-
Call Now
1800-102-2727
Dry Cell - Definition, Working Principle and Types of Dry Cell
In our daily life, we use many electronic devices that use dry cells, such as wall clocks, watches, battery toys, remote control of TV, cameras etc. It is a type of simple electric battery with electrochemical cells. One of its important working principles is to convert chemical energy into electrical energy. Thus, these cells are used to generate electricity.
Table of Contents:
- What is a Dry Cell?
- Types Of Dry Cells
- Working Principle of Primary Cell
- Working Principle of Secondary Cell
- Advantages of Dry Cells
- Applications of Dry Cells
- Practice Problems On Dry Cell
- Frequently Asked Questions
What is a Dry Cell?
The dry cell is a simple electrochemical cell used as an electricity-producing source. It converts chemical energy into electrical energy to run electronic devices. This cell was developed by a French engineer named Georges Leclanche in 1866, after which dry cells are named Leclanche cells.
A certain number of cells combine together to form a battery. It consists of one or more electrochemical cells, which convert chemical energy into electrical energy. The lead-acid battery is an advanced dry cell. However, earlier, it was heavy and easily destructible. With the same principle, an advanced version of the dry cells is introduced and comes in different voltages and sizes.
In 1881, Carl Gassner of Mainz invented the modified form of the Leclanche battery, which is the commercial form of a zinc-carbon cell. It has many applications, such as flashlights, calculators, radios, toys, etc.
Types Of Dry Cells
Dry cells are classified into two types based on their nature. They are primary cells and secondary cells.
Primary Cells
- In primary cells, the chemical reactions are irreversible.
- Once the electrochemical reactions consume chemical reagents, the cells are dead and fail to produce electricity. They can’t be recharged again.
- Primary cells are smaller, and they are easy to transport.
- The fixed output voltage is 1.5 V
Secondary Cells
- In secondary cells, the chemical reactions are reversible.
- These cells can be reused and recharged multiple times.
- Once the electrochemical reactions consume chemical reagents, the cells can be recharged by using battery charges to generate electricity.
- Secondary cells can also be transported; however, they are larger than primary cells.
- The output voltage is not fixed, and it depends upon the load attached to them.
Working Principle Of Primary Cell
1. Zinc Carbon Cell
The chemical reaction in primary cells takes place only once. In a zinc-carbon cell, the zinc performs as the anode. The carbon or graphite rod acts as the cathode. The rod touches the brass cap on the top. The zinc is covered on the sides but not at the bottom. It is surrounded by low moisture electrolytes such as ammonium chloride (NH4Cl) paste, manganese dioxide (MnO2) and zinc chloride (ZnCl2). The reaction in the anode and cathode are as follows:
At Anode:
Oxidation refers to the loss of electrons. At the anode, oxidation takes place. As a result of oxidation, the zinc corrodes over time.
Zn(s) → Zn2+ + 2e-
At Cathode:
Reduction refers to the process of gaining electrons. At the cathode, reduction takes place. The manganese oxidation state is reduced from the +4 to +3 oxidation state. Ammonia that is released is trapped by Zn2+ ions to form [Zn (NH3)]+2. It is in aqueous form and soluble in nature.
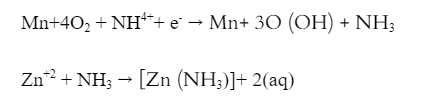
2. Mercury Cell
In mercury cells, the cathode is HgO, and the anode is the zinc metal. The reaction takes place as follows:
At Anode:

3. Alkaline Battery
Like zinc-carbon cells, the alkaline battery has the same half-cell reactions.

Working Principle of Secondary Cell
1. Nickel-cadmium Cell
The Nickel-cadmium cell is also called the NiCd cell. It comprises cadmium and nickel, in which cadmium acts as an anode and nickel plate acts as a cathode. Sodium hydroxide or potassium hydroxide acts as an electrolyte. During disposal, cadmium releases harmful substances to the environment. So NiCd cells are not used now.
2. Lithium-ion Cell
Lithium-ion batteries are popularly used on many electronic devices such as cell phones, laptops, iPods, etc. Lightweight carbon and lithium are used to make the electrodes of these cells. During disposal, they are less harmful to the environment than NiCd cells.
Advantages of Dry Cells
Some of the advantages of Dry cells are listed below:
- Dry cells are simple and easy to use in electronic devices.
- They are cheap and easily available.
- They have high energy density and are easy to create in compact sizes.
- Dry cells are easy to carry and safe during transportation.
- They have very low hazardous effects on the environment.
Applications of Dry Cells
- Primary cells are used in small electronic gadgets that can be transported, such as clocks, watches, calculators, etc.
- Lithium batteries are used in devices like smoke detectors and cameras as they only require minimum output.
- Lead storage cells are used in automobiles and inverters.
Practice Problems On Dry Cell
Q1. Which of the following is used as the cathode in a Zinc carbon cell?
a. Zinc
b. Carbon rod
c. Ammonium chloride
d. None of the above
Ans: b. Carbon rod
In a zinc-carbon cell, the zinc performs as the anode. The carbon or graphite rod acts as the cathode. The rod touches the brass cap on the top.
Q2. Which of the following is incorrect about secondary cells?
a. Irreversible
b. Rechargeable cells
c. Reusable cells
d. None of the above
Ans: a. Irreversible
In secondary cells, the chemical reactions are reversible. These cells can be reused and recharged multiple times.
Q3. Which of the following cells is used in inverters?
a. Lithium batteries
b. Lead storage cells
c. Nickel-cadmium Cell
d. Zinc Carbon Cell
Ans: b. Lead storage cells
Lead storage cells are used in automobiles and inverters.
Frequently Asked Questions
Q1. What is meant by an electrode?
Ans: An electrode is a conductor that is used to create electrical contact with non-metallic solids or liquids or any substance of the circuit.
Q2. What is meant by cations?
Ans: Cations are referred to the ions that have a net positive charge and can move towards the negative electrode.
Q3. What is meant by anions?
Ans: Anions are referred to the ions that have a net negative charge and can move towards the positive electrode.


