-
Call Now
1800-102-2727
Ethers Preparation: Introduction of Ethers, Ethers Preparations, Uses of Ethers, Practice Problems, Frequently Asked Questions
You must have seen tile flooring in a lot of locations.
Ever wonder how these tiles are so firmly attached to the floor?
To adhere the tile to the floor surface, we use free-flowing mortar, and this combination contains cellulose ether to make the mortar free-flowing and aid in the firm fixing of the tiles. One use for ethers is in this situation.

The realm of medicine also makes use of ethers. Ethers have been used as anaesthetics in the past. Ether was initially employed as an anaesthetic, according to historical documents, in 1842. Painkillers also contain ethers as an ingredient. Codeine, a commonly used painkiller, is morphine that has been etherized.
Excellent solvents include ethers. They are used in a variety of chemical processes, including extraction. Because they are volatile, they are utilised to start diesel and gasoline engines in cold areas.
MTBE (methyl tert-butyl ether), a kind of ether, is added to gasoline to increase octane while reducing nitrogen oxide pollution. The use of dimethyl ether as a refrigerant is common.
On this concept page, we will discover more about ethers, their preparation, and their applications.
Table of contents:
- Ethers - Introduction
- Ethers Preparations
- Uses of Ethers
- Practice Problems
- Frequently Asked Questions(FAQs)
Ethers - Introduction
A chemical bond between two alkyl or aryl groups via an oxygen atom defines the class of organic compounds known as ether. Water, alcohol, and ether all have comparable chemical compositions. Water has two hydrogens connected through an oxygen atom. You replace one hydrogen with a hydrocarbon group of alkyl or aryl, you have alcohol or phenol. Replacement of both hydrogens by hydrocarbon groups produces ethers.
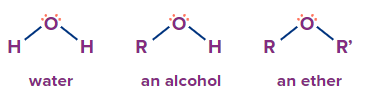
Ethers are colourless liquids with a pleasant aroma at room temperature. Ethers are less dense, have lower boiling points, and have lesser miscibility with water compared to alcohol. Ethers are the preferred solvents for quick-drying combinations containing perfumes, and as a nonreactive solvent for waxes, fats, gums, dyes, and resins. The high volatility of ethers is useful in the preparation of spray chemicals containing pesticides, insecticides, and fumigating agents in their gaseous state to improve resistivity.
Alcohol (except methanol) and ethers exist as isomers. Ethers lack the highly polarised chemical bond that exists in alcohols between the hydrogen and oxygen atoms. Ethers are unable to form hydrogen bonds with one another as a result.
However, the oxygen atoms in ether possess uncoupled electron pairs that can be used to create hydrogen bonds with molecules other than ethers. For instance, O-H or N-H bonds, which are hydrogen bonds, can form with alcohols or amines.
Classification of ethers:
Based on the substituent group attached to its oxygen atom, ethers can be divided into two main categories: symmetrical ethers and asymmetrical ethers.
- Symmetrical ethers or simple ethers: The same alkyl or aryl groups are attached on either side of the -O- functional group in these ethers.
Example: (diethyl ether), (dimethyl ether)
- Unsymmetrical ethers or mixed ethers: The different alkyl or aryl groups are attached on either side of the -O- functional group in these ethers.
Example: (ethyl methyl ether), (methyl phenyl ether).
Ethers Preparations:
1. Intermolecular dehydration of alcohol:
Alcohol dehydrates under various conditions when protic acids (sulfuric acid, phosphoric acid) are present, resulting in the production of alkenes and ethers. The reaction products formed depend on the conditions under which the reaction was conducted.
For instance, ethanol dehydrates to ethylene at 443 K when sulfuric acid is present. In contrast, ethanol produces ethoxyethane at 413 K when sulfuric acid is present.
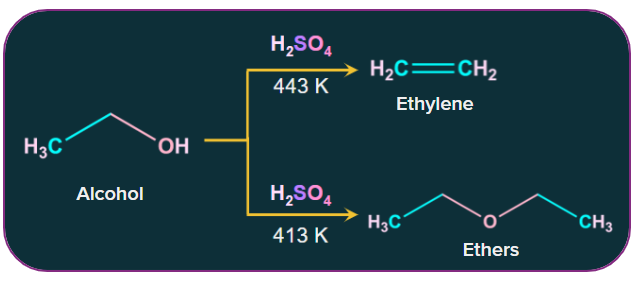
Ethyl alcohol on heating in the presence of acid as a catalyst, 443K form diethyl ether.
2CH3CH2OH → CH3CH2OCH2CH3 + H2O
The formation of ether by intermolecular dehydration of alcohol occurs by an SN2mechanism.
Mechanism:
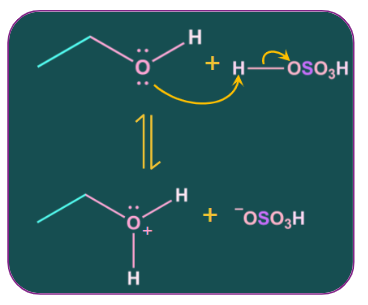
Step-1: Protonation of alcohol.
Proton from acid gets attached with the lone pair of electrons of oxygen of the alcohol. Alcohol acts as a nucleophile and the proton acts as an electrophile.
Step-2: Attack of nucleophile
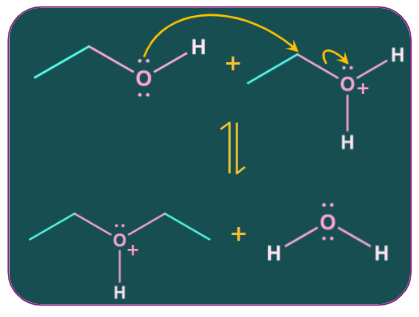
Because of the positive charge on oxygen, the alpha carbon atom to oxygen becomes partially positive., facilitating a nucleophilic attack by the oxygen of a second alcohol molecule on it. The attack results in an intermediate of protonated ether and neutral water molecules.
Step-3: Deprotonation
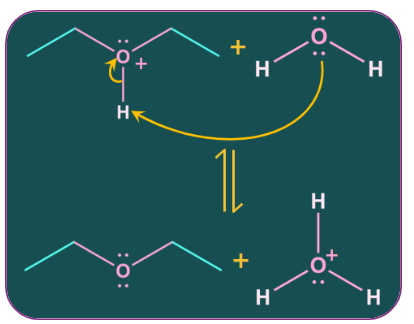
Deprotonation of the hydrogen ion takes place as an intramolecular attack on it by a water molecule which produces an ether.
Limitations of intermolecular dehydration:
The method of dehydration of alcohol to produce ethers is used to produce ethers containing primary alkyl groups. Among the precautions taken to ensure that the reaction proceeds properly are:
1. The presence of other groups should not interfere with the alkyl groups present in the alcohols used.
2. The temperature must be kept as high as possible.
Taking care of these details will ensure that ethers rather than alkenes are formed in the reaction. The steps with secondary or tertiary alcohol follow the SN1 mechanism rather than the SN2. However, the dehydration reaction for secondary and tertiary alcohols is not as successful as that of primary alcohols because, instead of substitution, elimination occurs, resulting in the formation of alkenes.
Additionally, only symmetrical ethers can be formed through the dehydration of alcohol to produce ethers. The preferred method of making unsymmetrical ethers, such as ethyl methyl ether, is to use suitable sodium alkoxides and an alkyl halide, as in the Williamson synthesis.
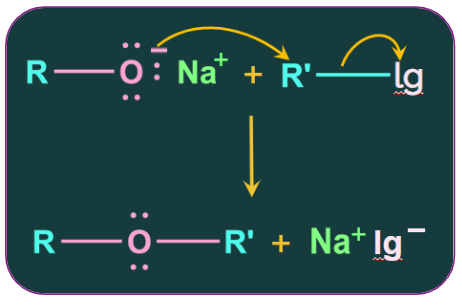
2. Williamson Ether synthesis:
This is a crucial technique for creating symmetric and asymmetric ethers in a laboratory.
An ether is produced in the Williamson synthesis when an alkyl halide and a sodium alkoxide react.
Nature of alkoxide:
Alkoxide acts as a strong nucleophile with a lone pair of electrons of oxygen. So, the higher the nucleophilicity of the oxygen, the stronger will be the nucleophilic attack on the alkyl halide. Among the alkoxide, it is known that nucleophilicity increases from the primary to a tertiary alkyl group.
Tert. alkyl > sec. alkyl > primary alkyl.
Hence a tertiary alkoxide will be highly suitable for Williamson synthesis of ether.
Nature of alkyl halide:
An alkyl halide is the target of an SN2 attack by an alkoxide ion in this reaction. The suitability of the alkyl halide depends on-
- Having a good leaving group of halides.
The leaving group ability follows the order, I > Br > Cl > F.
So, an alkyl iodide facilitates Williamson's synthesis.
- Attack on the halide-attached carbon must be easy, which means that the halide carbon should be sterically unhindered. Steric hindrance increases in the order, tert > secondary > primary.
Primary carbon has the least steric hindrance.
A primary iodide hence will be the most ideal halide for the formation of ether by Williamson's synthesis.
A primary or secondary alkyl halide with tertiary alkoxide forms ether by Williamson's synthesis.
4. Reaction of an alkyl halide with dry silver oxide ( Ag2O):
Ether is formed when the dry silver oxide is used to treat alkyl halide.
Uses of Ethers:
- Due to diethyl ether's additional favourable therapeutic index, which is a greater distinction between an effective dose and a potentially toxic dose, it largely replaced the use of chloroform as an anaesthetic.
- In the creation of fragrances or fragrant goods like soaps, detergents, and everyday household items like essential oils, medications etc some new ether compounds, like diethyl ether, are particularly beneficial.
- Dimethyl ether (DME), a solvent used in lithium batteries, is easily miscible with water.
- Because it is water soluble, the cyclic ether 1,4-dioxane is frequently used as a solvent in laboratories for the synthesis of chemicals.
Practice Problems:
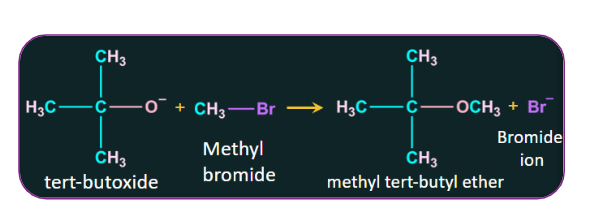
Q1. The reaction of the tert-Butoxide ion with methyl bromide to give methyl tert-butyl ether is known as
(A) Williamson ether synthesis
(B) Swartz reaction
(C) Wurtz reaction
(D) None of the above
Answer: (A)
Solution: Williamson ether synthesis is the process by which the tert-Butoxide ion reacts with methyl bromide to produce methyl tert-butyl ether. Following is the reaction:
When performing a Williamson ether synthesis,
- The alkyl halide should have a less hindered halide carbon for nucleophilic attack by the nucleophile and substitution..
- The alkoxide should have more substitution by electron-donating groups, to increase the nucleophilicity of oxygen.
Q2. Ethers in reaction with atmospheric oxygen in the presence of sunlight give
(A) Peroxide
(B) Alcohol
(C) Superoxide
(D) None of the above
Answer: (A)
Solution: When ethereal oxygen comes into contact with atmospheric oxygen in the presence of sunlight, it reacts with oxygen to form ether peroxide because it has a single pair of electrons. Serious accidents frequently happen because ether peroxide is incredibly unstable and violently explodes when heated. As a result, boiling an ether sample that has been stored for a long time is risky.
Q3. The formula of Methyl phenyl ether is
(A)
(B)
(C)
(D) None of these
Answer: (A)
Solution: The formula of methyl phenyl ether is and its common name is anisole. The common name of phenoxy benzene is diphenyl ether and its formula is . The common name is benzophenone.
Q4. The IUPAC name of diethyl ether is
(A) Ethoxy ethane
(B) Methoxy ethane
(C) Methyl phenyl ether
(D) None of the above
Answer: (A)
The common names of ether are written by mentioning the two alkyl groups in alphabetical order with an ending of ‘ether’.
For instance, t-butyl methyl ether contains the alkyl groups t-butyl and methyl group separated by an oxygen atom.
Ethyl methyl ether contains ethyl and methyl alkyl groups connected to an oxygen atom.
In IUPAC nomenclature, ether is considered a substituted product of a hydrocarbon. As usual, hydrocarbon has the highest carbon chain. The alkyl group with a relatively less carbon chain is considered a substituted group along oxygen and is mentioned as an alkoxy substituent.
Ethers are thus alkoxy alkanes in the IUPAC system.
So the IUPAC name of ethyl methyl ether is methoxy ethane and its formula is
Frequently Asked Questions(FAQs)
Q1. Why do ethers, which have the same molecular weight as alcohols, have lower boiling points?
Answer: Ethers are unable to form hydrogen bonds with one another. However, the oxygen atoms in ether have unbonded electron pairs that can be used to form hydrogen bonds with molecules that are not ethers.
Ether's ability to form hydrogen bonds with other compounds allows it to be used as a solvent for other organic and inorganic compounds. Ethers cannot form hydrogen bonds with one another. Because of this, they have a lower boiling point than alcohols with the same molecular weight.
Q2. Why do ethers have lower reactivity?
Answer: Most substances, particularly bases and alkali metals, are chemically inert to ethers. When reacted with mineral acids and Lewis acids, they form oxonium compounds due to their weak basicity caused by the presence of non-bonded electron pairs on the oxygen atom. It is inert in comparison to alcohol because alcohol has an activated O-H bond, whereas in ether both oxygen atom bonds are attached to an alkyl group, making it more difficult to attack such species and thus less reactive.
Ethers are only reactive when oxygen is protonated in an acidic medium. The ether undergoes a number of reactions to lose its positive charge on oxygen. SN1 and SN2 are the two most well-known cleavages.
Q3. Why are ethers effective as solvents in many organic reactions?
Answer: Alcohols are both H-bond donors and acceptors, whereas ethers can only accept H-bonds. Ethers make excellent solvents for a variety of organic compounds due to their capacity to accept H-bonds and the London forces created by the alkyl groups attached to the oxygen. Ethers are a preferred solvent for many organic reactions due to their low chemical reactivity. Ethers high volatility also makes it possible for them to evaporate when separating reaction products.
Q4. What exactly are crown ethers?
Answer: Crown ethers are cyclic polyethers composed of two or three carbon atoms separated by four or more oxygen atoms. Crown ethers are named using both the total number of atoms in the ring and the number of oxygen atoms and have the general formula or As a result, 18-crown-6 is an 18-membered ring containing six oxygen atoms. A cavity in the centre of all crown ethers is lined with oxygen atoms and can accommodate an alkali metal ion. The cation is stabilised by interacting with lone pairs of electrons on the oxygen atoms around it.
Crown ethers are helpful for dissolving ionic substances in organic solvents, like KMnO4 dissolving in toluene, by sequestering the cations inside a cavity that is hydrophilic while the outer shell, made up of C-H bonds are hydrophobic.


