-
Call Now
1800-102-2727
Extraction of Mg and Cu – Occurrence, Position in the Periodic Table, and Methods of Extraction of Magnesium and Copper
Everyone enjoys the milk-based treats and sweets that our mothers or grandmothers make, right?
We are aware that dairy products have a number of health advantages and that the majority of milk products are produced by cattle. Have you ever wondered how raw milk is converted into milk products? For instance, raw milk is used to make butter by skimming the cream off the top and allowing the milk to sit for about 12 hours to allow the cream to develop. After that, a sieve is used to separate the solid butter from the bulk. Isn't it remarkable how extraordinary goods like butter can be produced from relatively commonplace liquids like milk?
Similarly, various unprocessed minerals known as ores are used to extract pure metals. There are very few metals in nature that are found in their pure, unaltered state, including noble metals like gold, silver, and platinum. The remaining metals, which are crucial to human life, are found in combination with ores or minerals. A variety of small impurities can be found in ores. There are several ways to remove metals from their ores.
We'll learn about the extraction of metals in this article, particularly copper and magnesium.
TABLE OF CONTENTS
- What is Extraction?
- Occurrence and Position of Magnesium
- Extraction of Magnesium
- Occurrence and Position of Copper
- Extraction of Copper
- Practice Problems
- Frequently Asked Questions – FAQ
What is Extraction?
The earth's crust contains variable amounts of metal ores. We can utilise the minerals in the earth because metals can be extracted from ores. Mining is the process of extracting metal ores from subterranean deposits. Metallurgy refers to the entire process of extracting pure metal from one of its ores, on a large scale while maintaining economic viability. Several physical and chemical processes are employed to extract metals from ores. The process used in a particular situation is determined by the ore's nature, the metal's characteristics, and the local conditions. As a result, a universal approach for extracting all metals from their ores is not possible. A metal's metallurgy consists of five primary operations.
- Concentration or dressing of the ore.
- Calcination and roasting are used to transform concentrated ore into a form that is appropriate for reduction, often an oxide, depending on the type and chemical makeup of the ore.
- Metal production through reduction.
- Metal purification and refinement.
- Pollution-control procedure for handling waste gases and materials.
Occurrence and Position of Magnesium
Magnesium does not occur naturally in its pure form. Humphry Davy, a British chemist, isolated pure elemental magnesium in 1808 by electrolysing a mixture of magnesium oxide and mercuric oxide. Magnesium in the combined form is found in minerals such as Magnesite (MgCO3), Dolomite (MgCO3.CaCO3), Kieserite (MgSO4.H2O), Epsom salt (MgSO4.7H2O), Carnallite (KCl.MgCl2.6H2O), and Asbestos [CaMg3(SiO3)4]. After Cl- and Na+, magnesium ion (Mg2+) is the third most prevalent dissolved ion in the oceans. As a result, the oceans offer a rich source of magnesium ions, from which the metal can be easily extracted. Moreover, it is a fundamental component of chlorophyll, the green pigment in plants.

The periodic table's group IIA has six elements: beryllium, magnesium, calcium, strontium, barium, and radium. Because their oxides are basic (alkaline) in nature and are found in the earth's crust, these elements are together known as alkaline earth metals.
Extraction of Magnesium
Utilising the distinct physical and mechanical characteristics of the element and its alloys, magnesium metal is employed in a wide range of industries and applications. Due to its lightweight characteristics, which help to reduce weight, improve fuel efficiency, and lower greenhouse gas emissions, magnesium is employed more and more every year. Let’s discuss a few methods of extraction of magnesium.
From Magnesite
Magnesite (MgCO3) is calcinated to produce magnesium oxide (magnesia). Magnesium is mostly obtained from the mineral magnesite, which is a form of magnesium carbonate (MgCO3) and a member of the calcite group of carbonate minerals. The mineral was created as a byproduct of the alteration of rocks high in magnesium or as a result of the action of magnesium-containing fluids on calcite.

Either electrolysis or thermal reduction of magnesium oxide can be used to produce magnesium. Molten magnesium, barium, and sodium fluorides are combined with the oxide to form the solution. Utilising cast iron rod as cathode and carbon rod suspended in the molten mixture as the anode, electrolysis of the mixture is carried out at 650°C. Magnesium is produced during electrolysis in a molten condition. Moreover, reducing agents such as carbon, silicon, aluminium, or calcium carbide can be used to thermally reduce magnesium oxide.
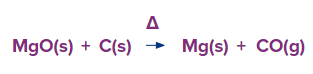
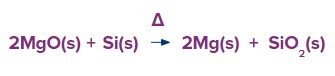
Electrolysis of Anhydrous Magnesium Chloride
The process of isolation of magnesium by the electrolysis of anhydrous magnesium chloride consists of the following three steps.
- Preparation of hydrated MgCl2
- Conversion of hydrated MgCl2 into anhydrous MgCl2
- Electrolytic reduction of anhydrous MgCl2
Let’s discuss each step in detail.
- Preparation of hydrated MgCl2
The preparation of hydrated MgCl2 can be done by two different methods.
- Preparation of hydrated MgCl2 from carnalite (KCl.MgCl2.6H2O)
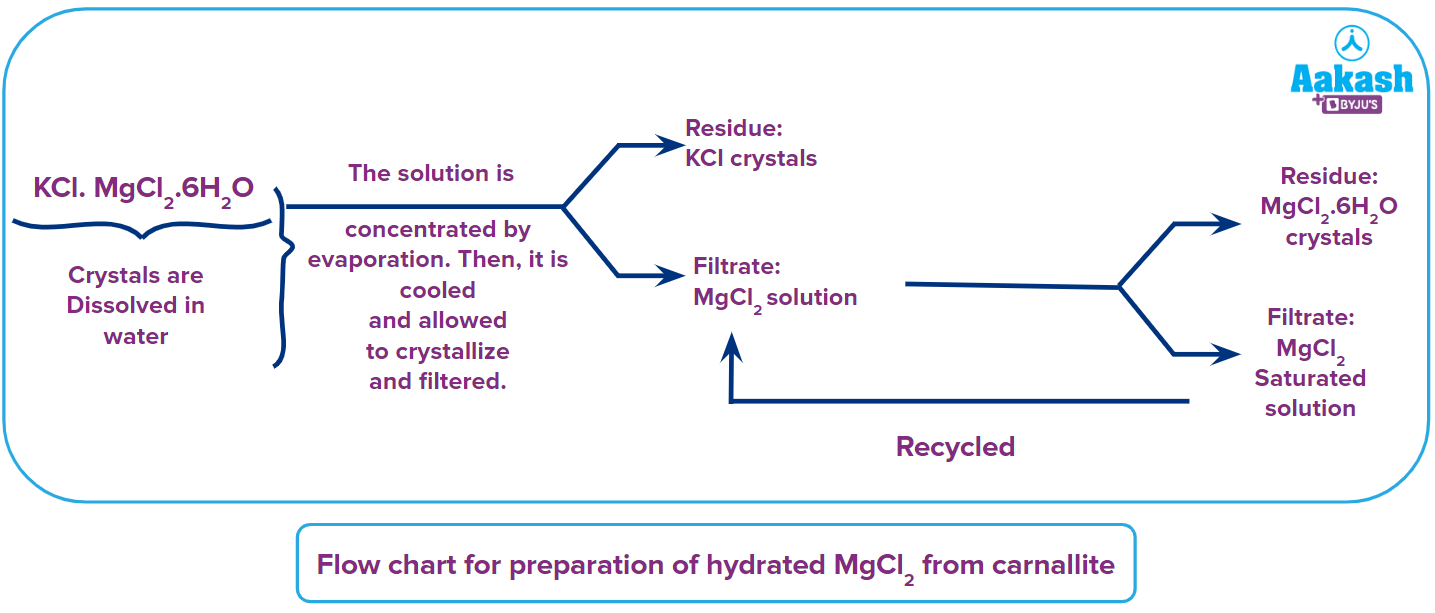
The principle behind this whole process of preparation of hydrated MgCl2 is that MgCl2 is more soluble than KCl and KCl crystallises first.
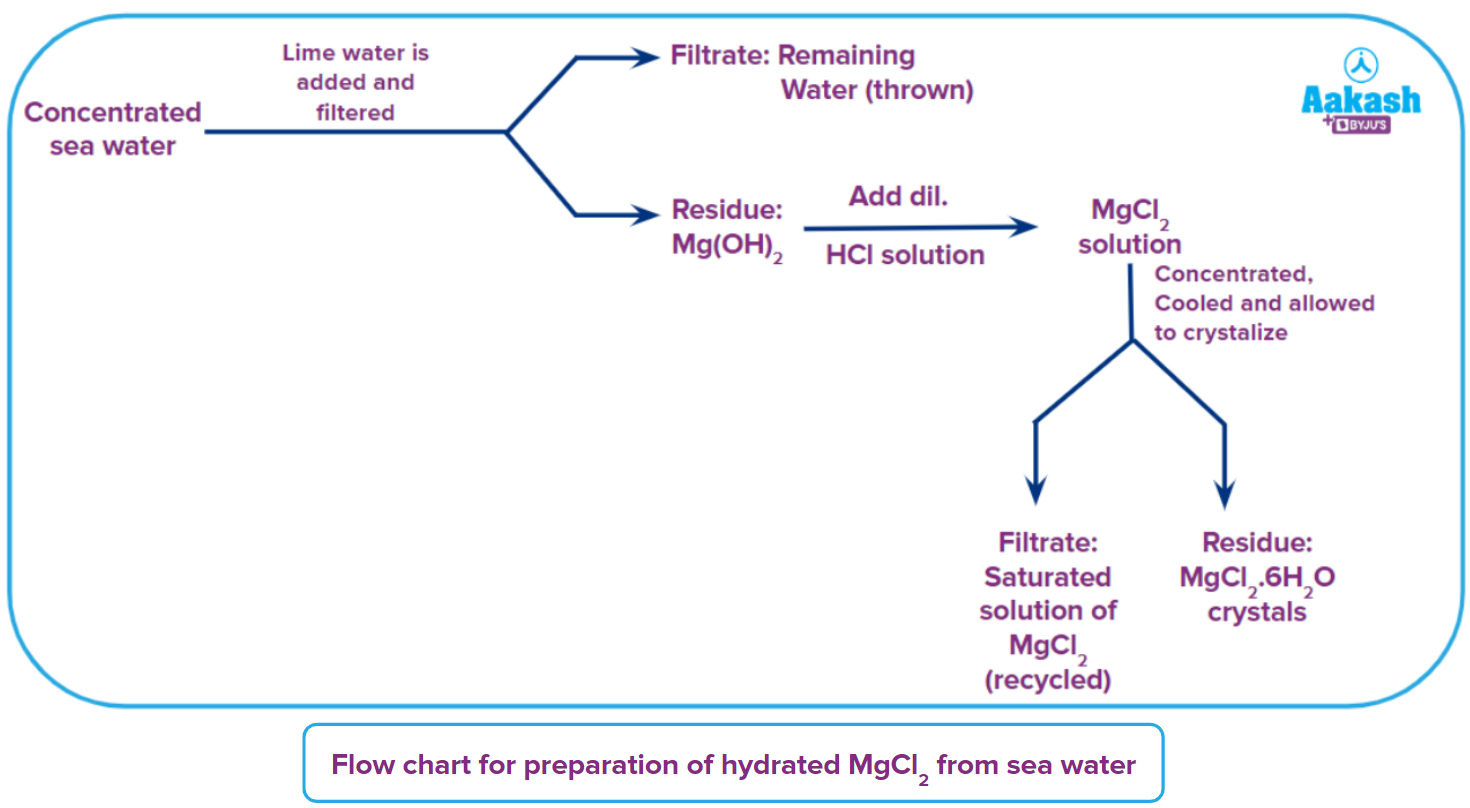
- Preparation of hydrated MgCl2 from sea water
Magnesium compounds can be found in seawater. Magnesium hydroxide precipitates when magnesium compounds are treated with calcium hydroxide. This process of extraction of MgCl2 from seawater is known as the Dow seawater process. The principle here is that Ca(OH)2 is more soluble than Mg(OH)2 and it precipitates.
- Conversion of hydrated MgCl2 into anhydrous MgCl2
We cannot obtain anhydrous MgCl2 on direct heating of hydrated MgCl2 (MgCl2.6H2O) as it gets hydrolysed.

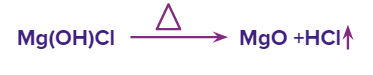
Moreover, this MgO which is formed is unwanted in the electrolytic reduction process as its melting point is very high. That is the reason why hydrated MgCl2 is heated to 175 ℃ in the current of dry HCl in vacuum and anhydrous MgCl2 is formed.
![]()
With the dry HCl gas present in the system, the equilibrium shifts towards the left side and with the heating the hydrated MgCl2 continues to decompose, which results in anhydrous MgCl2. MgO or seawater can also be used to make anhydrous magnesium chloride. Chlorine is sprayed on a combination of red-hot MgO and carbon.
- Electrolytic reduction of anhydrous magnesium chloride
The magnesium chloride is dried first. Anhydrous magnesium chloride is fused with anhydrous calcium chloride and sodium chloride in a ratio of 35% (MgCl2), 50 % (NaCl), and 15 % (CaCl2). In an electrolytic cell, the mixture is electrolyzed at 700°C in the presence of an inert atmosphere.
Reactions at the electrodes:
Reaction as anode:
Reaction at cathode:
Magnesium and chlorine are produced at the cathode and anode, respectively.
Magnesium, which is lighter than the fused mass, floats to the surface and is regularly extracted by perforated ladles. The addition of NaCl and CaCl2 to anhydrous MgCl2 lowers the fusing temperature and improves the electrical conductivity of the fused mass.
Occurrence and Position of Copper
Copper has been used by humans from prehistoric times. The word 'cuprum' comes from the island of Cyprus, where the metal was collected by the Romans and Greeks. Bronze is an alloy of copper and tin, consisting of around 88% copper and 12 % tin, and it was used in Egypt as early as 350 B.C. Copper is found in a tiny fraction of the earth's crust (0.0001%). Its deposits are few. Copper can be found in the following forms in nature.
- Native state: Copper is abundant in the metallic state in Lake Superior in the United States, as well as in the Ural Mountains in Russia and Sweden.
- Combined state: Sulphides, oxides, and basic carbonates are the most common copper ores. Being ductile (able to be formed into wire), malleable (able to be hammered and moulded), and a good conductor of electricity, copper (Cu) is a very important industrial metal (only silver is better). Some of the minerals in which copper can be found are Numerous minerals that exist in deposits big enough to mine contain copper. Azurite 2CuCO3·Cu(OH)2, culprit (Cu2O), malachite, chalcocite (Cu2S), acanthite, chalcopyrite, and bornite. Chalcopyrite (CuFeS2) is where most copper is found (around 79.8 % copper at pure form).
Position of Copper in the Periodic Table
Copper is found in group 11 of the periodic table.The differentiating electron is accommodated in the (n-1)d energy shell, making these d-block elements. Copper belongs to the 3d series of metals.
Extraction of Copper
Copper can be extracted in a variety of ways, depending on the type of ore and the amount of copper present. There are two types of methods that can be used.
- From Sulphide Ores:
- The dry process, often known as smelting, is used on ores with a copper concentration of more than 3 %.
- The wet process, also known as the hydrometallurgical process, is used on low-grade ores that contain a minor amount of copper.
- From Non-Sulphide Ores: Copper is collected primarily from copper pyrites using a dry method or smelting technique. The steps involved in extracting copper from copper pyrites are as follows.
- Concentration of ore: The froth flotation method is used to concentrate sulphide ore. The ores are pulverised and sieved before being dumped into water tanks containing pine oil and potassium xanthate. A powerful rush of air is released, agitating the entire mass. The froth flotation method is used to concentrate sulphide ore. The ores are pulverised and sieved before being dumped into water tanks containing pine oil and potassium xanthate. A powerful rush of air is released, agitating the entire mass.
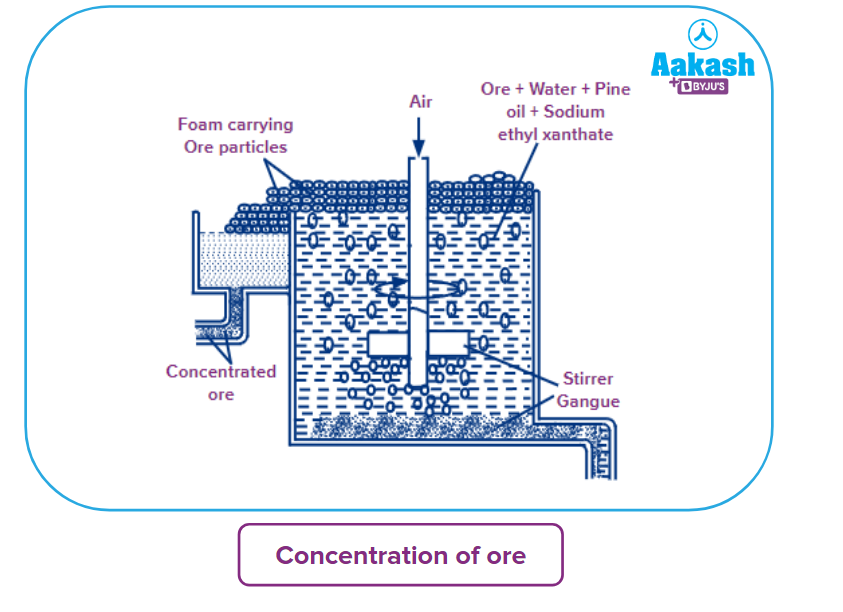
The ore particles are carried to the surface of the liquid by froth, whilst the contaminants drop to the bottom of the tank. The froth is separated on a regular basis. This is the ore that has been concentrated.
- Roasting: The concentrated ore is rapidly heated in a stream of air on the reverberatory furnace's hearth. The following changes occur during roasting.
- Sulphur dioxide is produced by oxidising free sulphur.
- Arsenic and antimony present in the ore are extracted as volatile oxides.
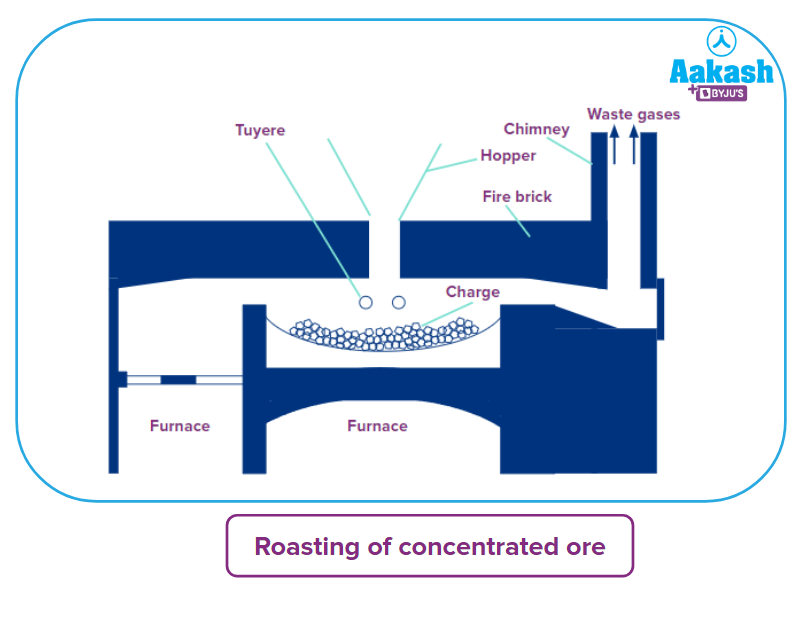
- With the evolution of sulphur dioxide, the pyrite is transformed into cuprous sulphide and ferrous sulphide.
- Copper and iron sulphides are partially oxidised.
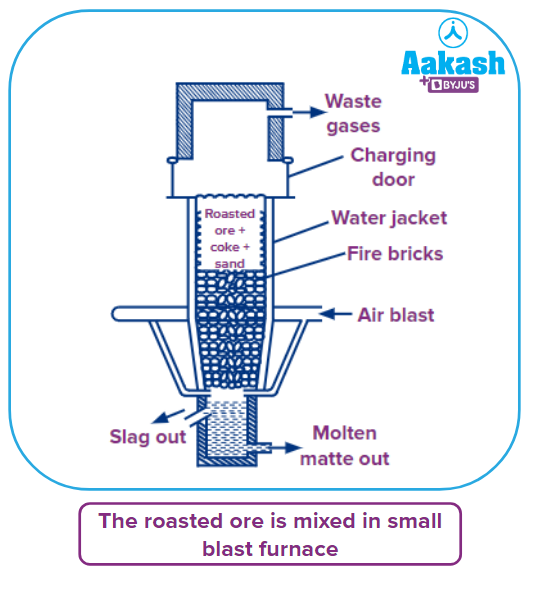
- Smelting: The roasted ore is combined with coke and silica in a small blast furnace.steel with refractory bricks on the inside. It stands between 15 and 20 feet tall. It is water jacketed all the way around and has a waste gas outlet near the top.
- Bessemerisation: The matte from the smelting process is fed into a Bessemer converter. A blast of air is pushed across the molten mass after some sand (silica) is added. A pear-shaped steel vessel lined with magnesite and quartz is used as a Bessemer converter. It has air-blast tuyeres and is placed in a way that allows it to be tilted in any direction. The Bessemer converter undergoes the following reactions.
- The ferrous sulphide that remains is oxidised.

- Slag is formed when ferrous oxide reacts with silica and is drained off at intervals by tilting the tank. All of the iron is eliminated as ferrous silicate in roughly three hours.
- The air blast continues for nearly another two hours. Excess silica is absorbed by the converter's basic lining, and a portion of cuprous sulphide is oxidised, forming free copper metal with the remaining cuprous sulphide.
This is an example of auto-reduction in action. The molten copper is poured off and allowed to cool before being used again. The dissolved sulphur dioxide evaporates after cooling, forming huge blisters on the surface. As a result, the metal created is known as 'blister copper.' Blister copper is made up of 98% copper and 2% impurities.

- Refining: Blister copper is refined using one of the two procedures listed below.
- Refining by Poling: The impure metal is melted in a silica-lined reverberatory furnace. A little amount of copper metal is oxidised to cuprous oxide, which dissolves in the melt and provides oxygen to the more basic elements present as impurities. Slag is formed when these components volatilize or mix with silica. By inserting poles of green woods, the amount of copper oxide that remains in the mass is lowered. The gas bubbles (hydrocarbons) that come from the wood act as reducing agents. With these poles, the mixture is vigorously churned. The procedure is known as poling. Tough pitch copper is made using this technique, which yields copper with a purity of 99.5 %.
- Electrolytic refining: Electrolytic refinement is the most common method of copper refinement. The electrolytic bath is composed of a 15% copper sulphate solution with 5% sulphuric acid. The anodes are blister copper, and the cathodes are pure copper thin sheets. Copper from the anodes dissolves as the current flows, whereas pure copper is deposited on the cathodes.
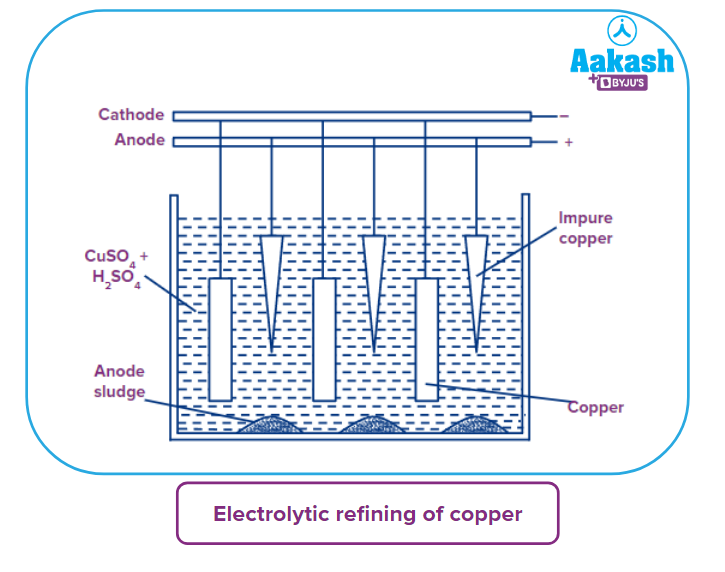
Cu2+ get deposited at cathode electrode whereas SO42- get deposited at anode electrode. Less electropositive impurities such as Ag, Au, and Pt gather below the anode in the form of anodic mud as more electropositive impurities dissolve in the solution, such as Fe,Zn,Ni,Co and so on. The purity of electrolytic copper is 99.96-99.99 %
Recommended Videos
Metallurgy Class 12 Chemistry 📑📑 (Ch-6) | Important Topics for JEE Main | JEE 2022 Exam Preparations
Practice Problems
1. ____________ is used to crush and concentrate the non-sulphide ores like oxide and carbonate ores.
a. Gravity Process
b. Bessemerisation
c. Hydrometallurgical Process
d. Electrolytic Refining
Answer: A
Solution: Gravity is used to crush and concentrate the oxide and carbonate ores. In a reverberatory furnace, the concentrated ores are calcined. Impurities either volatilize or are oxidised as the carbonate decomposes into oxide.
The resulting oxide is either reduced with carbon in a reverberatory furnace or leached with dilute H2SO4, depending on the method used.
So, option A is the correct answer.
2. ___________ is a copper carbonate ore.
a. Cuprite
b. Malachite
c. Bornite
d. Chalcopyrites
Answer: B
Solution: Malachite (CuCO3.Cu(OH)2 is one of copper carbonate ore. Chalcopyrites (CuFeS2) and Bornite (Cu3FeS3) are sulphide ores of copper. Cuprite (Cu2O) is one the oxide ore of copper.
So, option B is the correct answer.
3. Automobiles, aviation parts, and power tools are all made from__________.
a. tin alloys
b. magnesium alloys
c. copper alloys
d. aluminium alloys
Answer: B
Solution: Magnesium alloys comprise aluminium as well as minor amounts of other metals to provide strength and corrosion resistance. Magnesium alloys are utilised in the manufacturing of automobiles, aviation parts, and power equipment.
So, option B is the correct answer.
4. ___________ metal is also employed as a reducing agent in the tetrachloride production of titanium and zirconium.
a. Aluminium
b. Sodium
c. Magnesium
d. Copper
Answer: C
Solution: Magnesium metal is also employed as a reducing agent in the tetrachloride production of titanium and zirconium. The reaction can be written as
So, option C is the correct answer.
Frequently Asked Questions – FAQ
1. What are the top two considerations when deciding whether to extract a metal?
Answer: Before choosing to extract metal from its ore, a number of criteria need to be taken into account, including (i) the type of impurity, (ii) the type of metal, and (iii) the facilities that are accessible.
2. What is solvent extraction?
Answer: The transfer of one compound from one solvent to another due to the difference in distribution coefficient or solubility between two immiscible solvents is called solvent extraction. It has a stronger separation effect than chemical precipitation, as well as a higher degree of selectivity and faster mass transfer than the ion exchange strategy when compared to other separation procedures.
3. What do you mean by multiple extraction?
Answer: An amount of solvent is used to extract one layer (typically the aqueous layer) many times in succession in a multiple extraction technique. If the component has a low partition coefficient in the organic solvent, the extraction is performed two to three times, or even more times. This is known as multiple extraction.
4. Magnesium is collected from seawater in a variety of ways. Explain?
Answer: Solar evaporation, which causes chemical precipitation, is the most popular method for separating magnesium from seawater and brines. In saltwater, magnesium is the third most prevalent dissolved element. Solar evaporation is commonly used to handle highly concentrated brines like those found in the Dead Sea and the Great Salt Lake.


