-
Call Now
1800-102-2727
Electrolysis of Sodium Chloride
Most bulky chemicals with industrial use, like sodium hydroxide and chlorine, are produced by a crucial process called sodium chloride electrolysis. Sodium chloride can be electrolysed in both aqueous and molten forms. Additionally, other salts are present during electrolysis to facilitate the redox reactions.

Image: Components of electrolysis
Table of Contents
- What is Electrolysis?
- Electrolysis of Molten Sodium Chloride
- Electrolysis of Aqueous Sodium Chloride
- Importance of Electrolysis of Sodium Chloride
- Practice Problems
- Frequently Asked Questions
What is Electrolysis?
Electrolysis is a technique that utilises an electric current to execute a chemical reaction. It involves the dissociation of compounds into constituent elements or ions. The compounds are dissolved in an electrolyte, a liquid that facilitates the flow of electric current.

Image: Electrolysis
Electrolysis of Molten Sodium Chloride
Electrolysis involves the movement of ions to an electrode. Electrolysis cannot be performed in a solid state because ions cannot travel. Sodium chloride dissociates into sodium and chloride ions during high-temperature melting, allowing electrolysis to form sodium atoms and chlorine gas.
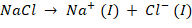
At Cathode, the reaction occurs will be 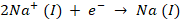
At Anode, oxidation takes place such that 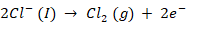
Net reaction takes place 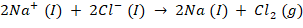
The down process involves certain points that include
- Sodium chloride would melt at a very high temperature of around 801 degrees Celsius.
- The melting point decreases to 580 degrees Celsius by adding anhydride chloride in the ratio of

- Iron cathode and graphite anode are utilised in the electrolysis procedure, where iron gause is utilised to hamper the mixing of products, i.e., chlorine and sodium.
- Chlorine gas and the metal sodium is the product of the electrolysis of molten sodium chloride.
Electrolysis of Aqueous Sodium Chloride
Sodium chloride, when present in an aqueous solution, is dissociated into sodium and chloride ions. The dissociation is easier in the aqueous solution. However, water can also experience reduction and oxidation reactions at several potentials. Therefore, the oxidised or reduced substance may contain sodium and chloride ions and involve the water molecule.
The cathode and anode can have two competing reactions, where a reduction reaction occurs at the cathode at pH 7. Both sodium ions and water can be transformed into hydrogen gas and sodium metal, respectively.
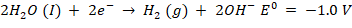
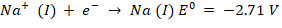
At pH 7, an oxidation reaction occurs at the anode. Water can be transformed into oxygen or chlorine molecules by oxidising chloride ions, i.e.,
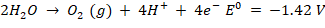
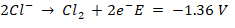
Consequently, the final product of electrolysis of aqueous sodium chloride might range from
- At the cathode, sodium metal or hydrogen gas,
- Chlorine or oxygen gas is formed at the anode.
The sodium chloride aqueous solution concentration influences electrolysed sodium hydroxide side product, which is generated when sodium reacts with water. Moreover, the reaction can occur in two forms, either very diluted aqueous sodium chloride solution or at high concentrations.

Image: Electrolysis of aqueous sodium chloride
Importance of Electrolysis of Sodium Chloride
There are numerous significant importance of sodium chloride electrolysis which are as follows.
- Aids in the production of chlorine gas, which is widely utilised by industries.
- Production of sodium hydroxide (NaOH) takes place, which is useful in the pulp, paper, textile and detergent industries.
- Generation of hydrogen gas, which is used as a fuel source in numerous industries.
- Helps in water treatment.
- Helpful in research of electrolytic cells involving studying distinct behaviour of ions and redox reactions.
- Aids in pharmaceutical and chemical industries for drug formulation.
- Utilised in alkaline batteries.
- It can be used in the production of bleach.
Practice Problems
Q1. Electrolysis of sodium chloride aids in the production of
a. Chlorine
b. Sodium hydroxide
c. Hydrogen
d. All of the above
Ans: d. All of the above
Explanation: Electrolysis of sodium chloride aids in the production of chemicals of industrial utility like sodium hydroxide, hydrogen gas and chlorine.
Q2. In electrolysis of aqueous sodium chloride, chlorine or oxygen is formed at
a. Anode
b. Cathode
c. Gel
d. None of the above
Ans: a. Anode
Explanation: In electrolysis of aqueous sodium chloride, oxygen gas or chlorine is framed at the anode.
Q3. Aqueous sodium chloride electrolysis results in the formation of ____ at the cathode
a. Sodium metal
b. Hydrogen gas
c. Either a or b
d. None of the above
Ans: c. Either a or b.
Explanation: In the electrolysis of aqueous sodium chloride, hydrogen gas or sodium metal is generated at the cathode.
Frequently Asked Questions
Q1. What is a redox reaction?
Answer: Redox reaction, also called oxidation-reduction reaction, involves the transmission of electrons from one molecule or species to another. The species which gain electrons are known as reduced species, and the one that loses become oxidised.
Q2. State two common gels used to perform electrolysis.
Answer: The most widely used gels used for electrolysis are polyacrylamide and agarose gels.
Q3. What is the rule of the cathode?
Answer: According to the rule of the cathode, the production of metals at the cathode depends on their position in the metal reactivity series. The metals that are less reactive than hydrogen are produced at the cathode.


