-
Call Now
1800-102-2727
Disaccharides – Structure, Types, Categories, Importance, Practice Problems and FAQ
We do a variety of things every day, like studying, going to classes, travelling, shopping, working out, and more.
Have you ever wondered how we manage to do so much in a single day?
Think about what would occur if we went a day without eating. Will you have the same amount of energy to complete each of these tasks?
Definitely not. So, exactly what is it about the meal that gives us energy and enables us to work all day? The answer is biomolecules, and one of these is a molecule called a carbohydrate. As the primary source of energy for your body, carbohydrates should make up a significant portion of your daily diet. Consuming enough carbohydrates will keep you healthy and give you the energy you need for day-to-day life.
Disaccharides are a subcategory of carbohydrates that, like other carbohydrate types, are crucial to our daily lives.
Let's examine what it is and why it is important to us.
TABLE OF CONTENTS
- Disaccharides
- Sucrose
- Lactose
- Maltose
- Different Categories of Disaccharides
- Food containing Disaccharides
- Importance of Disaccharides
- Practice Problems
- Frequently Asked Questions – FAQ
Disaccharides
Condensation of two molecules of either the same or different monosaccharides produces disaccharides. Looking at the classification of carbohydrates, we can clearly see that Disaccharides are simple carbohydrates.
When two monosaccharides (simple sugars) are connected by glycosidic linkage, a disaccharide (also known as a double sugar) is created. Disaccharides, like monosaccharides, are water soluble. Sucrose, lactose, and maltose are three common examples.
Disaccharides are carbohydrates that can produce two monosaccharide molecules that can be the same or different when they are hydrolyzed by acids or enzymes. The two monosaccharides are created by the oxide linkage, which forms when the water molecule is destroyed. When an oxygen atom connects two monosaccharide units, a glycosidic connection happens.
Sucrose
Sucrose is the most common disaccharide with the molecular formula . It is a white crystalline solid that is soluble in water. Sucrose is dextrorotatory and on hydrolysis with dilute acids, yields an equimolar mixture of D-(+)-glucose and D-(–)-fructose.
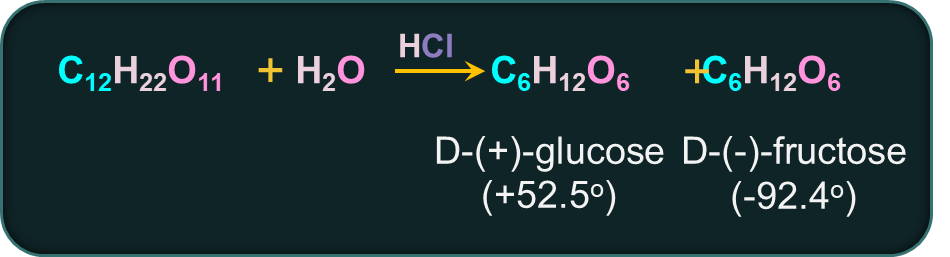
Condensation reaction takes place between C-1 of 𝛼-D-glucose and C-2 of β-D-fructose to form sucrose.
Sucrose is a non-reducing sugar since the glycosidic bond formation involves both the reducing groups of glucose and fructose. Due to the lack of a hemiacetal group, Sucrose is not a reducing sugar,
i.e., it does not reduce Fehling’s or Tollens’ reagent.
Since the laevorotation of fructose (–92.4°) is more than the dextrorotation of glucose (+52.5°), the mixture is laevorotatory. A mixture of glucose and fructose (1:1) is known as invert sugar.
Lactose
Lactose is a disaccharide present in the milk of humans, cows, and almost all other mammals. Because this disaccharide is found in milk, it is commonly referred to as milk sugar. It has the molecular formula . It is hydrolysed by dilute acids or by the enzyme lactase to give an equimolar mixture of D-(+)-glucose and D-(+)-galactose.
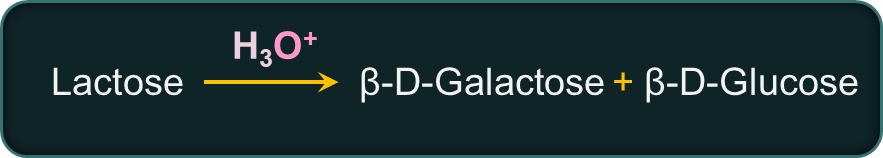
In lactose, glycosidic linkage is between C1 of β-D-Galactose and C4 of β-D-Glucose.
Lactose is a reducing sugar, i.e., it reduces Fehling’s or Tollens’ reagent. This is also a sugar that helps you lose weight.

Maltose
Maltose is produced by the action of malt (which contains the enzyme diastase) on starch. It has the molecular formula .

Maltose produces two molecules of D-(+)-glucose when it is hydrolyzed by maltase or diluted acids.
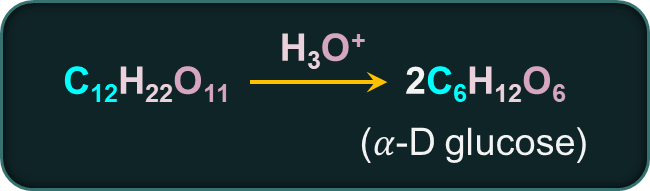
In maltose, glycosidic linkage is between C1 of 𝛼-D-Glucose and C4 of 𝛼-D-glucose.
Maltose is a reducing sugar because at least one hemiacetal group (of the two glucose molecules) is present, which causes it to reduce Fehling's or Tollens' reagent.

Different Categories of Disaccharides
There are a few additional kinds of Disaccharides that aren't as well-known, such as
Trehalose
Two molecules of glucose combine to form the sugar trehalose. Tremalose and mycose are other names for it. It is produced by some plants, fungi, bacteria, and invertebrate animals as a source of energy and to survive lack of water and cold temperatures.
Lactulose
One molecule each of galactose and fructose combine to form the sugar lactulose. Lactulose, a non-absorbable sugar, is used to treat hepatic encephalopathy and constipation. It is administered orally for constipation and intrarectally for hepatic encephalopathy.
Cellobiose
Cellobiose, like trehalose, also consists of two glucose molecules, but are organised in a different pattern to that of trehalose. Bacteriology, a type of chemical analysis, can be used to see these.
Chitobiose
It is made up of two connected glucosamine molecules. It can be found in bacteria, insect exoskeletons, as well as fish, octopus, and squid.
Food containing Disaccharides
Sucrose, maltose, and lactose are the most common disaccharides found in regular meals. Other, lesser-known forms of disaccharides, on the other hand, aren't readily present in our diets and must be manufactured artificially.
Foods containing sucrose
Sucrose, sometimes known as table sugar, is one of the most prevalent disaccharides and can be found in a variety of foods, including:
- Cakes
- Ice cream
- Chocolate
- Some salad dressings
- Breakfast cereals
- Watermelon
- Oranges
- Peaches
- Sweet potato
- Carrots
- Mangos
Foods containing maltose
If you look at the ingredients of the meals you eat and find anything with the word "malt" in it, there's a strong possibility maltose is one of the ingredients. Maltose can be found in a variety of foods, including:
- Sweet potatoes
- Instant coffee
- Honey
- White flour
- Glucose syrup
- Beer
Foods containing lactose
Lactose is present in different levels in most dairy meals. Because hard cheese has less lactose than other dairy products like milk, some lactose intolerant persons can eat modest amounts of it. Lactose is found in minor levels in other foods, such as bread. Lactose can be found in the following foods:
- Milk
- Bread
- Pancakes
- Cookies
- Cakes
- Breakfast cereals
- Soups
- Salad dressings
- Chips
- Coffee creamers and powdered milks
Importance of Disaccharides
Disaccharides, like any other carbohydrate, serve as an energy source for the body. Our systems break down disaccharides into simple sugars (monosaccharides) for absorption in the small intestine when we ingest meals that contain them.
Practice Problems
Q1. Which of the following is a disaccharide?
A. Glucose
B. Fructose
C. Maltose
D. Galactose
Answer: C
Solution: Glucose, fructose and galactose are monosaccharides, while maltose is a disaccharide. When it is hydrolysed with dilute acids or by the enzyme maltase, maltose yields two molecules of D-(+)-glucose.
So, option C is the correct answer.
Q2. Lactose on hydrolysis yields which two monosaccharides?
A. Glucose and Fructose
B. Glucose and Ribose
C. Glucose and Sucrose
D. Glucose and Galactose
Answer: D
Solution: Lactose is hydrolysed by dilute acids or by the enzyme lactase to give an equimolar mixture of D-(+)-glucose and D-(+)-galactose.

So, option D is the correct answer.
Q3. The number of monosaccharides that are needed to be condensed into disaccharides is
A. 1
B. 2
C. 3
D. 4
Answer: B
Solution: When two monosaccharides (simple sugars) are connected by glycosidic linkage, a disaccharide (also known as a double sugar) is created. The condensation of two molecules
of either the same or different monosaccharides produces disaccharides.
So, option B is the correct answer.
Q4. There is a linkage between which of the following carbons in maltose?
A. C1, C2
B. C2, C3
C. C1, C4
D. C2, C4
Answer: C
Solution: Maltose is produced by the action of malt (which contains the enzyme diastase) on starch. It has the molecular formula . When it is hydrolysed with dilute acids or by the enzyme maltase, maltose yields two molecules of D-(+)-glucose. In maltose, glycosidic linkage is between C1 of 𝛼-D-Glucose and C4 of 𝛼-D-glucose.

So, option C is the correct answer.
Frequently Asked Questions – FAQ
Q1. What is the general formula of carbohydrates?
Answer: Carbohydrates have the generic formula . Because carbohydrates (or sugars) contain the general formula , they were once thought to be "hydrates of carbon."
Q2. What is the most frequent disaccharide's molecular formula?
Answer: Disaccharides are carbohydrates that break down into two identical or distinct carbs when hydrolyzed. is their usual formula.
Q3. Why are disaccharides soluble in water?
Answer: In contrast to polysaccharides, which have a complicated structure, monosaccharides and disaccharides are soluble in water because they contain a number of free-OH groups that are polar and strongly attracted to water molecules.
Q4. Why are disaccharides vital in human nutrition?
Answer: Disaccharides are made up of three primary compounds: sucrose, lactose, and maltose, and they are a key source of energy in the diet. Sucrose, sometimes known as table sugar, is the most widely distributed disaccharide and the most widely used naturally occurring sweetener.


