element after actinium (Ac) in the periodic table. The actinide series is also known as the second inner transition series, as it contains 14 elements spanning from Th(90) to Lw(103). Despite the fact that actinium (Z=89) contains no 5f electrons, actinides are commonly used to study it.
Let's look at some key tendencies in the periodic table:
Electronic configuration:
The electrical configuration of 7s2 is thought to be shared by all actinides, with varied occupancy of the 5f and 6d subshells. The fourteen electrons are formally added to 5f, though not in thorium (Z = 90) but from Pa onwards the 5f orbitals are completed at element 103. The stabilities of the f0, f7, and f14 occupancies of the 5f orbitals are related to the abnormalities in the electronic configurations of actinides, as they were also in lanthanide. Am and Cm configuration are [Rn]5f77s2and [Rn]5f76d17s2 respectively. Although the 5f orbitals are similar to the 4f orbitals in terms of their angular component of the wave function, they are not as buried, and so 5f electrons can engage in bonding to a far higher amount.
Electronic configurations of actinides are given below:
|
Atomic Number |
Element |
Symbol |
Configuration |
|
89 |
Actinium |
Ac |
|
|
90 |
Thorium |
Th |
|
|
91 |
Protactinium |
Pa |
|
|
92 |
Uranium |
U |
|
|
93 |
Neptunium |
Np |
|
|
94 |
Plutonium |
Pu |
|
|
95 |
Americium |
Am |
|
|
96 |
Curium |
Cm |
|
|
97 |
Berkelium |
Bk |
|
|
98 |
Californium |
Cf |
|
|
99 |
Einsteinium |
Es |
|
|
100 |
Fermium |
Fm |
|
|
101 |
Mendelevium |
Md |
|
|
102 |
Nobelium |
No |
|
|
103 |
Lawrencium |
Lr |
Atomic size and Ionic size:
The actinides, like the lanthanides, have a similar general tendency.
The size of atoms or M3+ ions diminishes gradually throughout the run. Actinoid contraction is the term for this (similar to lanthanoid contraction). The shrinkage is higher from element to element in this series due to inadequate shielding by 5f electrons.
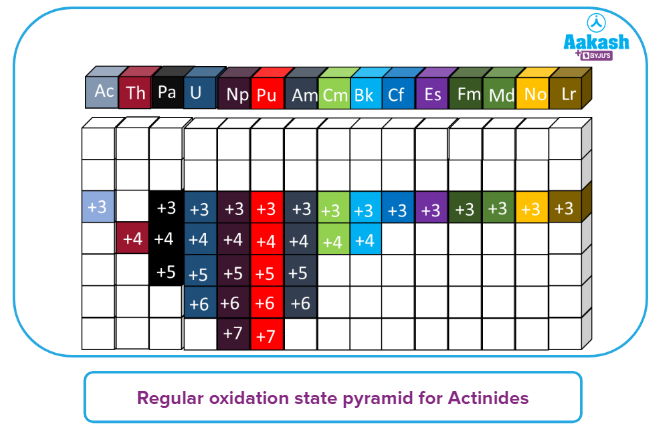
Oxidation states of Actinides:
- The actinides, like the lanthanides, have the most frequent oxidation state of +3. However, unlike the first four elements ( Th, Pa, U, Np) this state is not always the most stable.
- In the gas phase and in solution, for example, U3+ is easily oxidized. For the later elements , the +3 state is the most stable (except No).
- Th(+4), Pa(+5), and U(+6) are the most stable oxidation states for the first four elements. All of the outer electrons, including the f- electrons are used for bonding in these high oxidation states.
- Np has an oxidation state of +7, however, it is oxidizing, and its most stable state is +5.
- Pu shows all oxidation states from +3 to +7, with +4 being the most stable.
- Am represents oxidation states ranging from +2 to +6. The configuration of the Am2+ is f7. It's the equivalent of Eu2+ However, it only exists as fluoride in solid form. However, the +3 state is the most stable for Am and practically all of the remaining elements.
- From Th to Bk, all the elements have a +4 oxidation state.
- Ions of Cf2+, Es2+, Fm2+, Md2+and No2+ are present in solution. Their characteristics resemble those of alkaline earth metals, especially Ba2+.
- In No2+ it resembles an f14 arrangement and is No's most stable oxidation state.
Color of the ions :
- The majority of actinide ions are coloured.
- The quantity of electrons in 5f-orbitals affects the color of the ions. The ions with zero 5f-orbital electrons (i.e., 5f0) or seven 5f-orbital electrons (i.e., 5f7 ) are colorless.
- Both in a crystalline structure and in an aqueous solution, ions with 2 to 6 electrons in 5f-orbitals exhibit color. The f-f transition is what gives the color.
The coloured ions in different charges are shown in the below table:
|
Ions |
Inner configuration |
color |
|
Th4+ |
5f0 |
Colorless |
|
U3+ |
5f3 |
Red |
|
Np3+ |
5f4 |
Purple |
|
Pu3+ |
5f5 |
Violet |
|
Am3+ |
5f6 |
Pink |
|
Cm3+ |
5f7 |
Colorless |
|
U4+ |
5f2 |
Green |
|
Np4+ |
5f3 |
Yellow green |
Actinide contraction:
- From Thorium (Th) to Lawrencium (Lr), tri-positive actinides' atomic sizes are seen to steadily shrink. The electrons entering the inner orbital (n-2)f and the rising nuclear charge are the causes of this. Actinide contraction describes the shrinkage of an actinides' element caused by an increase in atomic number.
Magnetic behavior:
- The majority of the actinide ions are paramagnetic because they have unpaired electrons.Th4+(5f0), Pa4+(5f1), U3+(5f3), Np5+(5f2), Pu4+(5f4), Am5+(5f4) etc. are showing paramagnetic character.
- Actinide cations that only have paired electrons are diamagnetic.A few examples are Th4+(5f0), U6+(5f0), Lr3+(5f14) .
Formation of complexes:
- Compared to lanthanides, actinides have a slightly higher propensity to form complex compounds. This is brought on by their smaller ions and higher charge.
- The majority of the actinide halides combine with alkali metal halides to generate complicated combinations.
- Actinides combine with organic substances like oxine and EDTA to generate chelates.
- For the ions M4+, , M3+ and, the degree of complex formation declines in order.
Radioactivity:
- The radioactivity of all actinide elements is inherent.
- Actinides are heavy elements that contribute to the radioactivity of spent fuel in addition to the fission products present in nuclear reactor cores. Because many actinides are radioactive and/or unstable, they need to be handled carefully. The chemistry and arrangement of the particles in crystals are significantly influenced by the radiation in actinides.
- They are able to experience nuclear reactions.
- The most prevalent isotope of the actinide, which is by far the most frequently generated, is plutonium (239). 'Minor' actinides like neptunium (238) and americium 241 and 243, as well as curium 244 and 245, are also produced in lower amounts by nuclear reactors.
Related video link: https://www.youtube.com/watch?v=EAQGKdam8sE (40-37 to 1:18:25)
D and F Block Elements Class 12 Chemistry One Shot & Mind Maps L- 2 (Ep 29) | NEET 2022 Exam Prep
Practice Problems:
Q1. The electronic configuration of cerium in the +3 oxidation state is:
Answer: C
Solution: The electronic configuration of cerium is . Cerium in the +3 oxidation state loses +3 electrons and thus gets a configuration of .
So, Option C is the correct answer.
Q2. ___________salts are used in glass industries for imparting colors.
A. Uranium
B. Plutonium
C. Americium
D. Californium
Answer: A
Solution: Uranium salts are used in the glass industry (to impart a green color), the textile business, the ceramic industry, and medicines.
Q3. Which actinide oxidation state is the most stable?
A. +2
B. +3
C. +4
D. +5
Answer: B
Solution: Because of the smaller energy difference between the 5f, 6d, and 7s orbitals, actinoids exhibit varying oxidation states. Other oxidation states are possible despite the fact that 3+ is the most stable oxidation state due to the efficient shielding of f-electrons.
Q4. What type of plutonium is contained in nuclear weapons?
A. Pu-238
B. Pu-239
C. Pu-240
D. Pu-241
Answer: B
Solution: The most frequent form of plutonium in a normal nuclear reactor is fissile Pu-239, which is created when a neutron is captured from U-238 and then undergoes beta decay. Fissioning Pu-239 produces roughly the same amount of energy as fissioning U-235.
Frequently asked questions-FAQs:
Q1. How stable are f-block components?
Answer: The deep-seated 4f and 5f orbitals of the f-block chemical elements (lanthanum and actinium) have been filled with electrons as the atomic number has increased. All f-block elements have a stable or common oxidation state, which is their trivalent oxidation number or state.
Q2. What are the major lanthanides' sources?
Answer: The main source of lanthanides is monazite sand, which is made up of phosphates of lanthanum, thorium, cerium, and neodymium. The phosphate portion of monazite also contains trace amounts of other lanthanide ions. Promethium is the only lanthanide that is produced artificially through a nuclear reaction.
Q3. Can inner transition elements be used for medical purposes?
Answer: These metals are utilized for medical purposes in addition to powering nuclear power reactors. Uranium, for example, is employed as a barrier against radiation.
Q4. What is the difference between transition elements and inner transition elements?
Answer: The transition elements are in the d-block, and in the d-orbital have valence electrons. They can form several states of oxidation and contain different ions. They are known as transition elements because of the d-d transition. Inner transition elements are in the f-block, and in the f-orbital have valence electrons.


