-
Call Now
1800-102-2727
Entropy
Entropy plays a key role in measuring the quantity and quality of energy within a system in thermodynamics. Entropy is a term that originated from the observation that some processes in nature happen on their own while others do not. It facilitates foreseeing processes' spontaneity, measuring energy conversion efficiency, assessing chemical reaction feasibility, and maximising system efficiency. Entropy is crucial to the study of physics, chemistry, engineering, and environmental science. It also serves as a basis for understanding and quantifying the behaviour of systems.
Table of Contents
- What is entropy?
- Entropy and the laws of thermodynamics
- Entropy of fusion
- Entropy of vaporisation
- Entropy and enthalpy
- Practice problems
- FAQs
What is Entropy?
Entropy is a key concept in thermodynamics and statistical mechanics that quantifies the degree of disorder or unpredictability of a system. It counts the total number of small formations or arrangements in accordance with the system's overall configuration.
Entropy, in a nutshell, measures the overall distribution of matter and energy inside a system. A highly structured or organised system has low entropy, whereas a more disordered or random system has high entropy.
Entropy is expressed by the symbol S and is commonly measured in joules per kelvin (J/K) or calories per kelvin (cal/K). The change in entropy, ΔS, is often utilised for studying and predicting the course and randomness of processes and reactions.
The formula for change in entropy is given by

Where
Q = heat transfer in the system
T = absolute temperature
|
Did you know? Negentropy is the opposite of entropy. It means that processes become more in ‘order’. It indicates the opposite of randomness and degree of disorder. |
Entropy and the Laws of Thermodynamics
Entropy is associated with the laws of thermodynamics that constitute the fundamental concepts guiding how energy and systems behave. Entropy and the laws of thermodynamics are related in the following ways:
First Law of Thermodynamics
The first law states that energy is conserved in all processes and systems. It means that energy cannot be created or destroyed and that the total amount of energy in a closed system is constant.
When a solid turns into a liquid and a liquid turns into a gas, entropy increases. The total entropy of an enclosed system is likely to increase or stay constant. However, it never decreases during any kind of energy transfer or change.
Second Law of Thermodynamics
The second law deals with the idea of entropy and how it functions in processes. Thermodynamically, complete heat transfer into work is not possible without some amount of energy being dissipated. The change in total entropy is always positive. The total entropy of a system with the entropy of the surroundings will be greater than zero.
The second law highlights the likelihood of greater disorder or unpredictability and the rise in entropy in natural systems. It suggests that spontaneous activities take place in a way that raises the entropy of the system as a whole and its surroundings.
Third Law of Thermodynamics
According to the third law of thermodynamics, when the temperature approaches absolute zero, the entropy of a system decreases until it reaches a minimum value of zero. Any crystalline solid's entropy decreases as the temperature approaches absolute zero. It's because a crystal at absolute zero has complete order.
The limitation of the statement is that many solids lack zero entropy at absolute zero.
Entropy of Fusion
The entropy of fusion defines the change in entropy that happens when a substance melts down, transitioning from its solid to the liquid state.
The entropy of fusion is generally positive. Liquid molecules are more mobile and free to move than solid molecules, which are more rigidly structured and organised, leading to an increase in entropy.
The entropy of fusion is determined using the enthalpy of fusion and the fusion temperature.
The formula is given as

Where
ΔHfus = enthalpy of fusion
Tf = melting point or fusion temperature
Entropy of Vaporisation
The entropy of vaporisation is used to describe the changes in entropy that take place when any substance is converted into a gaseous state by boiling.
The entropy of vaporisation is generally positive, indicating an increase in entropy. The entropy increases due to the breakdown of the intermolecular links when the liquid is converted to gas.
The entropy of vaporisation can be calculated by multiplying the enthalpy of vaporisation by the boiling temperature. The formula can be given as

Where
ΔHvap = enthalpy of vaporisation
Tb = boiling point
Entropy and Enthalpy
Entropy and enthalpy play an important role in every chemical and physical process. The change in entropy (ΔS) defines the degree of disorder or randomness related to any process. At the same time, the change in enthalpy (ΔH) measures the heat exchange with the surrounding of the process. If ΔH is positive, it indicates that heat has been absorbed by the system. If ΔH is negative, it indicates that heat is released by the system.
The relation between entropy and enthalpy can be given by the formula
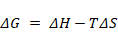
Where
ΔG = change in Gibbs free energy
ΔH = enthalpy change
ΔS = entropy change
T = temperature in Kelvin
Practice Problems
Q 1. What is the SI unit of entropy?
a. KJ/mol
b. J/K
c. J/mol
d. J/C
Ans. b. The entropy is calculated by the change in energy per unit temperature in Kelvin. The SI unit is given as Joules/Kelvin.
Q 2. Which has the highest entropy?
a. Solid
b. Liquid
c. Gas
d. None of the above
Ans. c. The gaseous state has the highest entropy because of the free movement of the molecules.
Q 3. The entropy of any system can not be
a. Increase
b. Zero
c. Constant
d. Decrease
Ans. d. The entropy of any system can increase and remain constant if the process is reversible.
FAQs
Q1. Is it possible to have zero entropy?
Ans. The entropy is zero in an Adiabatic process where there is no exchange of heat and mass transfer possible.
Q2. What is an example of entropy?
Ans. A few examples of entropy include the making of popcorn, boiling water for tea and sugar dissolving.
Q3. What are the factors that affect entropy?
Ans. The factors that affect entropy are changes in temperature, changes in phases, changes in the number of particles and mixing of particles.


