-
Call Now
1800-102-2727
Exothermic and Endothermic Reactions-Definition, Plots of Exothermic and Endothermic Reactions, Difference Between Exothermic and Endothermic Reactions
It's a cold winter night and a man is all clad in a jacket warming himself up in front of a fireplace.

It’s the night that this hiker has to spend under an open sky and it is going to be pretty cold out there. But this fire here will surely keep him warm. What do you think would be happening while he is sitting next to the fire and absorbing all the heat? But how do you look at this process from a thermodynamic point of view? Do you think he is only absorbing the heat energy or do you think that he is also emitting some energy in the form of heat as well? Let’s look at two types of processes/reactions in detail which occur in nature where heat energy is absorbed and released.
Table of Contents
- Introduction to Exothermic and Endothermic Reactions
- Endothermic Reactions
- Exothermic Reactions
- Difference Between Endothermic and Exothermic Reactions
- Ways To Identify Exothermic and Endothermic Reactions
- Practice Problems
- Frequently Asked Questions – FAQs
Introduction to Exothermic and Endothermic Reactions
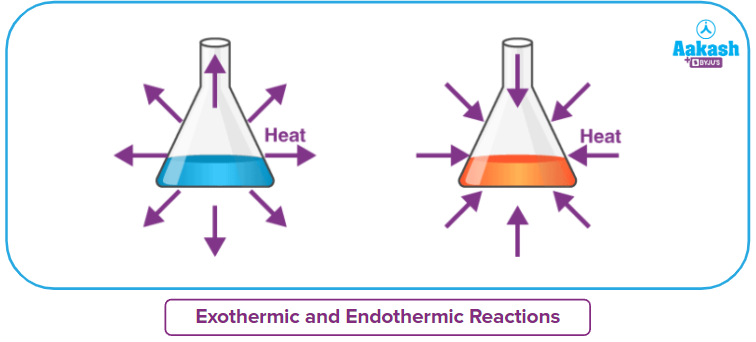
Let’s look at what is happening to the wood which is burning and it actually giving out heat. This means that this combustion process is giving out heat and the burning process is called an exothermic process in thermodynamics. The word “Exo” stands for external and “thermo” for heat. That makes the word exothermic refer to a process where there is a release of heat or one that gives out the heat to the surroundings.
But if we think in this way and consider this man near the fireplace as the system, this man is absorbing the heat and this process is called an endothermic process in thermodynamics. “Endo” stands for internal and therefore the word endothermic collectively refers to a process that absorbs the heat.
So, chemical processes or reactions are broadly classified into these two types: Exothermic & Endothermic reactions.
So, the endothermic reactions absorb energy from the surrounding in the form of heat. On the other hand, an exothermic reaction releases heat energy into the surrounding from the system.
Photosynthesis is an example of an endothermic process. In the photosynthesis process, plants absorb energy from the sun and convert carbon dioxide and water into glucose and oxygen. The reaction between sodium and chlorine, which produces a bright yellow light and heat energy is an example of an exothermic reaction.
Endothermic Reactions
The endothermic reaction is defined as a reaction where the system (where the reaction is occurring) absorbs the energy from its surrounding in the form of heat. A few examples of the endothermic process are photosynthesis, evaporating liquids, melting of ice, vapourisation of dry ice, cracking of alkanes, thermal decomposition, ammonium chloride in water and much more.
Let’s consider a reaction:
A + B → C + D; ΔH = +ve
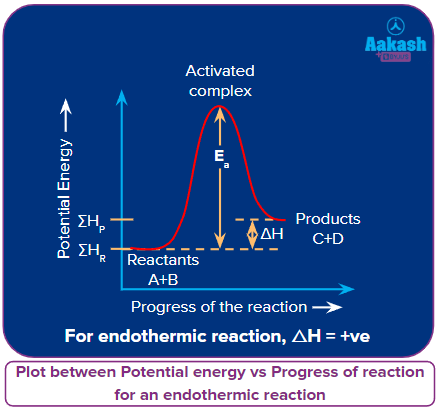
for an endothermic reaction.
In the endothermic reactions, the enthalpy during the conversion of reactants to products, increases i.e.,
Exothermic Reactions
The exothermic reaction is defined as a reaction where the system (where the reaction is occurring) releases the energy to the surrounding in the form of heat. A few examples are neutralisation reactions, burning of substances, burning of fuels, deposition of dry ice, respiration, dissolution of sulphuric acid into water etc.
The term ‘Exo’ refers to ‘to release’ and ‘thermic’ refers to ‘heat.’
Let’s consider a reaction:
A + B → C + D; ΔH= -ve
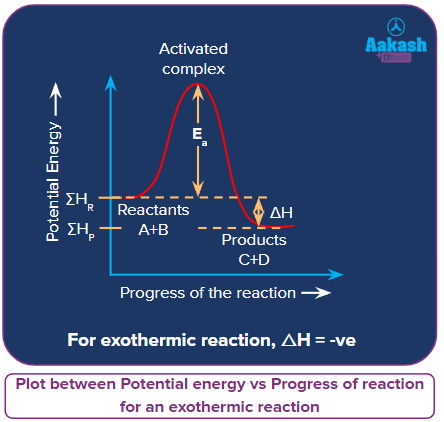
for an exothermic reaction.
In the exothermic reactions, the enthalpy during the conversion of reactants to products, decreases i.e.,
The heat energy is produced as a result of the reaction happening where reactants are converted into products. It occurs as a result of the dissociation of the bonds between the molecules. The heat energy is then released through the formation of new bonds.
Difference Between Endothermic and Exothermic Reactions
Endothermic Reaction |
Exothermic Reaction |
|
Endothermic reactions are chemical reactions in which the reactants absorb heat energy from the surroundings to form products. |
Exothermic reactions are chemical reactions in which energy is released in the form of heat to the surroundings to form products. |
|
The energy is absorbed from the surrounding into the reaction. |
The energy is released from the system to the surroundings |
|
The change in enthalpy for endothermic reactions is +ve. i.e., ΔH= +ve |
The change in enthalpy for exothermic reactions is -ve. i.e., ΔH= -ve |
|
Examples: Melting of ice, evaporation, cooking and photosynthesis |
Examples: Rusting iron, explosions, nuclear fission and fusion reactions |
Ways To Identify Exothermic and Endothermic Reactions
Procedures to differentiate between exothermic and endothermic reactions are as follows:
- Monitoring the change in temperatureThe temperature of a system (where the reaction is occurring) rises in an exothermic reaction when the heat energy is given out to the surrounding. Whereas the temperature of the reaction decreases when energy is absorbed. You can monitor changes in temperature by placing a thermometer in the reaction mixture.
- Enthalpy change of reaction (rH )The enthalpy change when the reactants are converted into products in a particular chemical reaction at any temperature and pressure is known as the enthalpy change of a reaction, which is represented as and is calculated as follows:
For example, consider the reaction given,
- Where, Hf = Enthalpy of formation
The reactions that absorb heat energy are known as endothermic reactions. In such cases, the enthalpy of the reactants is less than the enthalpy of the products so that rH is positive.
The reactions that release heat, i.e., the reactions that are accompanied by the evolution of heat are known as exothermic reactions. In such cases, the enthalpy of the products is less than the enthalpy of the reactants, so that rH is negative.
Example: The molar enthalpy of water vapour at 298 K and 1 atm can be represented as follows: Hm (H2O, g, 298 K, 1 atm) Where, g represents the state of the substance, i.e., a gaseous state.
rH = (Sum of enthalpies of products) – (Sum of enthalpies of reactants)
rH = energy utilized to break the bonds of reactants + energy that is released to make the new bonds of the product
The values of the enthalpies are added in the equation mentioned above as, according to the description, the energy that is utilized in breaking the bonds of a reactant tends to be positive (+) and energy that is given out in order to make the bonds of products tends to be negative (−).
A reaction is exothermic when the rH is negative as the energy given out to form the products is much greater than the energy used to break the bonds of reactants.
A reaction is endothermic when the rH is positive as the energy that is released to make the products is a lot lesser than the energy that gets absorbed to break the bonds of reactants.
Practice Problems
Q. The reactions which can not occur spontaneously are:
a. Exothermic
b. Endothermic
c. Isothermic
d. None of the above.
Answer: (B)
Solution: Endothermic reactions absorb heat from the surroundings, so this reaction will occur only when the heat will be provided. So they do not occur spontaneously.
Q. Which of the following is an example of an endothermic reaction?
a. Reaction of carbon with oxygen forming carbon dioxide
b. Melting of ice
c. Burning of fuels
d. None of the above
Answer: (B)
Solution: The melting of ice is the process, where ice melts into the water at its melting point i.e., 100 ℃. This melting process happens when the ice absorbs the heat from the surrounding. When the heat is absorbed, the force that holds the molecules together weakens up and solid ice is converted into liquid water, the process of absorbing heat from the surroundings is known as the endothermic process. In all other options, heat energy is being released making them exothermic processes.
Q. Which of the following reaction is an endothermic reaction?
a.
b.
c.
d.
Answer: (B)
Solution: An endothermic reaction is the type of process where an increase in the enthalpy of the system occurs. Enthalpy change of those reactions is positive. The formation of NO is an example of an endothermic reaction as energy will be required to break the triple bonds in N2.
Frequently Asked Questions – FAQs
Q. In a chemical reaction, why is heat released or absorbed?|
Answer: Chemical bonds are either broken or formed in any chemical reaction. Heat is released when chemical bonds are formed, and heat is absorbed when chemical bonds are broken. Since molecules want to remain together, forming chemical bonds between them requires less energy than breaking them, resulting in heat being absorbed from the surroundings.
Q. Define the enthalpy of a reaction.
Answer: The enthalpy of a reaction is defined as the heat energy change that occurs when reactants become products. ΔH is positive if heat is absorbed during the reaction; if heat is released, ΔH is negative.
Q. During the endothermic reaction, what type of energy conversion will take place?
Answer: This is the conversion of kinetic energy to chemical energy. Heat is absorbed and converted into chemical energy during an endothermic reaction.
Q. Why is respiration regarded as an exothermic reaction?
Answer: Since energy is released during the process of respiration, it is an exothermic process. The carbon dioxide in the food is broken down to form glucose during the process. This glucose combines with oxygen in our cells, releasing a large amount of energy.
Q. What is the main difference between an exothermic and an endothermic process?
Answer: The main difference between exothermic and endothermic reactions is that an endothermic reaction absorbs energy in the form of heat from the surroundings, whereas an exothermic reaction releases energy to the surroundings.


