-
Call Now
1800-102-2727
Entropy and Second Law of Thermodynamics: Entropy, Laws of Thermodynamics, Entropy and Second Law of Thermodynamics, Entropy Calculation, Practice Problems, FAQs
You must have played with balloons in your younger days. Can you tell me, whether filling up a balloon or releasing the air from it, was easier or spontaneous (taking place on its own, without external assistance)?

Consider a box partitioned into two compartments. One compartment contains greenish-yellow chlorine gas, while the other is filled with violet iodine vapours. If you remove the partition, each gas moves to the other compartment and get mixed, as shown by the colour change. Is there a chance to force them back to individual boxes? No.
In both cases, movement(process) to one side is spontaneous, while the reverse process is non-spontaneous.
In the above cases again you can clearly say which process is spontaneous. But there are many cases, both physical and chemical processes which cannot be seen. If so, how to predict spontaneity? The second law of thermodynamics gives a factor to predict spontaneity.
Come let us know about this entropy and the Second law governing it now.
Table of Content
- Entropy
- Laws of Thermodynamics
- Entropy and Second Law of Thermodynamics
- Entropy Change Calculation
- Practice Problems
- Frequently Asked Questions
Entropy
Entropy is defined as a measure of the degree of randomness or disorderliness in a system. The greater the disorderliness in an isolated system, the higher the entropy. Entropy is denoted by the letter 'S'.
Remember, how do you describe the three states of matter? The random movement is maximum for molecules in the gaseous state followed by the liquid state and then the solid state.
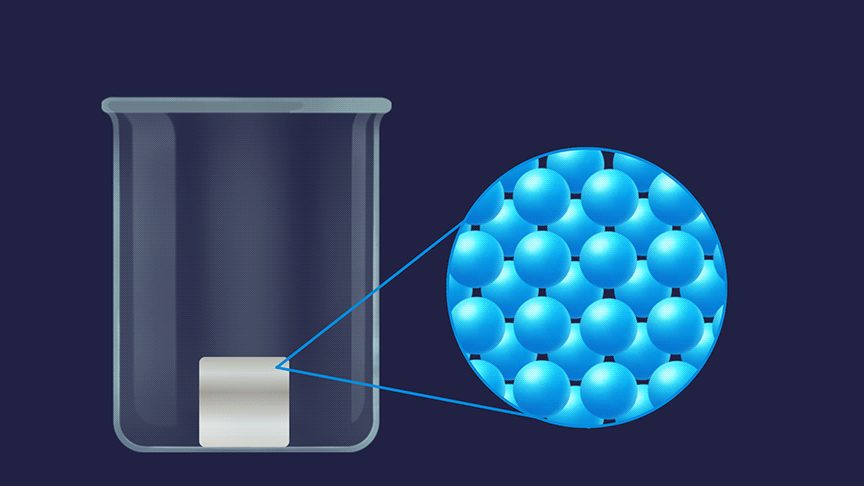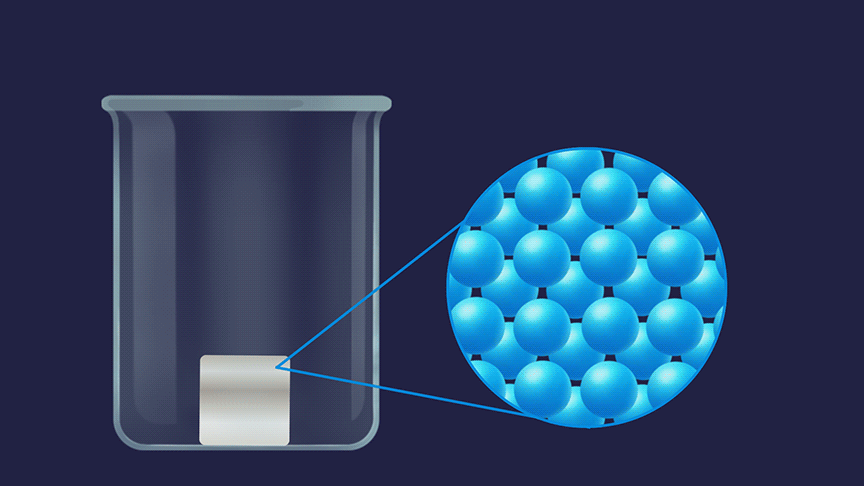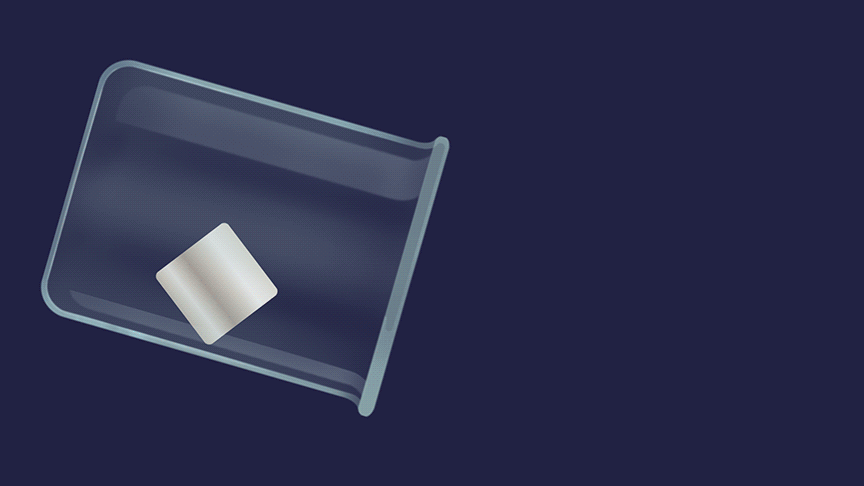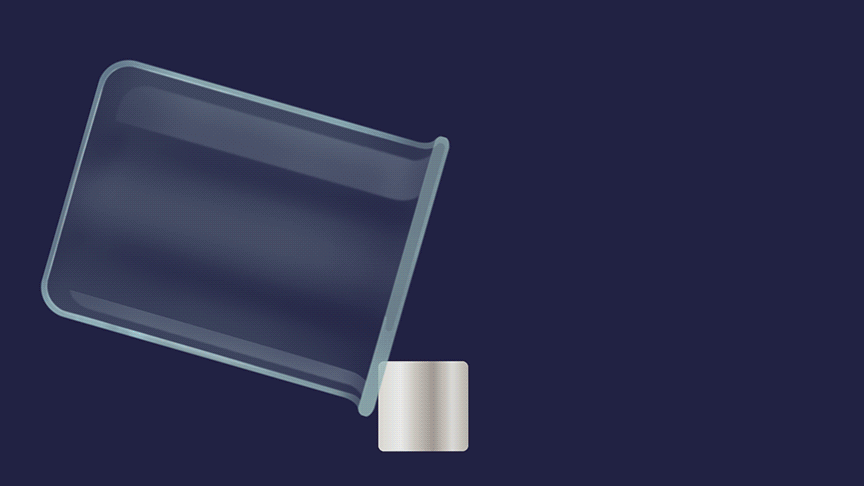
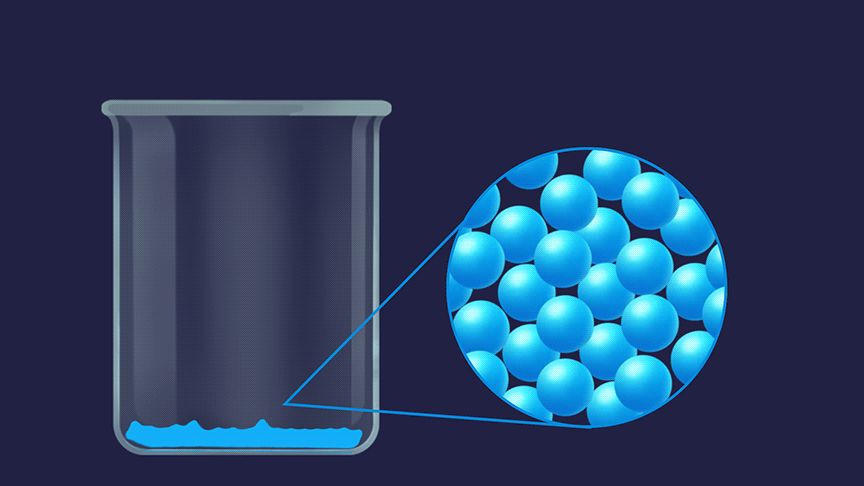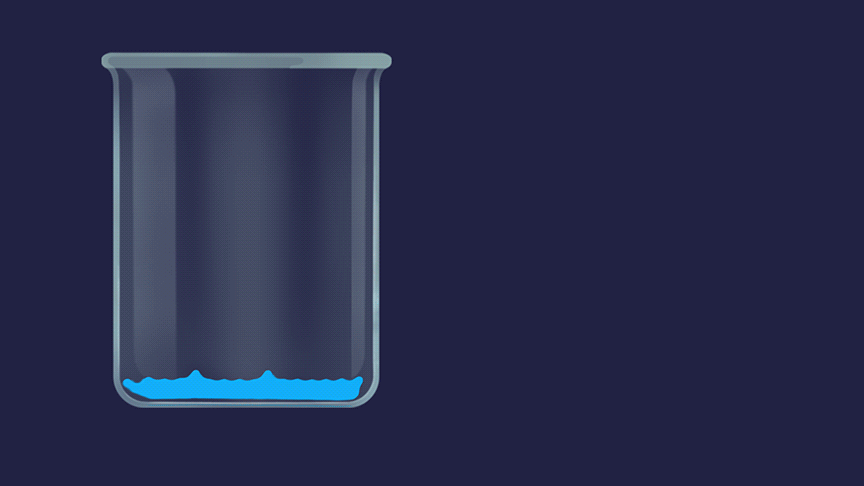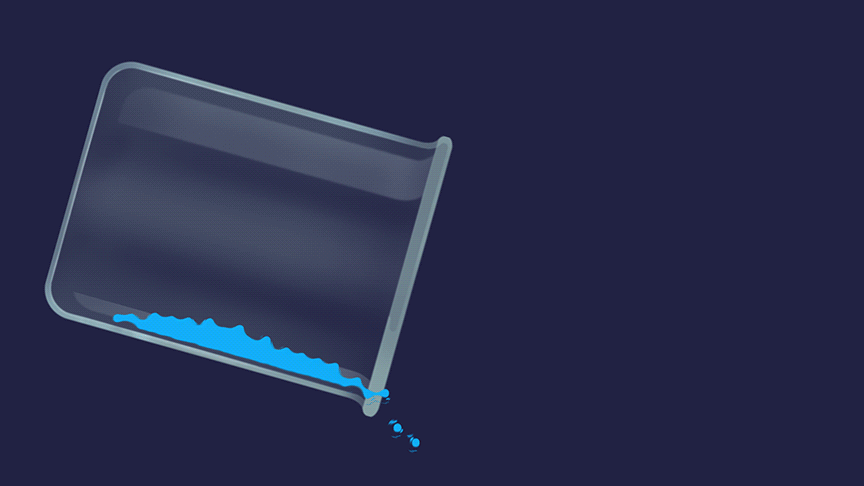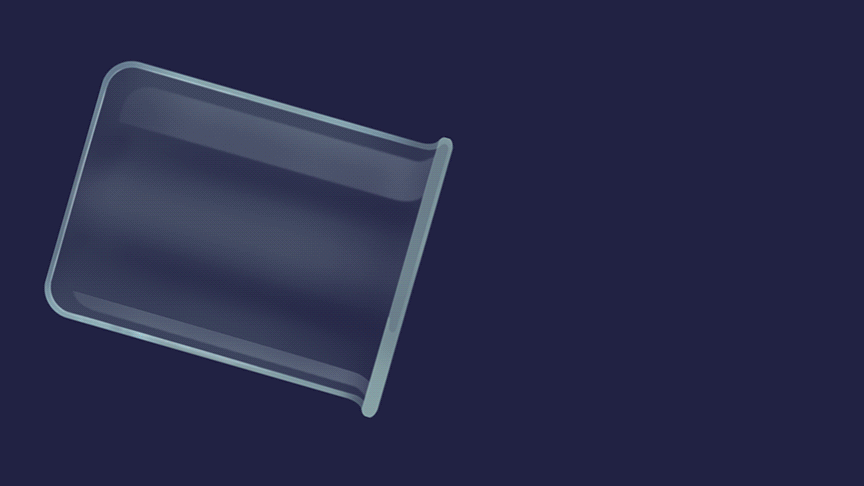
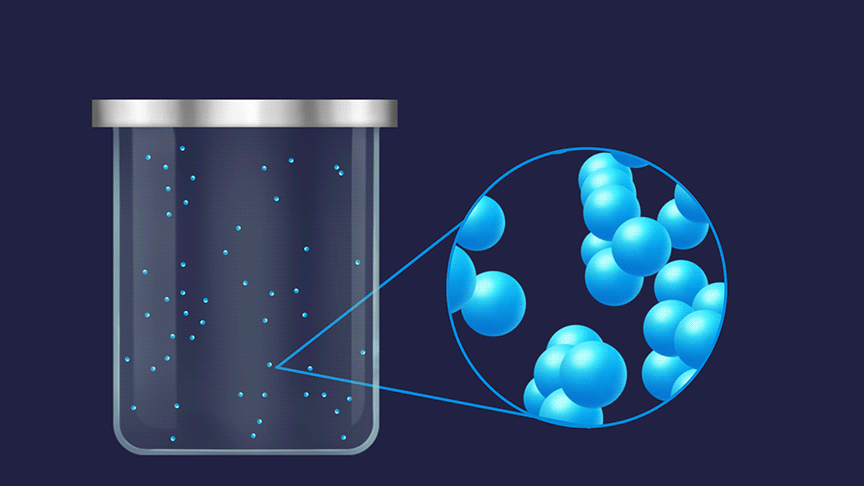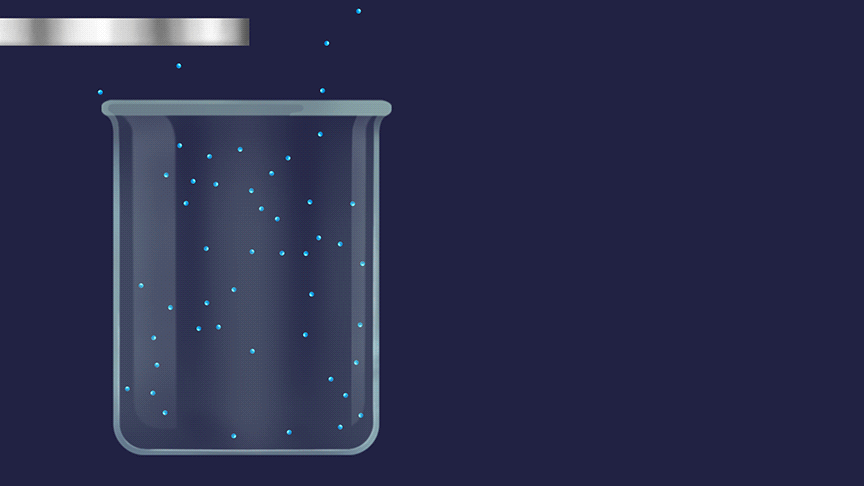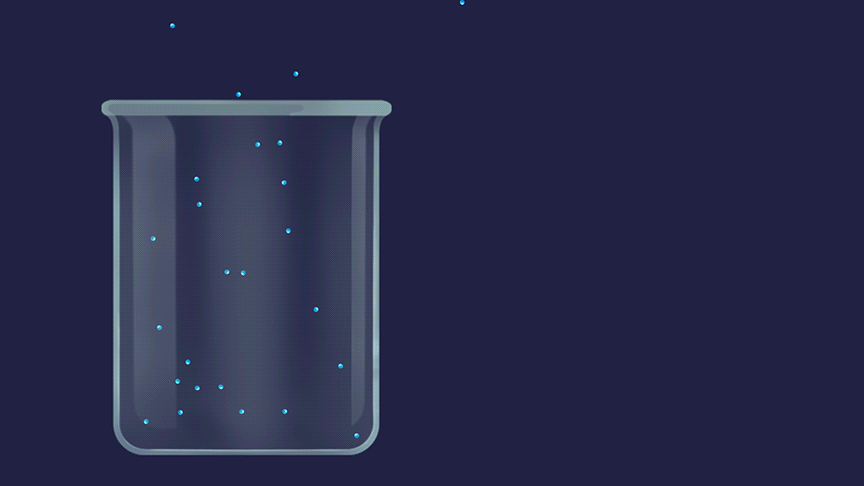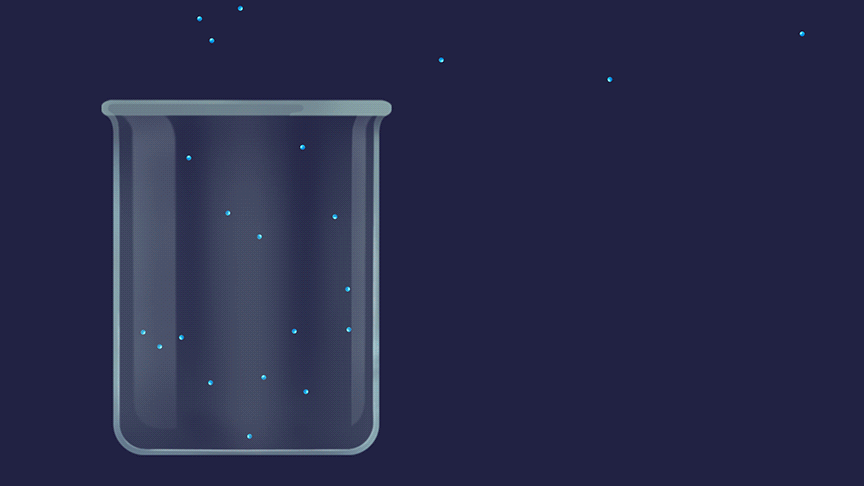

The order of entropy for different physical states is given as:
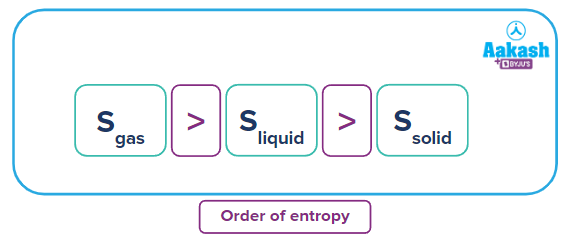
Now looking back to the spontaneous-nonspontaneous examples we have seen,
Air is freer to move or has more disorderliness in the atmosphere, while inside the balloon, the space is small and hence restricted to move and has less disorderliness. For filling a balloon, you have to do work or push air and hence is a nonspontaneous process. A nonspontaneous process needs external work to make it happen. On opening the nozzle of a filled balloon, air automatically, or spontaneously escapes out into the atmosphere. This spontaneous escape is accompanied by an increase in entropy, which needs no external support.
Laws of Thermodynamics
Thermodynamics details the relation between heat and the internal properties of matter in four laws. The first two laws, namely zeroth and first laws talk about the equilibrium of hot substances in contact and the Constance of the total energy of the universe, respectively.
The first law of thermodynamics does not give any information about the direction of the flow of heat energy.
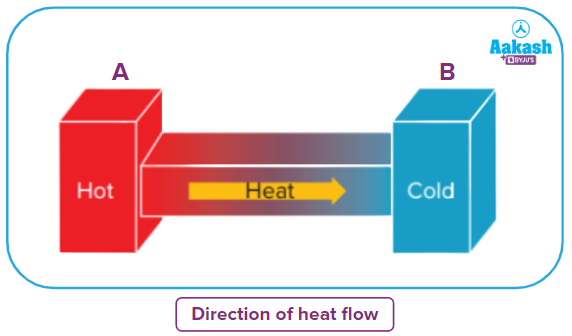
For example, if two systems, A and B, which are capable of exchanging heat, are brought in contact with each other, then the first law of thermodynamics will only tell us that one system loses the energy and the other system gains the same amount of energy.
However, the law fails to tell whether the heat will flow from systems A to B or vice versa or about the direction of spontaneity. That’s where the second law of thermodynamics comes into the picture which states that heat can flow spontaneously from a hot object to a cold object; heat will not flow spontaneously from a cold object to a hot object.
The second law deals with the conditions of the spontaneous and nonspontaneous direction of heat or in general any process.
Entropy and Second Law of Thermodynamics
Entropy is an extensive and state property. When a system changes in its physical or chemical entities, like internal energy and enthalpy, the absolute entropy of the initial and final states need not be known. It is enough if the change in the entropy (ΔS) of the system between the final and initial states is known.
The Universe is the system with the immediate surroundings for practical purposes.
The second law of thermodynamics states that the entropy of the universe increases in a spontaneous process. i.e., ΔSuniv ≥ 0. Mathematically,
(for a spontaneous change)
For a reversible process, i.e., it can proceed in both the direction spontaneously, so this can happen only when there will be no change in the entropy of the universe.
( for a reversible spontaneous change)
For an irreversible process, i.e., it can proceed only in the forward direction spontaneously, so this happens as the change in entropy is greater than zero.
(for an irreversible spontaneous change)
For an isolated system, as there is no interaction between the system and the surroundings, the system itself is considered as the universe. Hence, ΔSsys ≥ 0, for a spontaneous process in an isolated system, the entropy of a system always increases (in case of the irreversible process) or remains constant (in case of the reversible process).
Entropy Change Calculation:
- Entropy changes with temperature ie transfer of heat between the hot and cold body.
Entropy is defined as the change in entropy per unit of temperature,
Example - Cooling of a Hot Body.
Consider hot tea at a temperature of 60°C = 333K
Assume 20J of heat is lost (ΔQ= -20J) by tea and the same amount received (ΔQ= -20J) by the surroundings.
Entropy change of the tea cup system =
Entropy change of the surroundings =
As entropy change of the universe is positive ie increases on the loss of heat from the teacup, cooling will be a spontaneous process.
Cooling of a hot body is hence is a spontaneous process.
Example - Heating of a Hot Body
Consider hot tea at a temperature of 60°C = 333K
Assume 20J of heat is lost (ΔQ= -20J) by the surroundings and the same amount received (ΔQ= 20J) by the hot tea cup.
Entropy change of the tea cup system =
Entropy change of the surroundings =
Since the change of the entropy of the universe is negative, transfer of heat from the cold to the hot body will not take place or it is a non-spontaneous process.
Note:
Universal entropy change hence can be used to predict the spontaneity of a process.
All natural processes accompany an increase in entropy and hence are spontaneous processes.
- Entropy change of the system at constant temperature
For an isothermal expansion or compression of a gas, the entropy change of the system is given as,
Where n is the number of moles of the gas, R is the gas constant. V is the volume of final and initial states. P is the pressure of the initial and final states.
- Entropy change of the system at constant volume and constant pressure
ΔS = nCvlnTfTi: where Cv is the constant volume molar heat capasity
- Entropy change of the system at constant volume, with change in T and P,
ΔS = nCvlnTfTi-nRlnPfPi:
- Entropy change of the system at constant pressure, with change in T and V,
ΔS = nCvlnTfTi+nRlnVfVi:
Recommended Videos: https://www.youtube.com/watch?v=hXS8xNTP5pMEntropy |
Practice Problems
Q1. For an irreversible spontaneous change, the entropy of the universe is:
A) Constant
B) increasing
C) decreasing
D) Zero
Answer: (B)
Solution: ( For an irreversible spontaneous process )
The entropy of the universe tends towards a maximum.
Q2. What will happen to the entropy of the system when an ice cube is placed at room temperature?
a. Increases
b. Decreases
c. Constant
d. Zero
Answer: (A)
Solution: The entropy of a system will increase because initially the molecules of ice are tightly packed but when we place it at room temperature then the molecules will move randomly as the ice melts and gets converted into liquid. As the randomness of the system increases, then entropy will also increase.
Q3. For an irreversible spontaneous process, the change in entropy of the system is 500 J and the change in entropy of the universe is 700 J. The change in entropy of the surroundings would be:
a. 200J
b. 250J
c. 300J
d. 550J
Answer: Option (A)
Solution: Given,
(for an irreversible spontaneous change)
(for an irreversible spontaneous change)
(for an irreversible spontaneous change)
Now,
(for an irreversible spontaneous change)
Putting the given values,
Q4. For a spontaneous reversible process, the change in entropy of the universe will be:
a. Increase
b. Decrease
c. Constant
d. Zero
Answer: Option (D)
For a spontaneous reversible process, there will be no change in the entropy.
(for a reversible spontaneous change)
Q5. Why does an air conditioner cool a room while a refrigerator warms the room?
Solution: Air conditioners can cool your house because part of the unit is outside. That way the air conditioner can pump the heat out of your house and releases it to the outdoors. So just as your refrigerator heats your kitchen while cooling what is inside the refrigerator, air conditioners heat the outdoors while cooling your house. But in both cases, the heat is transferred from a lower temperature to a higher temperature by using electrical work.
Frequently Asked Questions (FAQs)
Q1. What is an example of the second law of thermodynamics in practice?
Solution: One of the real-life examples of the second law of thermodynamics is explained below:
When we put an ice cube in a cup with water at room temperature. The water releases the heat and the ice cube melts. Hence, the entropy of water decreases. The ice cube then absorbs the same amount of heat which was released by water and therefore its entropy increases. The entropy increased in this process is much greater than the entropy decreased.
Therefore we can say that there is a net increase in entropy.
Q2.Why is the second law of thermodynamics necessary?
Solution: The second law of thermodynamics helps in determining whether a reaction is feasible or not and also indicates the direction of heat transfer. It also indicates that heat energy cannot be entirely converted into equivalent work, some amount of heat energy will be rejected as well. The heat energy which is converted into work is known as useful energy.
Q3. Why does entropy increases with the increase in temperature?
Solution: With the increase in temperature, randomness (entropy) increases because the motion of particles increases and the speed of particles also increases so they have more entropy at a lower temperature.
Q4. What are the factors that affect entropy?
Solution: There are several factors that affect the amount of entropy in a system.
- More energy is put into the system at a constant temperature, more will be the randomness of the particles which excites the particles and more will be the entropy and vice versa.
- With the increase in temperature, randomness increases because the motion of particles increases, so they have more entropy at a lower temperature.


