-
Call Now
1800-102-2727
Dual Nature of Matter - Definition, Examples, Practice Problems & FAQs
Why can the flow of air rotate a paddle wheel? Why flow of air can’t cast shadow but light can cast shadow on an object? Can you guess at microscopic or macroscopic level any object having both the characteristics?
Flow of air can rotate a paddle wheel because it has mechanical properties or particle nature and light can cast shadow of any object because light has wave properties. Light has a dual character in both wave and particle nature, with the help of their wave properties we can explain phenomena like interference, diffraction and particle nature of light perfectly explains blackbody, photoelectric process.
Now, what if I say that matter also has a dual nature. Would you agree with this statement?

In the same way, Louis de Broglie proposed a hypothesis that a particle shows a wave nature. So, let’s see how it was proved that particles also have the wave characteristics.
Table of Contents
- de Broglie’s Hypothesis
- Davisson Germer Experiment
- Other Mathematical form of De-Broglie Wavelength
- Practice Problems
- Frequently Asked Questions-FAQs
de Broglie’s Hypothesis
According to de Broglie, the matter also has a wave character and he proved this theoretically by giving a mathematical relation between the wavelength and the momentum of the particle.
He used Planck's equation and Einstein's mass-energy connection to calculate the wavelength of this relation which is called de Broglie’s equation.
According to the Plank’s equation;
E = h𝜈....(i)
𝜈 is the frequency of the photon
λ is the wavelength of the photon
c is the speed of the light
h is the Planck’s constant
According to the Einstein’s equation;
m is the mass of the particle
c is the speed of the light
Equating equation (i) and (ii), we get;
Similarly for particles (matter),
where pis the momentum of the particle.
These relationships allow us to link the wavelength of a wave to the mass and velocity of particles. The de Broglie connection connects the two representations of the electron, which may be described as a particle or a wave. The circular stationary orbits predicted by Bohr are no longer accepted for electrons. Electrons, according to de Broglie, travel in waves.
Consider an electron that is orbiting a nucleus in a circle of radius r. Evidently, the radius of the circular orbit must be an integral multiple of wavelength lambda if the wave is to remain consistently in phase.
Where v is the velocity of electrons
Thus, the angular momentum can be calculated;
This is considered as Bohr’s postulate.
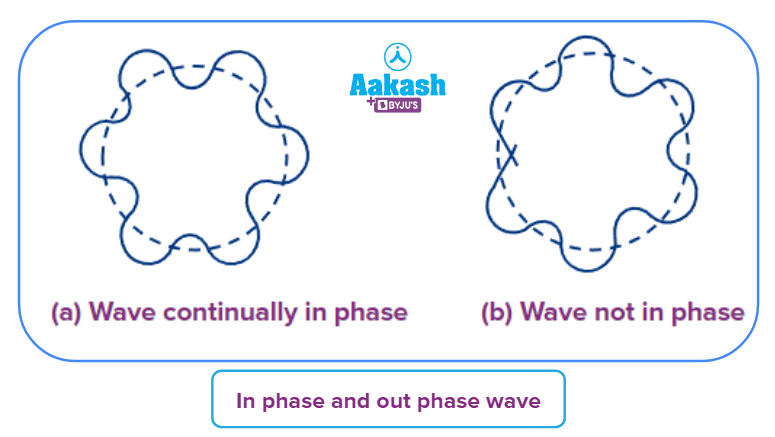
Evidently, the wave will no longer remain in phase if the circumference is more or lower than the value nh/2 specified in the above equation.
Some aspects of electrons may be explained in terms of wave nature, while others can be explained in terms of particle nature, just like electromagnetic radiation.
According to the de-Broglie theory, matter possesses both wave and particle qualities. However, it was only a theoretical notion until Davisson and Germer carried out a renowned experiment that verified the concept, earning Davisson the Nobel Prize for his efforts.
Davisson Germer Experiment
Clinton J. Davisson and Lester H. Germer studied the diffraction of an electron beam being dispersed by nickel crystals in order to show the wave nature of electrons. The experiment also confirmed the early theoretical de-Broglie theory that matter, in addition to its particle character, has a wave component.
Experimental apparatus of Davisson Germer experiment:
The graphic below is the arrangement used by Clinton J. Davisson and Lester H. Germer to demonstrate the wave nature of electrons. The setup includes an electron cannon with a tungsten filament that is heated by a low tension voltage source. The accelerator in the cannon is in charge of accelerating thermally excited electrons from the filament by providing the appropriate potential difference using a high tension voltage source. As a result, the electrons' velocity may be changed using the accelerator. A fine stream of electrons is impinged on the nickel crystal positioned in its path. The device is housed in a vacuum chamber to prevent electrons from colliding with atoms in the path of an electron beam. A mobile collector with a rotating galvanometer is put in the direction of the diffracted electron beams, as can be seen in the image. Because the nickel target is moveable, the collector is essentially a Faraday cup electron detector that measures the number of electrons diffracted after impacting it at various angles.
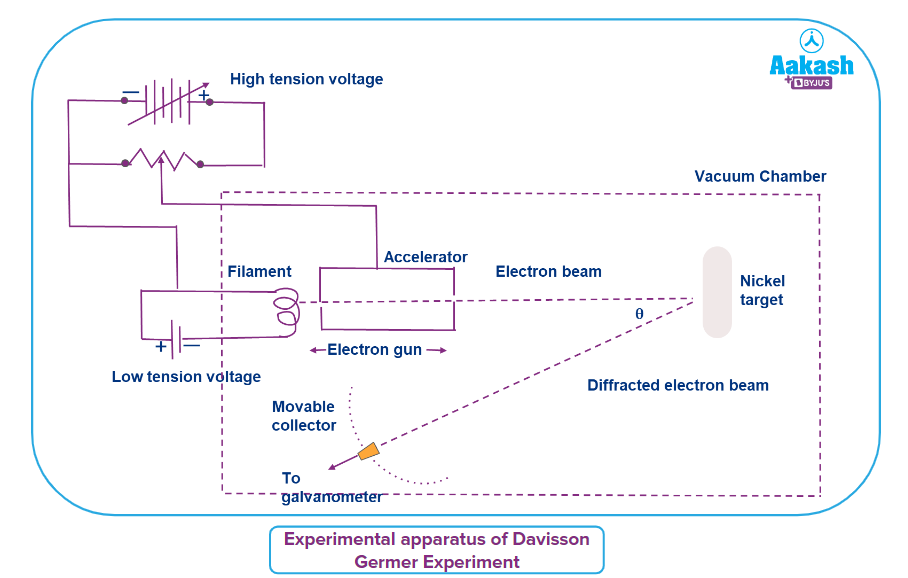
Working of Davisson Germer experiment:
The thermionic emission of electrons occurs when the filament is heated; the electrons are then accelerated using an accelerator to form an electron beam. The electron beam is then impacted on a nickel target as usual. The nickel target's atoms scatter the incoming electrons in diverse ways. By rotating the mobile collector in a circle, the intensity of the dispersed electron beam in a particular direction may be determined. The moveable collector is connected to a galvanometer, which measures the photocurrent and so determines the electron beam intensity.
Other Mathematical form of De-Broglie’s Wavelength
We can write
Multiplying''m' on both the sides, we get;
Substituting this expression of p in (iii), we get;
When a charge 'q' is accelerated by a potential difference of 'V' volts from rest, the charge's kinetic energy equals q × V .
If an electron is accelerated through a potential difference of 'V’ volt from rest then:
On putting the values of h,me and qein the above equation.
h = 6.626 × 10-34 Js
me= 9.1 × 10-31 kg
qe=1.6 × 10-19 C
We get,
Practice Problems
Q1. What will be the mass associated with a ball having de Broglie wavelength 6.626 10-37m moving with velocity 100 m sec-1?
- 10 g
- 100 g
- 1000 g
- 10000 g
Answer: (D)
Solution: Let the mass of ball = x g
We can see, order of the wavelength of the macroscopic particle is extremely small, therefore de Broglie wavelength calculation for the macroscopic particles has no physical significance.
Q2. The mass of an electron is 9.1 10-31kg.The wavelength associated with it is 2377 nm. The kinetic energy of the electron is:
- 3.5 10-25J
- 3.5 10-26J
- 3.5 10-27J
- 4.5 10-26J
Answer: (B)
K.E =3.5 10-26 J
Q3. The approximate ratio of de Broglie’s wavelength of neutron and electron moving with the same speed is:
- 1:4
- 1:1839
- 1:1
- 1:2
Answer: (B)
Solution:
Mass of electron = 11837 mass of 1 proton
mass of electron = 11839 mass of 1 neutron
According to the question, both electron and neutron have the same velocity.
( for electron)
( for neutron)
Q 4. Which of the following has the longest de Broglie’s wavelength?
(All have the equal velocity)
- Hydrogen gas molecule
- Electron
- Proton
- Nitrogen gas molecule
Answer: (B)
We know,
For species which have the equal velocity, wavelength is inversely proportional to their mass. An electron has the least mass in all the given options. So, it has the longest de Broglie’s wavelength.
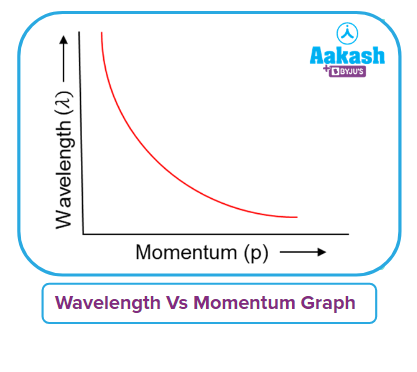
Q5. Which of the following graph types best shows the change in de Broglie’s wavelength as a function of momentum?
- Rectangular hyperbola
- Parabolic
- Linear
- Can’t be predicted
Answer: (A)
Solution: The matter wave of a particle has a de Broglie wavelength that is inversely proportional to its momentum.
Due to this, the lighter particle will have a longer wavelength and less momentum. The electron has the longest wavelength and is the lightest particle. The curve between the de Broglie’s wavelength and the momentum will be a rectangular hyperbola.
Frequently Asked Questions-FAQs
Q. What is the de-Broglie hypothesis's flaw?
Answer: Because the order of the wavelength of macroscopic particles is so tiny that de Broglie wavelength computation macroscopic particles has no physical meaning.
Q. What do you understand about the dual nature of electrons?
Answer: Electrons may be thought of as both a particle and a wave at the same time. Bigger the particle size, the smaller the wavelength of the particle. The dual nature of matter is proved by de Broglie using the famous de Broglie equation.
Q. Which experiment conclusively proved the de-Broglie hypothesis?
Answer: The Davisson and Germer experiment revealed the electrons' wave character, validating de Broglie's previous notion. When electrons are dispersed from crystals with correctly spaced atoms, they display diffraction.
Q. de-Broglie was awarded the nobel prize in physics for which achievement?
Answer: de Broglie awarded nobel prize in physics (1929) for discovery of wave nature of electrons. He proved that even particles have the wave nature. Bigger the particle size, the smaller the wavelength of the particle. He proved the dual nature of matter by giving the famous de Broglie equation.


