-
Call Now
1800-102-2727
Electrolytes- Electrolytes, Their Types, Degree of Ionization, Practice Problems and FAQs
Assume you are in your final chemistry practicals. The invigilator posed a question to you, "look, young lad, There are two beakers A and B, each containing a fine white powder. One of them contains salt, while the other contains sugar. Can you tell which beaker contains salt without tasting it?"
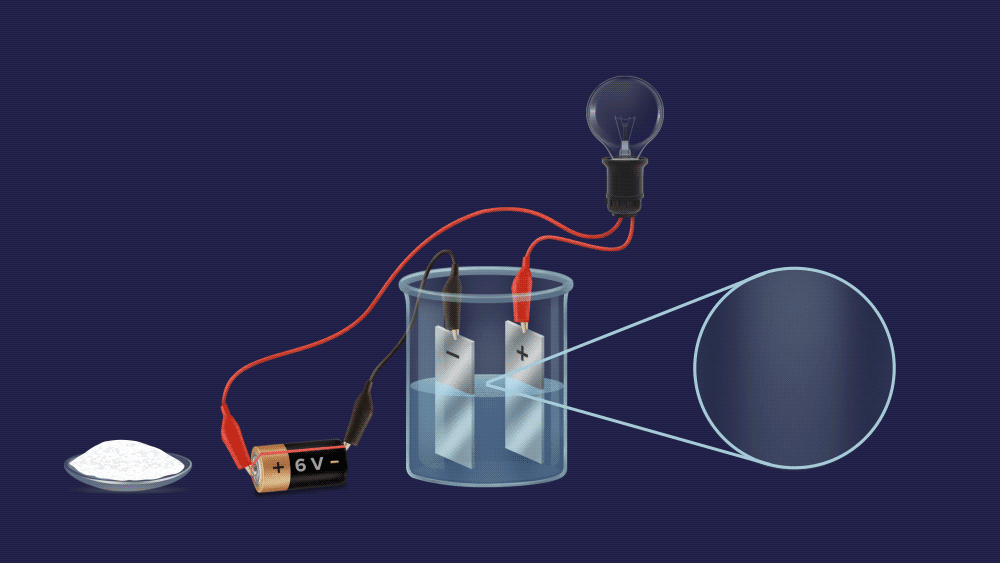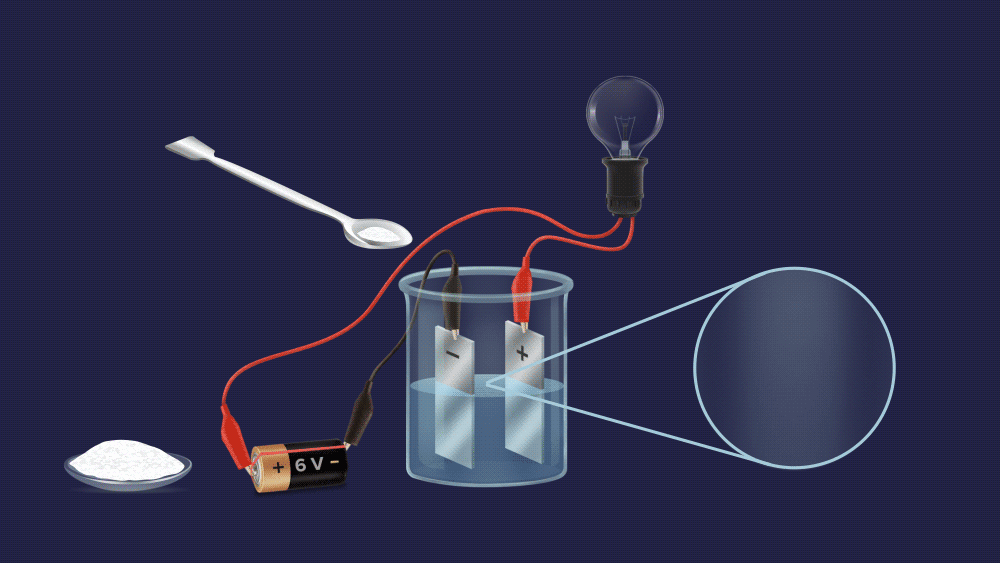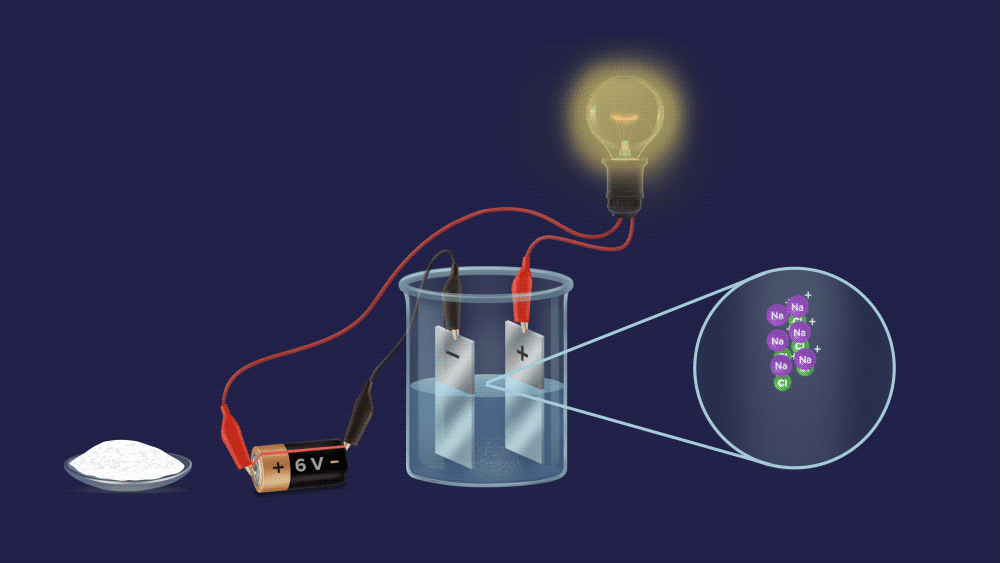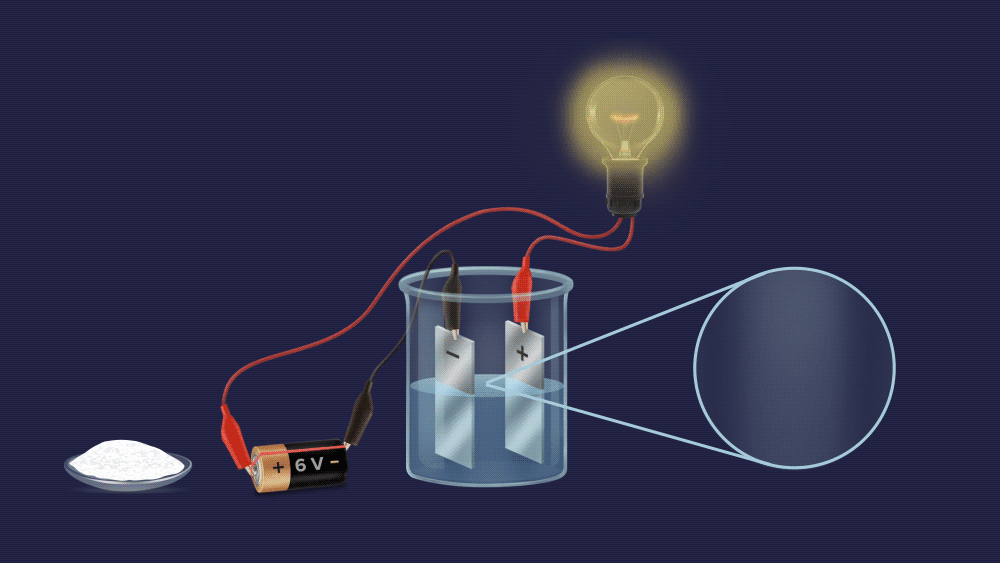
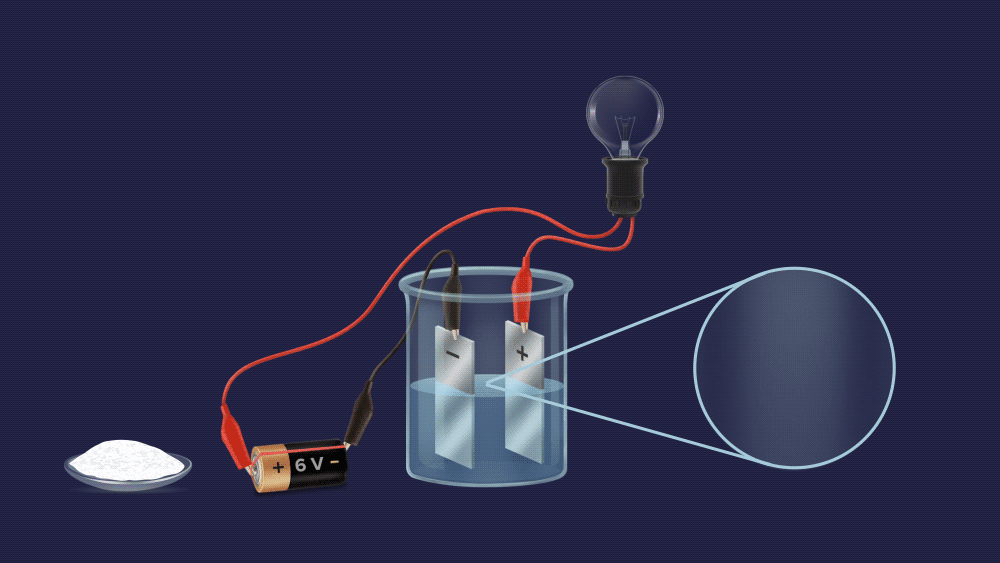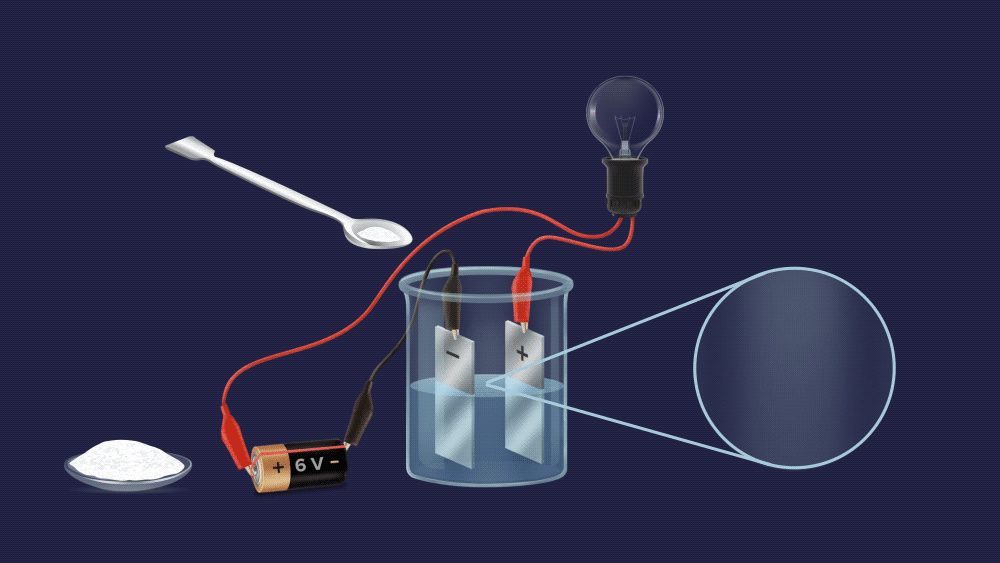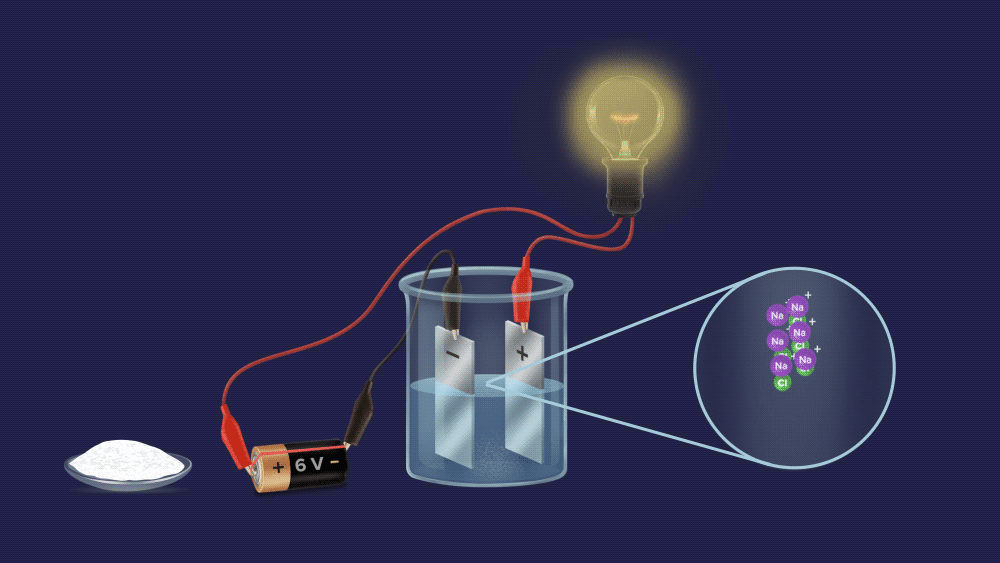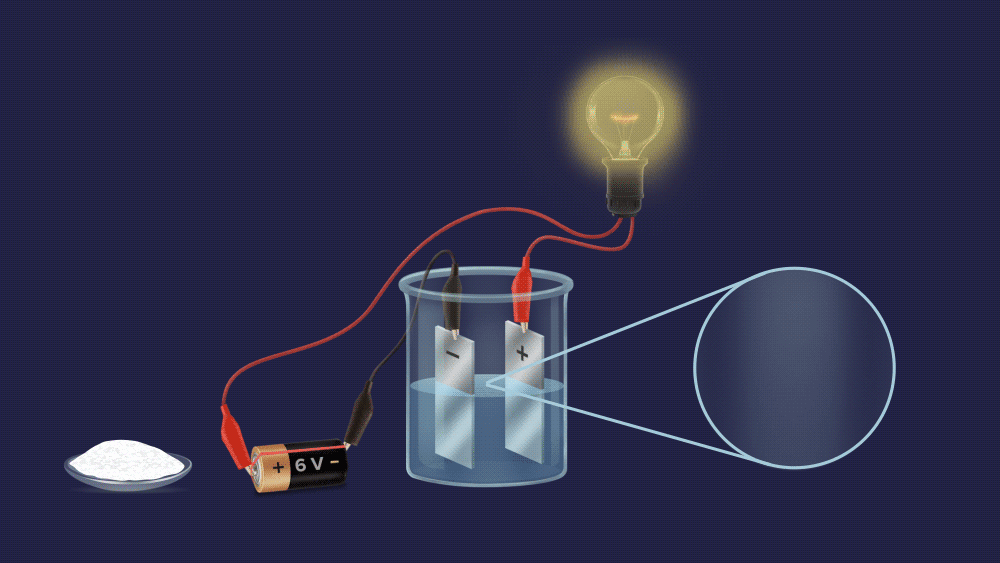
We can easily tell them apart, as you can see. To begin with, make an aqueous solution of both powders and construct an electrochemical circuit by connecting a battery, electrodes, and a bulb.
In case of a salt solution, the bulb glows brightly.
The bulb does not light up in case of a sugar solution.
As a result, you can quickly identify the salt solution. But, if you're wondering why this happens, read on.

![]()
![]()
Table of Contents
- Electrolytes
- Degree of ionization
- Types of Electrolytes
- Strong Electrolytes
- Weak Electrolyte
- Ionization of Weak Electrolytes
- Practice Problems
- Frequently Asked Questions
Electrolytes
An electrolyte is a substance that can conduct electricity either in a molten state or dissolved in a polar solvent like water.
It is made up of oppositely charged cations and anions. It dissociates into ions and conducts electricity when dissolved in water. .
Most of the electrolytes are ionic compounds, which means they are made up of oppositely charged ions. In water, the electrostatically bound ions dissociate and are hydrated by water molecules. They can conduct electrical current not only in an aqueous state but also in a molten state, due to the charged ions. As an example, consider acids, bases, and salts.
In an aqueous state, some polar covalent molecules can react with water to form cations and anions and conduct electricity.
Example:
Degree of ionization
The degree of dissociation is defined as the fraction of the total number of molecules of the electrolyte dissolved that is dissociated into ions at equilibrium. It is represented by α.
The probability of interaction between dissociated ions decreasing with dilution. Thus, as the rate of backward reaction decreases, the rate of dissociation increases, forming more ions, i.e., as dilution increases, so does the extent of ionization.
At infinite dilution, the degree of dissociation of an electrolyte is assumed to be almost unity (α ≈ 1).
Types of Electrolytes
Electrolytes are classified into two types. 1) Strong electrolytes and 2) Weak electrolytes.
The following are the differences between strong and weak electrolytes:
Weak electrolytes are those ionic compounds that partially ionise, whereas strong electrolytes are are those ionic compounds that completely ionise
Strong Electrolytes
- Strong electrolytes are those electrolytes that are almost completely ionized or dissociated (i.e., ~100 per cent ionization) upon dissolution.
- Because strong electrolytes are completely ionized, the degree of ionization α is approximately equal to 1.
- Examples include common salt NaCl, HCl, and NaOH.
- Because HCl completely ionises into H+ and Cl-, the reaction is represented by a single-headed arrow:
Weak Electrolyte
Weak electrolytes are electrolytes that are only partially ionized upon dissolution.
- Because weak electrolytes are only partially ionized, the degree of ionization, α << 1,
- Acetic acid (CH3COOH), carbonic acid (H2CO3), and ammonium hydroxide are some examples
(NH4OH).
- Because NH4OH is a weak electrolyte, it partially ionizes into ammonium ions () and hydroxides (OH-) ions. As a result, an equilibrium exists between NH4OH and its constituent ions. Hence, their ionization is represented by putting double half arrows (⇌) and demonstrated by the following reaction:
- It can thus be concluded that ionic equilibrium exists in weak electrolytes.
|
Strong Electrolytes |
Weak Electrolytes |
|
They dissociate almost completely in their aqueous solutions. |
They dissociate partially in their aqueous solutions. |
|
Strong acids (HCl, H2SO4etc.) |
Weak acids (CH3COOH, H2C2O4etc.) |
|
Strong bases (NaOH, KOH etc.) |
Weak bases (Ca(OH)2, C5H5N etc.) |
|
Example: All soluble salts (NaCl, CH3COONa etc.) |
Example: All sparingly soluble salts (AgCl, BaSO4 etc.) |
Ionization of Weak Electrolytes
In general, the ionization of a weak electrolyte, AB, is represented as follows:
Such an equilibrium is known as an ionic equilibrium between the ions and the undissociated
electrolyte. Applying the law of chemical equilibrium to the given equilibrium, the Equilibrium constant in terms of concentration,
Strong and weak electrolytes exhibit different variations in ionic concentration with dilution and hence their molar conductivity..
The molar conductivity gradually rises with dilution for strong electrolytes. A straight line with a y-intercept equal to the value of the limiting molar conductivity ( maximum molar conductivity) appears when the molar conductivity and (where c is the concentration) are plotted.
But the graph between molar conductivity and for weak electrolytes is not a straight line. On dilution, week electrolyte dissociates more to give more ions and hence more molar conductance than strong electrolytes.
Practice Problems
Q1. One thing unites all electrolyte solutions:
A. always contain acids or bases
B. react with other solutions
C. contain molecules
D. conduct electricity
Solution: Electrolytes are substances that conduct electricity, according to the definition. Option (D) is the best option for distinguishing electrolytes.
Q2. Which of the following substances conduct electricity?
A. Urea
B. Naphthalene
C. Sugar
D. Table salt
Solution: Urea, naphthalene and sugar are organic compounds that do not dissociate to generate ions. When we dissolve it in water, it does not become ionized and generate its constituent's ion. As a result, they are examples of non-electrolyte.
Table salt has a chemical composition NaCl and dissociates into ions. Hence, it is an electrolyte.
Hence, the correct option is (D).
Q3. One experimental setup is created to determine the electrolytic nature of the solute. In the beaker, a salt solution is added.
Observation: When a salt solution is added to a beaker, the bulb glows.
![]()
What should the outcome of this experiment be?
A. Ions are not produced as a result of dissociation. As a result, salt is a nonelectrolyte.
B. Ions are produced through dissociation. As a result, salt is an electrolyte.
C. Ions are produced through dissociation. As a result, salt is a nonelectrolyte.
D. Ions are not produced through dissociation. As a result, salt is an electrolyte.
Solution: A electrolyte is a compound that conducts an electric current in an aqueous solution or molten form. These compounds produce ions when they dissolve in water.
If the bulb light up, this substance can conduct electricity. As a result, salt is an electrolyte. Hence, the correct option is (B).
Q4. Which of the following salt can be classified as electrolytes?
A. Copper Sulfide
B. Ferric oxide
C. Anthracene
D. Calcium Chloride
Solution: Copper sulfide and anthracene are insoluble in water, hence do not exist in ions and are non-electrolytes. Ferric oxide is the formula of rust which is also insoluble in water and does not dissociate in it. Hence it is also non-electrolyte. Calcium chloride is highly soluble in water and dissociates to give ions. Hence, it is an electrolyte.
Hence, the correct option is (D).
Frequently Asked Questions
Q1. Is salt a type of electrolyte?
Answer: Placing salt in a liquid (such as water) frequently contributes to the formation of an electrolyte solution. Smelted salts can also be used as electrolytes. For example, molten sodium chloride is a liquid that can conduct electricity.
Q2. Is water a type of electrolyte?
Answer: Water with a very low ion content cannot conduct electricity. Because the solution is a strong electrical conductor, when a solvent dissociates to form ions in water, it is referred to as an electrolyte.
Q3. Why is the study of electrolytes important?
Answer: Electrolytes are electrically charged particles composed of substances found in bodily fluids (ions). These ions store electrical energy that is needed for a variety of tasks, such as muscle contractions and nerve impulse transmission.
Q4. Is aqueous carbon dioxide electrically conductive?
Answer: When CO2 gas is dissolved in water, it forms a solution that contains hydrogen ions, carbonate, and hydrogen carbonate ions:
Because it contains ions, the resulting solution will conduct electricity. Because CO2 does not dissolve into ions, it is important to remember that it is not an electrolyte. Electrolytes are substances that dissolve into their constituent ions.


