-
Call Now
1800-102-2727
Ethers Classification: Introduction to Ethers, Ethers Classification, Properties of Ethers, Uses of Ethers, Practice Problem, Frequently Asked Questions
Have you heard of fresheners? Fresheners are used in rooms, halls, cars, toilets, and hospitals, either to mask bad odours or to have a feel-good smell for the occupants. These fresheners have some fragrances, disinfectants, and adsorbents as active materials. But, all of these are dispersed in a medium. Glycol ethers are one of the solvents used to disperse the active ingredients of fresheners for application.
Ethers themselves are a class of organic compounds differing in components, structure and uses.
Ethers are found most useful, particularly in the perfume industry.

Because of their olfactory and organoleptic properties, ethers are often employed in the fragrance and smell industries.
Anisole, often referred to as methyl benzene, belongs to the group of chemicals called ethers.
Methylbenzene (anisole) is a frequent component used to produce an anise seed essential oil smell.
Dimethyl ether (DME), a solvent used in lithium batteries, is easily dissolved in water.
Because it is water soluble, the cyclic ether 1,4-dioxane is frequently used as a solvent in laboratories for the production of compounds.
They improve the flavour of a wide range of foods and medications, including sweets, fruit jams, yoghurt, ice cream, bubble gum, fruit-based foods and beverages, and several pharmaceutical preparations.
Table of Content:
- What are Ethers?
- Nomenclature of Ethers
- Ethers Classification
- Properties of Ethers
- Uses of Ethers
- Practice Problems
- Frequently Asked Questions(FAQs)
What are ethers?
One oxygen atom joins two hydrocarbon groups in an organic molecule to generate ethers (alkyl or aryl). The R-O-R formula is used to represent it. The hydrocarbon group in the formula R′ could be the same as R or different from R. When an alkyl or aryl group is substituted for the hydrogen atom of the hydroxyl group in alcohols, ethers are produced.
The C-O-C bond's oxygen and carbon are sp3 hybridised. The oxygen atom's two lone pairs repel one another, creating the bending shape. A steric barrier is created by the presence of bulky groups on both ends of the oxygen atom and the bp-bp repulsion that results in a C-O-C bond angle of approximately 111.7o.

This subject is frequently studied in organic chemistry, and it also appears frequently in biochemistry, where we discover similar connections between lignin and carbohydrates.
Nomenclature of Ethers:
Two methods are followed in the naming of ethers - Common name and IUPAC nomenclature.
Common Name:
Here the ether compounds have their alkyl groups written in alphabetical order followed by the ending ‘ether’.
Example: methyl t-butyl ether and ethyl methyl ether are two examples.

In the case of the same alkyl or aryl group on either side of oxygen, the prefix ‘di’ is used before the alkyl or aryl name. For example, ethyl ether is named diethyl ether.
IUPAC Nomenclature:
In IUPAC nomenclature ethers are considered as a substituted hydrocarbon. The longest hydrocarbon of the two attached groups is taken as the parent hydrocarbon and the smaller hydrocarbon is placed as a prefix with an ‘oxy’ ending.
Ethers are therefore alkoxy alkanes according to the IUPAC nomenclature.
Examples: diethyl ether is ethoxyethane, methyl tert-butyl ether is 2- methoxy 2- methylpropane, diphenyl ether is phenoxy benzene and ethyl methyl ether is methoxy ethane.
Ethers Classification:
Based on the substituent group connected to its oxygen atom, ethers can be divided into two main categories: symmetrical ethers and asymmetrical ethers.
Symmetrical Ethers or simple Ethers: Simple ethers, also known as symmetrical ethers, have identical alkyl or aryl groups affixed to both sides of the functional group -O-.
Example: CH3-O-CH3 (dimethyl ether), C2H5-O-C2H5 (diethyl ether).
Unsymmetrical Ethers or mixed Ethers: Ethers with different alkyl or aryl groups connected on either side of the functional group -O- are referred to as unsymmetrical ethers or mixed ethers.
Example: CH3-O-C6H5 (methyl phenyl ether), C2H5-O-CH3 (ethyl methyl ether).
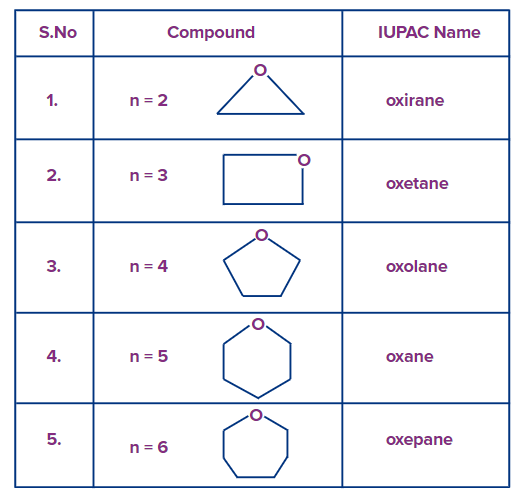
Cyclic Ethers: An example of a heterocycle is a cyclic ether, which has one or more oxygens inside the ring. Due to their inert nature, cyclic ethers are commonly utilised as solvents and have many common names. Numerous biological compounds, including sugars and DNA, also have these ring configurations.
For instance, oxirane, also referred to as epoxide, is a form of cyclic ether.
The prefixes "irane," "etane," "olane," and "ane" are added after the prefix "ox" in the IUPAC nomenclature for cyclic ethers to denote the number of carbon atoms in the three-, four-, five-, and six-membered ring systems, respectively.
Crown Ethers: Crown ethers are cyclic polyethers that have two or three carbon atoms between each of their four or more oxygen atoms. Crown ethers are named using both the number of oxygen atoms in the ring and the generic formula of (OCH2CH2)n or (OCH2 CH2CH2)n. A ring having 18 members and six oxygen atoms is called 18-crown-6. A K+ like alkali metal ion may fit into the central cavity of all crown ethers, which is lined with oxygen atoms. The lone electron pairs on the oxygen atoms in the vicinity interact with the cation to stabilise it.
As a result, ionic compounds like KMnO4 can be dissolved by crown ethers. Due to its hydrophobic nature, the ring can combine with nonpolar liquids. In nonpolar solvents like toluene, the dispersed ionic compounds will therefore dissolve. Ionic compounds can also be dissolved in nonpolar solvents using crown ethers.
Properties of Ethers:
- Methoxy methane and methoxy ethane are gases, while other components are tasteless flammable liquids. Lower homologues include colourless, fragrant, flammable liquids with distinct ether odour.
- Due to hydrophobic alkyl or aryl groups, ethers are only sparingly soluble in water.
- Ethers don't have hydrogen bonds among themselves. Because of this, their boiling points are much lower than those of comparable alcohols.
- Because diethyl ether vapours cause drowsiness when breathed, they are used as anaesthetics.
Uses of Ethers:
- Dimethyl ether is used as a solvent at ambient temperature.
- Diethyl ether is used as a solvent for gums, oils, and resins and diethyl ether vapours cause drowsiness when breathed, they are used as anaesthetics.
- In the creation of scents or aromatic goods like soaps, detergents, and everyday household things like essential oils, pharmaceuticals, etc., some novel ether compounds, like diethyl ether, are particularly beneficial.
- Because phenyl ether has a high boiling point, it can be utilised to transmit heat.
- The cyclic ether tetrahydrofuran (THF) is extensively used as an organic compound solvent. They are more suited for usage as solvents because of their lower boiling points since evaporation makes it straightforward to remove them from a molecule after a reaction mechanism.
Practice Problems:
Q1. Among the following, which one is not an aromatic ether?
(A) C6H5-O-C6H5
(B) CH3-O-C6H5
(C) C2H5-O-C6H5
(D) CH3-O-C2H5
Answer: (D)
Solution: Aromatic ethers are those in which there is at least one aryl group on either side of the oxygen atom. In the compound, CH3-O-C2H5, both groups are alkyl, making it an aliphatic ether.
Q2. Ethers have a boiling point that is _____alcohols with a similar molecular mass.
(A) higher than
(B) lower than
(C) Similar to
(D) little higher than
Answer: (B)
Solution: Ethers' boiling point is lower than that of isomeric alcohols but closer to that of alkanes of the same mass because they do not form intramolecular hydrogen bonds. Methoxy methane (CH3OCH3) has a lower boiling point than ethanol (CH3CH2OH) despite having the same chemical formula ( C2H6O).
Q3. Ethers are
(A) Volatile
(B) Non-volatile
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: Since the ethers lack hydrogen bonding they have the least intermolecular interactions, resulting in lower boiling point and higher volatility.
Q4. What kind of intermolecular forces exist in ethers?
(A) Dipole-Dipole interactions
(B) London dispersion forces
(C) Ion-Dipole interactions
(D) None of the above
Answer: (A)
Solution: The opposing partial charges on the two ethyl ether molecules attract one another when they are combined. Because it happens in polar molecules containing dipoles, this kind of intermolecular force is known as dipole-dipole interaction or dipole-dipole attraction.
Frequently Asked Questions(FAQs):
Q1. Why do ethers containing up to three carbon atoms dissolve in water?
Answer: In water, ethers comprising up to three carbon atoms are soluble. The nonpolar hydrophobic hydrocarbon chain and the polar hydrophilic oxygen end are the two different types of molecules that make up ethers. Hence, smaller ethers can form hydrogen bonds with water molecules. As the alkyl group size grows, ether becomes less soluble in water, because the hydrocarbon component is non-polar, that prevents hydrogen bonds from forming with the polar water molecules.
Q2. Is there a dipole moment in ether?
Answer: Yes, Ethers possess a dipole moment and are polar in nature. The cause of this is the difference in electronegativity among oxygen and carbon atoms. Because of the disparity in electronegativity, the oxygen atom draws the sharing electron pair closer to itself, which allows the oxygen atom to have a partial negative charge and the carbon atom to have a partial positive charge. In the ether, two polar C-O bonds confront one another at an angle of 111.7 degrees. The outcome is a net dipole moment since the two dipoles do not cancel out. In other words, the dipole moment in ether is ≠0.
Q3. Why is ether stored in a container with iron wire instead of being exposed to direct sunlight?
Answer: Ether powerfully reacts with the oxygen in the air when it is subjected to sunlight, yielding peroxide. When heated, peroxide ignites, causing a serious accident. Iron oxide is formed when oxygen mixes with iron to hold ether in a bottle with iron wire, preventing ether from turning into peroxide. Therefore, ether is kept in a container that also contains iron wire.
Q4. Ethers have a lower reactivity than alcohols and carbonyl compounds. Why?
Answer: Ethers are substantially less reactive than alcohols and carbonyl compounds, but substantially more reactive than alkanes. Ethers are often less reactive because they lack polarity or charge separation in the molecule. There is no transitory or partial positive charge, unlike carbonyl carbon. Therefore, there is no significant impact when nucleophiles interact with ethers (no reaction takes place). As a result, ethers are unaffected by bases, alkali metals, active methylene groups, etc.


