-
Call Now
1800-102-2727
Electrophile: Introduction, Characteristics, Factors Affecting and Examples of Electrophiles, Practice Problems & Frequently Asked Questions(FAQs)
Assume you won more money in the jackpot and became wealthy overnight.
So, if a poor person knocks on your door, you will undoubtedly assist that poor person and readily offer them some money.

In chemistry, we have electron-rich species that are willing to donate electrons and electron-deficient species that are poor and ready to accept electrons. Nucleophiles are electron-rich species, while electrophiles are electron-deficient species.
Table of content:
- Introduction
- Characteristics of electrophiles
- Factors affecting electrophile
- Examples of electrophiles
- Practice problems
- Frequently asked questions(FAQs)
Introduction
The name "electrophile '' is derived from electro which means “electrons” phile," which means "to love." In simple terms, it means "loving electrons." It is a reagent with a low electron density in its valence shell that makes a covalent bond with a molecule, ion, or atom with a high electron density. Hydrogen ions present in acids and methyl-carbocation are examples of electrophilic species. They lack sufficient electrons. A positive charge or a neutral species with vacant orbitals distinguishes an electrophile (not satisfying the octet rule).
Electrons flow from a high-density region to a low-density region, and opposite charges attract one another. This hypothesis explains the attraction of electrons by electron-deficient electrophile atoms, molecules, or ions. According to the definition of the Lewis acid, an electrophile is also referred to as a Lewis acid because it accepts electrons.
Characteristics of electrophiles:
- They are electron-deficient and have a strong attraction to electrons.
- They have either a positive charge or a neutral species.
- They attack places with a lot of electrons, like carbon-carbon double bonds or lone pairs.
- Electrophile favors electrophilic addition and electrophilic substitution reactions.
- Because they accept electrons, they are also known as Lewis acids.
Factors affecting electrophile:
The electrophilic atom is usually a carbon that is bound to an electronegative element, such as oxygen, nitrogen, sulfur, or a halogen, in the vast majority of nucleophilic substitution reactions . Electrophilicity is a basic concept: an electron-poor species is an attractive target for an electron-rich species, such as a nucleophile. However, the influence of steric hindrance on electrophilicity must also be considered. Furthermore, we must consider how the electrophilic carbon's nature, specifically the stability of a potential carbocationic intermediate, influences the SN1 vs SN2 character of a nucleophilic substitution process.
Consider two hypothetical SN2 reactions: one with a methyl carbon electrophile and the other with a tertiary carbon electrophile.
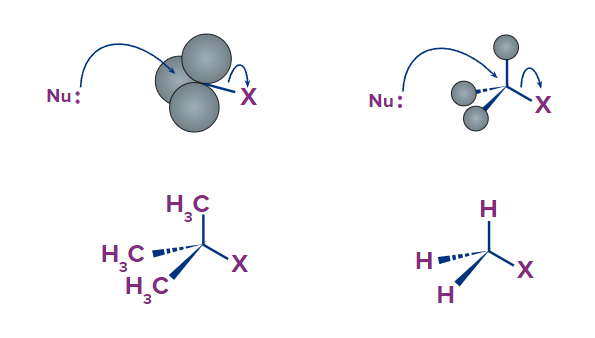
The nucleophile has a rather clean path for backside attack because the three substituents on the methyl carbon electrophile are small hydrogens. The larger methyl groups, on the other hand, prevent a backside attack on the tertiary carbon. The progress of an associative nucleophilic (SN2 ) displacement is hindered once more by steric hindrance, this time produced by bulky groups linked to the electrophile rather than the nucleophile.
The conditions outlined in the above paragraph, on the other hand, do not make a sterically-hindered carbon an ineffective electrophile; they simply make it less likely to be attacked in a concerted SN2 reaction. A two-step, dissociative (SN1) mechanism is more likely to occur in nucleophilic substitution reactions in which the electrophilic carbon is sterically hindered. From a geometric point of view, this makes perfect sense: steric restrictions are most noticeable in an SN2 displacement, when the electrophile being attacked is an sp3 hybridized tetrahedral carbon with its comparatively 'tight' angles of 109.4o. Remember that the nucleophile attacks a sp2 hybridized carbocation intermediate with trigonal planar geometry and 'open' 120oangles in an SN1 mechanism.
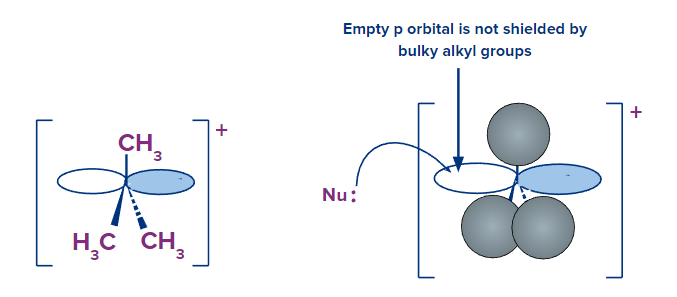
The bulky alkyl groups no longer significantly shielded the electrophilic carbocation's empty p orbital from the approaching nucleophile in this open geometry. A carbocation is a potent electrophile, and the nucleophilic step happens much faster than the first ionization stage.
Examples of electrophiles:
1. Positively Charged Electrophiles: They are electron deficient with positive charge.
Examples: H+, NO2+, CH3+,NO+etc.
Note: All positive charge carrying species are not electrophiles
Examples: NH4+, H3O+etc.
2. Neutral Electrophiles: They are neutral electron deficient molecules with incomplete octet.
Examples:
(a) Lewis acid- BeCl2, AlCl3,BF3, SO3, ZnCl2, FeCl3, SnCl2.
(b) Neutral atoms that have partial positive charge when linked to electronegative atoms like -O, -X, etc.
Examples: Acyl halides (-COCl), Carbonyl compounds (-CO-) and alkyl halides (RX)
(c) Free radicals, nitrenes and carbenes act as electrophiles.
Practice problems:
Q.1. Among the following, which is an electrophile?
(A) H2O
(B) SO3
(C) ROR
(D) NH3
Answer: (B)
Solution: Electrophiles are electron-deficient species that accept a pair of electrons. As a result of its electron deficient center and vacant d-orbitals, SO3 is an electrophile.While H2O, ROR, NH3are nucleophiles.
Q.2. Electrophiles and nucleophiles do not attack alkane molecules since they are:
(A) Non polar
(B) Polar
(C) Volatile
(D) Unstable
Answer: (A)
Solution: Electrophiles (positively charged species) and nucleophiles (negatively charged species) are positively and negatively charged radicals known as reagents that attack the electron rich or electron deficient site of an atom respectively. Alkanes are nonpolar saturated hydrocarbons in which the shared pair of electrons remain in the center of the bond, resulting in a zero dipole moment and the neutral compound the least reactive.
Q.3. Among the following, which is not an electrophile?
(A) NH3
(B) H+
(C) Cl+
(D) NO2+
Answer: (A)
Solution: Ammonia is a Lewis base, and it possesses a lone pair of electrons on nitrogen, which it can donate. As a result, it is a nucleophile rather than an electrophile.The three remaining species have a positive charge, are electron deficient, and act as electrophiles.
Q.4. Why (CH3)4N+ is neither an electrophile, nor a nucleophile?
(A) does not have an electron pair for donation and is unable to attract an electron pair
(B) Because all nitrogen shells are fully occupied, neither has an electron pair available for donation nor can it accommodate an electron.
(C) can act as Lewis acid and base
(D) none of these.
Answer: (B)
Solution: (CH3)4N+ does not have an electron pair available for donation as a nucleophile. The nitrogen valence shell is totally filled and has no scope for extra electrons to act as an electrophile.
Frequently asked questions(FAQs):
1. Why are aldehydes and ketones (carbonyl compounds) good electrophiles?
Answer: Because of the positive charge on the carbonyl carbon, the reactivity of the carbonyl as an electrophile is the most important theme in carbonyl chemistry. Carbonyl reactions almost often include the addition of an electron donor species to the carbonyl carbon.
Lewis acid is also known as an electrophile. Lewis acids attract electrons. Lewis acids have an atom with a positive charge, an atom with a partial positive charge, or an atom without an octet.
2. Explain why alkynes are less reactive to electrophilic addition reactions than alkenes.
Answer: In electrophilic addition processes, alkynes are often less reactive than alkenes because electrons are "held" more tightly in carbon triple bond carbon than in C=C bonds.
In other words, alkenes are more reactive to electrophilic addition reactions than alkynes.
This is because highly strained bridging carbocations develop (3 member ring with a double bond). Also, because the carbon atom in alkynes is more electronegative (has more s character), it strongly holds pi electrons.
3. Why is it that benzene easily undergoes ESR (electrophilic substitution reactions) whereas NSR (nucleophilic substitution reactions) are difficult?
Answer: Benzene is a planar molecule with electrons delocalized above and below the plane of the ring. As a result, it is electron-rich. As a result, it is very attractive to electron-deficient species, such as electrophiles. And it readily undergoes electrophilic substitution reactions.
Nucleophilic attack is difficult due to the presence of an electron cloud of delocalized electrons on the benzene ring. As a result, benzene repels them. And nucleophilic substitutions of benzene are difficult.
4. What is the major difference between electrophilic and nucleophilic attack?
Answer: The main difference between nucleophilic and electrophilic substitution reactions is that the nucleophilic attack involves a nucleophile displacing a leaving group, whereas the electrophilic attack involves an electrophile displacing a leaving group.


