-
Call Now
1800-102-2727
Electrolytic Cells: Electrochemical Cell, Types of Electrochemical Cell, Galvanic Cell, Electrolytic Cell, Types and Applications of Electrolytic cells, Difference Between Galvanic and Electrolytic Cell, Practice Problems, FAQs:
Beena, a student of class 12th went to her friend’s house. She rang the bell and her friend asked her to get inside. But the door handle was too tight, she was not able to open it.

Beena asked her friend why the handle of the door was looking rusty.
She explained her that due to excess exposure to outer environment, the metal of the handle has been corrode. And they are lookin to replace it with a new handle.
According to you, what metal will be best suited for this situation?
Well, we can pick any metal. All we have to do is make this electroplated.
Do you know how electroplating of metal works?
Well, to understand let’s understand the basic concept of electrolytic cell, which a type of electrochemical cell.
Table content
- Electrochemical cell
- Galvanic cell or voltaic cell
- Electrolytic cell
- Difference between Galvanic cell and electrolytic cell
- Practice problems
- Frequently asked questions(FAQs)
Electrochemical cell
An electrochemical cell is a equipment that may be defined as either that create electrical energy from chemical processes taking place inside it or use an external electrical energy source to facilitate chemical reactions inside it. Chemical energy can be converted to electrical energy or vice versa using this device.
Types of Electrochemical cell
Electrochemical cells are classified into two categories:
- Galvanic or voltaic cell
- Electrolytic cell
An electrochemical cell that produces an electricity to flow across a circuit from redox chemical reactions that occur within them is called as electrical cell or galvanic cell or voltaic cell. A conventional 1.5-volt cell, which is used to power numerous electrical items such as TV remotes and clocks, is an example of Galvanic cells or Voltaic cells.
In electrolytic cells, an exlectricity from an external source is utilized to undergo chemical reactions that normally will not take place.
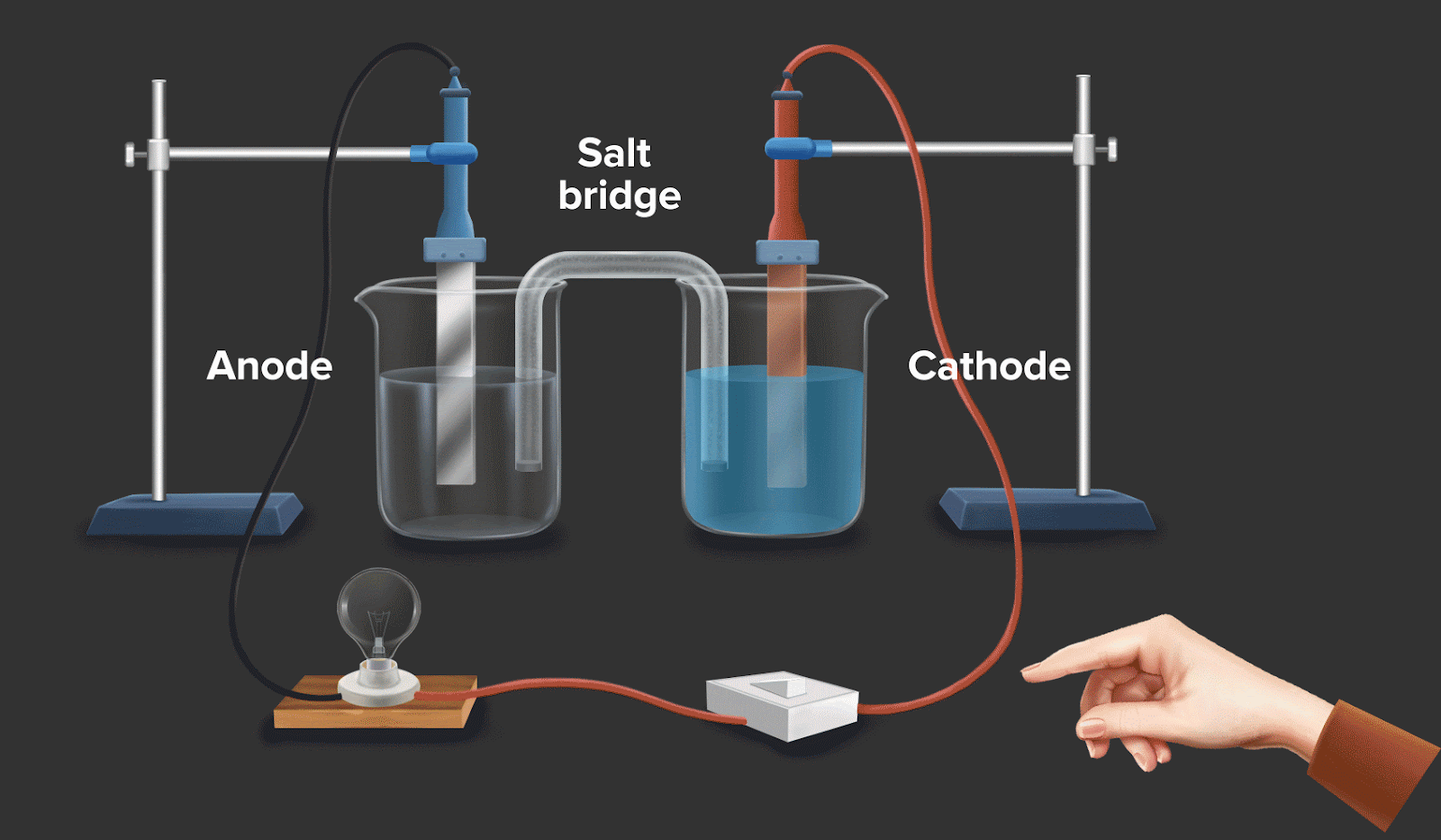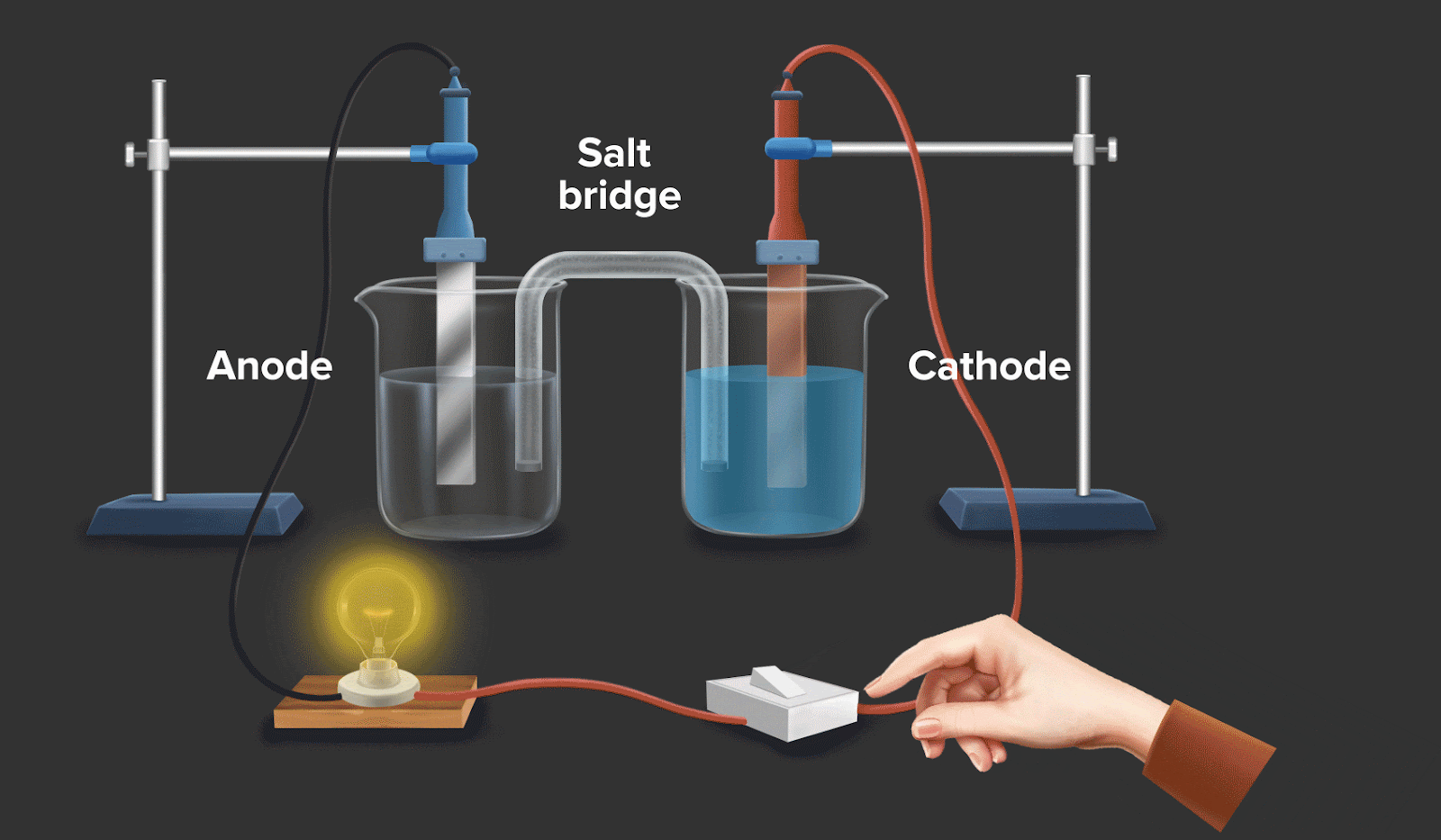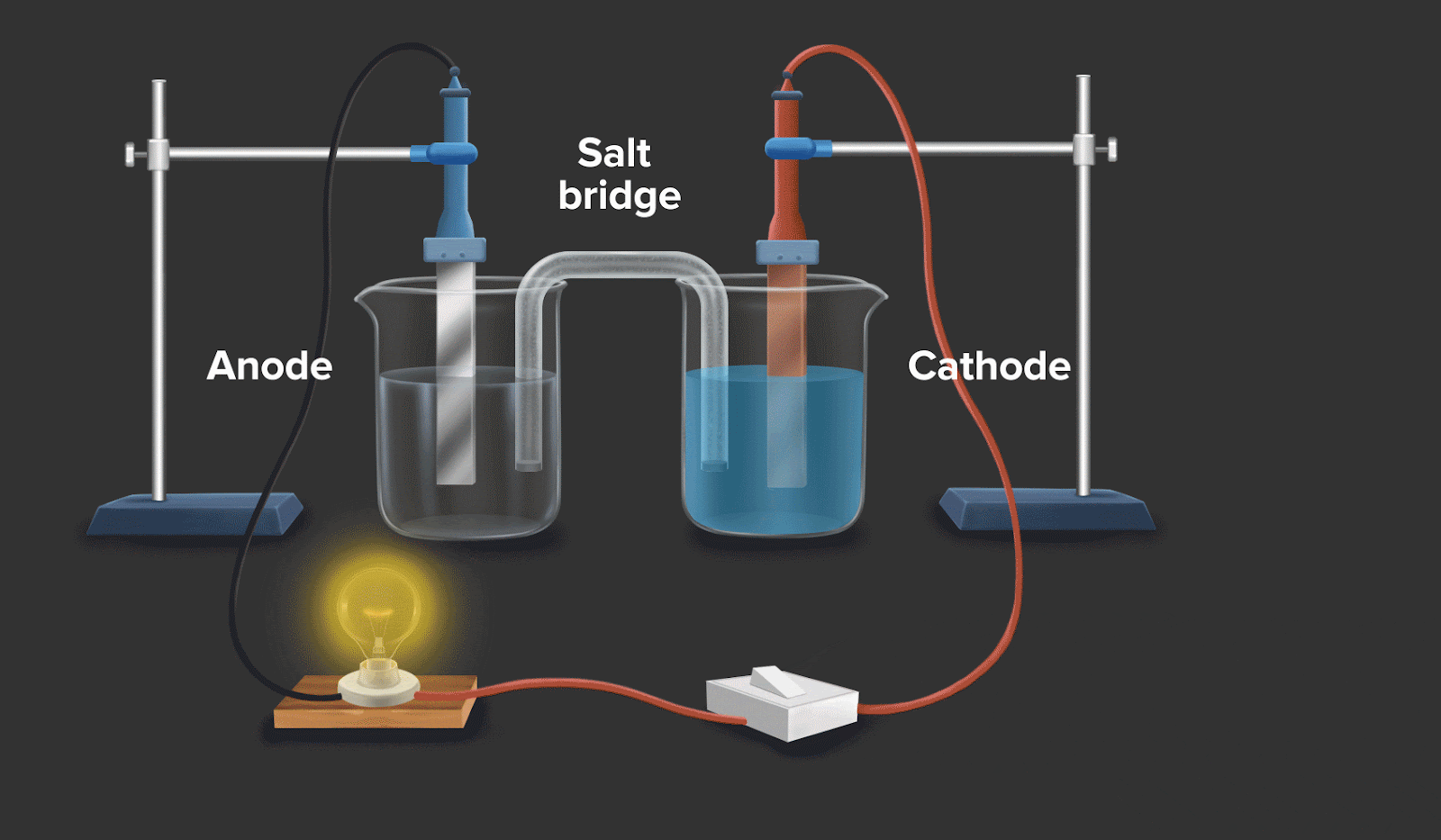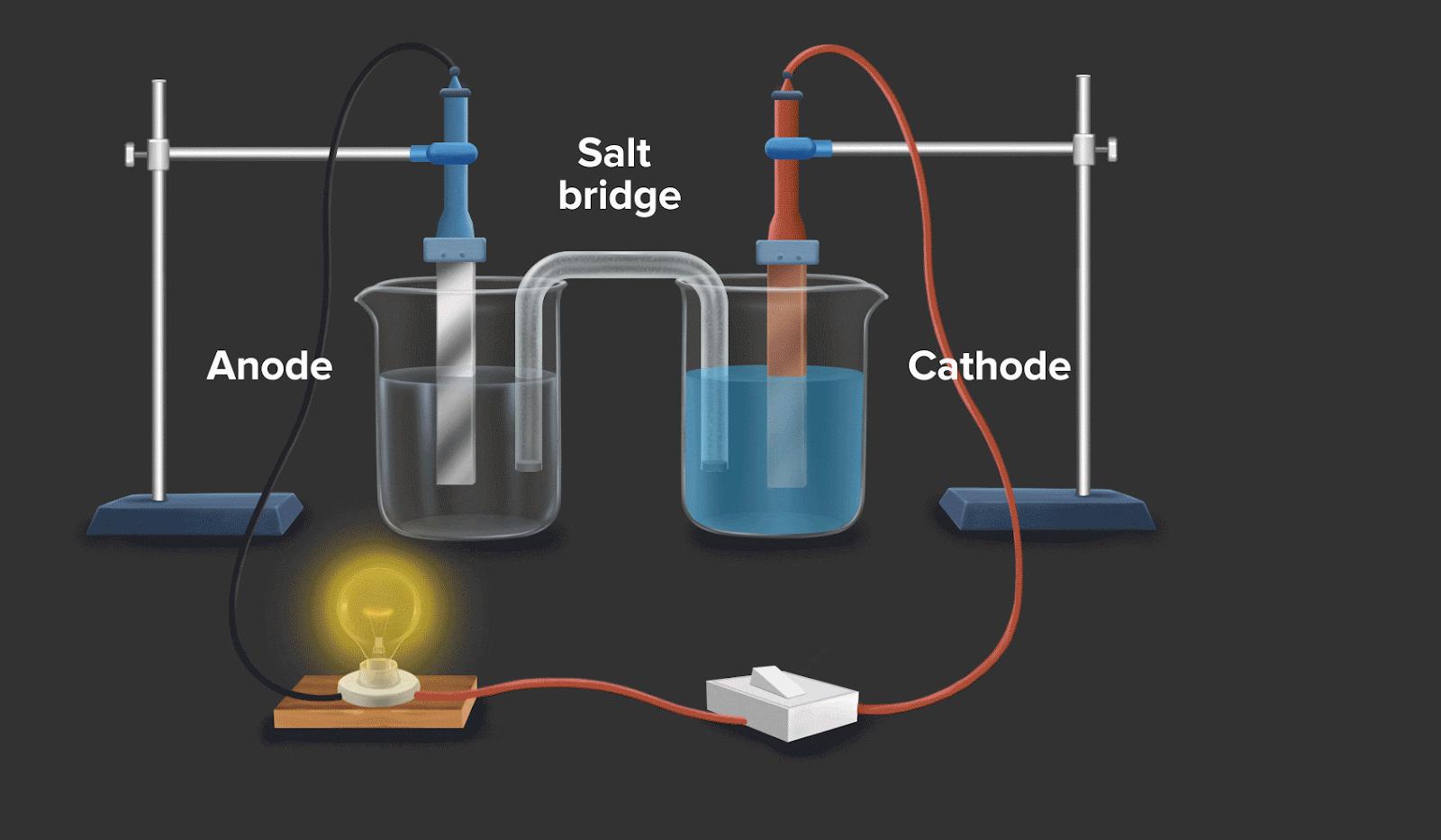
Galvanic cell or voltaic cell
A galvanic cell is an electrochemical cell which uses a spontaneous chemical reaction to produce electrical energy.
Example: The batteries in the automobile work on the same fundamental mechanism.
Consider a circuit as shown in galvanic cell. In the left container, oxidation takes place by convention, and in the right container, reduction takes place. If the overall reaction in this convention is spontaneous, then it can be said that it facilitates the flow of electrons and electric current.
![]()
Electrolytic cell
An electrolytic cell can be defined as an equipment where an electricity is used to make a nonoccuring redox reaction (reverse of a spontaneous reaction) to happen. If we connect a batter between two electrode half cell connected by a salt bridge and apply voltage equal to or slightly higher than the cell EMF, then the reaction takes place in the opposite direction of an galvanic cell, i.e. the total redox reaction which was non-spontaneous will be made feasible. So, in electrolyte cell electrical energy is used for creating chemical energy..
.
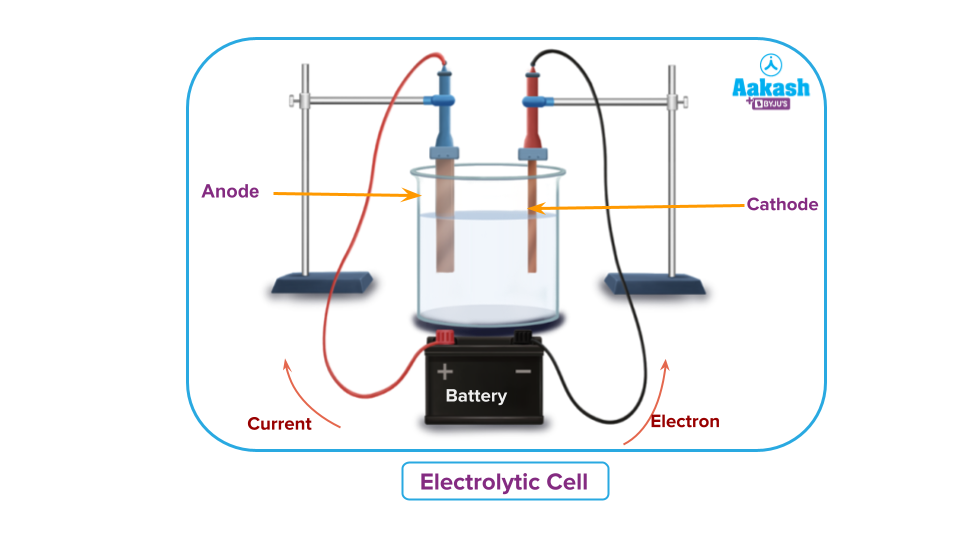
Electrolytic cell can be utilized for multiple uses.
For example, with the use of an electrolytic cell, molten sodium chloride (NaCl) can be electrolyzed into its pure elements of sodium and chlorine gas.
The cell contains molten sodium chloride with inert electrodes. On passing electric current, the cathode becomes rich in electrons and attracts the sodium cations. At the cathode, sodium ion takes up the electron from the cathode , gets neutralized leading to metallic sodium.
The chloride ions are attracted by the anode to which it releases its electron for the flow in the circuit. Two such neutral chlorine atoms combine to form chlorine molecule and escape at the anode..
Anode:
Cathode:
Overall reaction:
Hence, In an electrolytic cell, molten sodium chloride can be electrolyzed to produce metallic sodium and chlorine gas as products.
Types and Application of electrolytic cell
- Electrolysis: Electrolytic cells can be used to decompose compounds electrolytically to their elements.
Example: The creation of oxygen and hydrogen gas from water is the major use of electrolytic cells.
- Electrometallurgy- Is used for recovery of metals from their ore.
Example: They can also be used to recover aluminum from bauxite.
- Electroplating:A process of creating a thin protective layer of a specific metal on the surface of another metal. Chrome, silver, gold and other ornamental coatings are examples.
- Electro refining: Electrolytic cells are used in obtaining very pure metals from impure metals.
Many non-ferrous metals like copper are purified by electrolytic method. It should be mentioned that electrolytic cells are virtually usually used in the industrial manufacture of high-purity copper, high-purity zinc, and high-purity aluminum.
- Electrowinning: Electrolysis is used for the recovery of costly and precious metals from contaminants in the presence of leaching solutions of the metal salt.
Zinc metal recovery is one of the example of this electrowinning methodology.
- Electrosynthesis:
- Electrolysis are being used to manufacture chemicals that cannot be done otherwise.
Persuphate, chlor-alkali process are very voluminous chemicals prepared only by electrolysis.
Difference between Galvanic cell and electrolytic cell
|
Galvanic Cell |
Electrolytic cell |
|
|
|
|
|
|
|
|
|
|
Practice problems
Q1. Which of the following is not an electrochemical cell?
- Galvanic cell
- Voltaic cell
- Electrolytic cell
- None of the above
Answer: (D)
Solution: electrochemical cell is a device for the interconversion of chemical energy & electrical energy. Galvanic cell or voltaic cell, and electrolytic cells are different types of electrochemical cell.
Q2. Spontaneous reaction can be observed in which of the following cell?
- Galvanic cell
- Electrolytic cell
- Both A & B
- None of the above
Answer: (A)
Solution: The redox reactions that occur in galvanic cells are entirely spontaneous.
Q3. Choose the correct statement
- The redox reactions that occur electrolytic cells are entirely spontaneous.
- In electrolytic cell, The species that undergoes oxidation is where the electrons come from.
- In electrolytic cell, chemical energy is converted to electrical energy.
- A positively charged anode and a negatively charged cathode are used in electrolytic cell.
Answer: (D)
Solution: In electrolytic cell redox reactions are non- spontaneous. In these cells, Electrons are emitted from a source outside of the body (such as a battery), and here electrical energy is converted to chemical energy.
Q4. How do electrolytes conduct electricity?
- Due to the mobility of electrons from anode to cathode.
- Due to the mobility of electrons from cathode to anode.
- Both A & B
- None of the above
Answer: (B)
Solution: A substance develops the ability to conduct electricity when it splits into ions in solution or melt. Examples of electrolytes are Sodium chloride (NaCl) and Potassium chloride (KCl). They conduct electricity when mobility of electrons occurs from cathode to anode.
Hence, option (B) is correct.
Frequently asked questions(FAQs)
Q1. Can we recharge an galvanic cell?
Answer: It depends on the redox reactions taking place. Any additional reactions and loss of substances prevent the recharge of galvanic cells using an eternal source. Only rechargeable galvanic cells are capable of getting recharge and not all.
Q2. The mobile device that we use are electrolytic cells or electrochemical cells?
Answer: It is a galvanic cell function while the battery is in operation in the device (using the redox energy to produce electricity). It is an electrolytic cell function while the battery is charging (using outside electricity to reverse the completed redox reaction)
Q3. Is electrolytic cell endothermic or exothermic?
Answer: The chemical process in an electrolytic cell is endothermic. Because the reaction is not spontaneous, it necessitates the use of a power source. An electrochemical cell is a device that stores energy. When the cell is allowed to run in a galvanic mode, this energy can be released.
Q.4 What types of charges are associated with electrolytic cells' electrodes?
Answer: The cathode and anode in electrolytic cells have opposing electric charges. The cathode which is negative receives positively charged ions, while the anode with positive charge receives negatively charged ions.


