-
Call Now
1800-102-2727
Enantiomers – Stereoisomerism, Definition, Explanation, and Characteristics of Enantiomers, Practice Problems and FAQ
In this article, we will be discussing compounds that are mirror images of each other but are not superimposable on each other. That sentence was puzzling, right? Let's use an illustration to soften it a little.
Place your hand in front of a mirror and observe the image that it creates. Is the image observed on the mirror superimposable on your hand? They aren’t, right? Though your hand and the image observed in the mirror are the same, they are not superimposable.

Similarly, there are compounds in chemistry that are mirror images of each other, but are not superimposable. These compounds are called enantiomers.
Without any further ado, let’s get to know more about enantiomers and their characteristics.
TABLE OF CONTENTS
- Enantiomers
- Enantiomers – Structure
- Enantiomers – Properties
- Practice Problems
- Frequently Asked Questions – FAQ
Enantiomers
Enantiomers are a pair of molecules that are mirror images of each other but cannot be superimposable. Enantiomers are chemically similar to one another in every other way. As a result, a chemical compound's two enantiomers will have identical chemical bonds but radically different three-dimensional structures. When dissolved in a solution, enantiomers differ in the direction in which they rotate the plane of polarised light. The isomer which rotates the plane of polarised light in the clockwise direction is called the dextro-rotatory or (+) - isomer and the one that rotate the plane of polarised light in the anti-clockwise direction is called the levo-rotatory or (-) - isomer. Since they differ in the optical properties, these isomers are also called optical isomers.
When enantiomers are mixed together in equal amounts, the rotation caused by one enantiomer is exactly cancelled by an equal and opposite rotation caused by another enantiomer. Prefix (±) is used to specify the racemic nature of the particular sample.
Example: Equal amounts of d and l forms of lactic acid in a mixture counter-balances rotation of each other.
Let's look at what the enantiomer's absolute configuration for a particular pair.
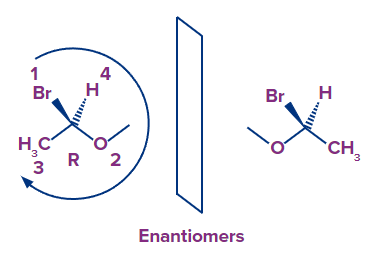
The carbon atom in this molecule is surrounded by four different groups. Therefore, the carbon is chiral. The numbering of groups around the chiral carbon is done according to the priority order given by the CIP (Cahn, Ingold and Prelog) rules.
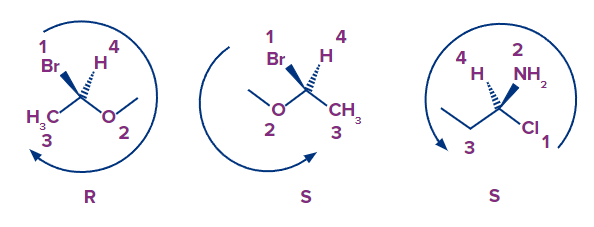
In the isomer given above, we can see that the arrow goes clockwise starting with bromine and moving on to oxygen and ultimately carbon. The absolute configuration is R if the arrow goes clockwise.
In the isomer given above, we can see that the arrow goes counterclockwise starting with bromine and moving on to oxygen and ultimately carbon. The absolute configuration is S if the arrow goes counterclockwise or anti-clockwise.
The arrow's direction is unaffected by the lowest priority. However, it is critical, and it is a must, that when assigning the R and S configurations: The priority with the lowest must face away from the spectator. To put it another way, the lowest priority must be a dashed line to assign the R and S based on the arrow's orientation, as we did earlier:
Another important example of an enantiomer pair is provided below.
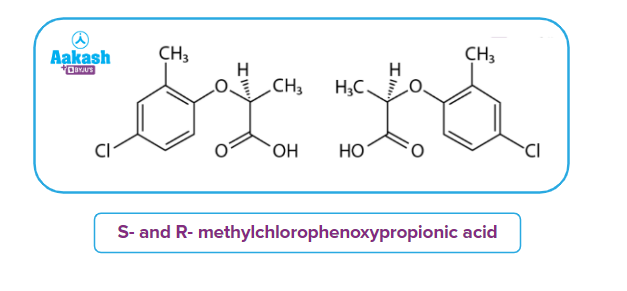
These isomers are known as S- and R-methylchlorophenoxypropionic acid (MCPP, referred to as mecoprop). It contains a combination of both S- and R- isomers. The R- enantiomer of this chemical is known to have herbicidal effects. As a result, this substance is frequently employed as a herbicide.
Contrary to cis and trans isomers, it is significant to note that nearly all pairs of enantiomers tend to share comparable physical characteristics, such as melting point and solubility. But it is well known that they rotate the light in the opposite way.
Chiral Recognition
The distinction between a chiral molecule's two enantiomers is known as chiral recognition. It is challenging to distinguish between the two enantiomers as the physical characteristics that are generally utilised to distinguish enantiomers are generally identical. Physical distinctions can only be seen when interacting with a secondary species that is capable of discrimination. Chirality is the structural underpinning of enantiomeric behaviour. Enantiomers are a pair of molecules that are chemically identical in all other respects but exist in two forms which are the mirror images of each other but are not superimposable on each other. A pair of enantiomers can be identified by the direction in which, when dissolved in solution, they rotate polarised light, either dextro (d or +) or levo (l or -). Hence, they are optical isomers.
Enantiomers – Structure
As seen below, the most frequent source of chirality is a tetrahedrally coordinated atom that is bound to four distinct substituents.
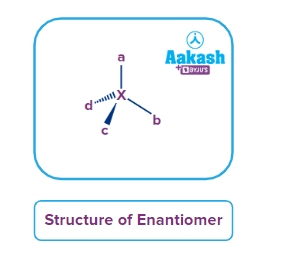
Enantiomers are a stype of stereoisomers which are a pair of non-superimposable mirror images of each other. A molecule is considered to be chiral or to exhibit chirality if it can only exist as a pair of enantiomers. An achiral molecule, on the other hand, can be superimposed on its mirror image. In reality, if there is an atom in a molecule that is tetrahedrally connected to four different groups/atoms, two enantiomers are possible. The four substituents should be different to one another, if not, the molecue will be achiral and can be superimposed on its mirror image. The atom that is joined to four different atoms is referred to as a stereogenic centre, or simply a stereocenter. Since chirality is a property of the molecule as a whole and cannot be localised around one atom or a group of atoms, the phrase "localised around the central atom" is a common but slightly inaccurate alternative term for a stereocenter. Despite being the most prevalent reason for chirality, a stereocenter does not by itself induce a molecule to be chiral.
Enantiomers – Properties
- Physical characteristics including boiling point, NMR spectra, melting point, and infrared absorptions, are often the same for enantiomers.
- While one enantiomer's melting point and other properties will be the same as the other enantiomer, it is vital to understand that a mixture of the two enantiomers may have a different melting point. This is due to the possibility that the interactions between similar enantiomers, which are between two molecules that are both R or both S stereochemistry, may differ from those between opposite enantiomers (R and S).
- The most used chiroptical technique is optical rotation, and it is the only class of physical methods that can discriminate between a compound's two enantiomers.
- The torsional angles' magnitude and sign, which are the only differences between enantiomers, as well as the bond lengths and angles, all affect a molecule's chiroptical characteristics.
Practice Problems
1. Select the correct relationship between d-tartaric acid and meso tartaric acid.
a. Diastereomers
b. Geometrical isomers
c. Enantiomers
d. Identical compound
Answer: A
Solution: D- tartaric acid is the optically active compound in which the -(OH)- group are present in the opposite direction attached to the chiral centre, but in case of meso tartaric acid, the -(OH)- group is present in the same side attached to the chiral centre and is optically inactive in nature. Meso can be obtained by changing the orientation of the valencies present along any one chiral centre present in the molecule. Therefore, D- tartaric acid and meso tartaric acid are diastereomers to each other.
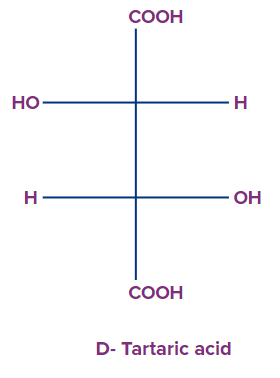
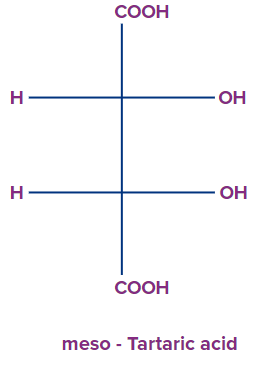
So, option A is the correct answer.
2. How can a pair of enantiomers be separated?
Solution: Since enantiomers will have similar properties except for the rotation of plane-polarised light, it is important that we first convert them to their respective diastereomers that can be easily separated out. Enantiomers are most frequently separated by converting them into a mixture of diastereomers, which will have various physical characteristics, such as melting point, boiling point, solubility, etc. Enantiomers of one substance should be combined with another molecule's single enantiomer to generate diastereomers. Then we can distinguish the diastereomers using conventional techniques (chromatography, recrystallization)
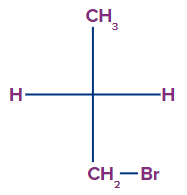
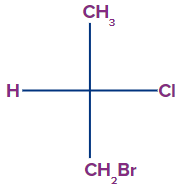
3. Select the correct option for the compound which contains a chiral centre.
Answer: D
Solution: A chiral centre is an atom with four different groups bonded to it in such a way that its mirror image is not superimposable. Nowadays, the term chirality centre is used interchangeably with the chiral centre. In the case of options A, B and C, two valency of the central carbon atom are hydrogen. Therefore, the compounds given in options A, B and C do not contain a chirla centre. In the compound given in option D, the central carbon atom is attached to four different groups/atoms namely, H, Cl, CH3 and CH2Br. Therefore, the compound given in option D contains a chiral centre.
So, option D is the correct answer.
4. Select the correct option for the number of stereogenic centres present in the given molecule.
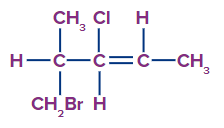
a. 2
b. 1
c. 3
d. 4
Answer: A
Solution: Stereogenic centre includes both the chiral centre as well as geometrical centre. In the given compound, there are two stereogenic centres which include one chiral centre also known as chiral carbon, as all the four valencies along that carbon are different, and one geometrical centre as there are two different groups attached along the carbon that constitutes the rotation restricted system.
So, option A is the correct answer.
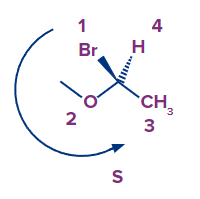
5. The arrangement is ___________ if we move our eyes counterclockwise from the ligand with the highest priority to the ligand with the lowest priority.
a. R-Configuration
b. S-Configuration
c. E-Configuration
d. C-Configuration
Answer: B
Solution: Our eyes go counterclockwise from the ligand with the highest priority to the ligand with the lowest priority in the S-Configuration.
Example:
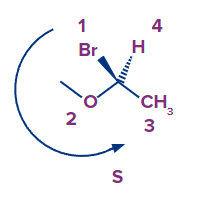
In the isomer given above, we can see that the arrow goes counterclockwise starting with bromine and moving on to oxygen and ultimately carbon. The absolute configuration is S if the arrow goes counterclockwise or anti-clockwise.
So, option B is the correct answer.
Frequently Asked Questions – FAQ
1. What distinguishes enantiomers from diastereomers?
Answer: Stereoisomers that are mirror images of one another and cannot be superimposed on one another are referred to as enantiomers. Contrarily, stereoisomers that contain at least two stereocenters are known as diastereomers. The fact that these isomers are not mirror reflections of one another should be noted.
2. Are enantiomers chiral?
Answer: Enantiomers can be considered as pairs of stereoisomers that are chiral in nature. A chiral molecule implies that its mirror image is non-superimposable, meaning that the mirror image is actually a separate molecule. The central atom in such molecules is called a chiral centre or a stereo centre.
3. Are enantiomers optically active compounds?
Answer: Yes, it is true that all enantiomers are known to be optically active. Optical activity is one of the core properties of enantiomers. This is because enantiomers are isomers which are non-super-imposable mirror images of each other and, therefore, tend to rotate light in opposite directions.
4. What is the meaning of diastereomers?
Answer: Diastereomers are stereoisomers that do not replicate each other’s images, and are not superimposable on each other. The diastereomers can be stereoisomers of two or more stereocenters. Determining whether two molecules are diastereomers is sometimes challenging.