-
Call Now
1800-102-2727
Dioxygen – Preparation, Physical and Chemical Properties, Practice Problems and FAQ
How old is the universe, do you know?
Our galaxy, the Milky Way, is about 13.61 billion years old and the universe is roughly 13.8 billion years old.
What staggering figures!
What about learning more about something we care about? What about the sun's and the earth's ages?
The age of the sun is roughly 4.603 billion years, whereas the age of the earth as we know it is roughly 4.543 billion years.
Would you like to learn more about us? How about when the first human was born?
Only two million years ago, the first person on earth began to walk and breathe.
Talking of breathing, do you know how old the oxygen present on earth is?
As a result of cyanobacterial photosynthesis, oxygen was created in the oceans as early as 3 billion years ago. We, humans, have only been on the planet for 2 million years, yet we have been breathing oxygen that was created for around 3 million years. Since oxygen is what we breathe to live, it is essential to understand its properties.
On this concept page, we will learn about dioxygen and its properties in detail.
TABLE OF CONTENTS
- Dioxygen
- Dioxygen – Preparation
- Dioxygen – Physical Properties
- Dioxygen – Chemical Properties
- Dioxygen – Anomalous Properties
- Practice Problems
- Frequently Asked Questions – FAQ
Dioxygen
One of the most prevalent forms of elemental oxygen is dioxygen, which is also referred to as dioxygen gas, molecular gas, or dioxygen. Some important characteristics of dioxygen are as follows.
- Two double bonds between two oxygen atoms serve as the Lewis structure of dioxygen.
- As a result, the molecule, O2, is linear and exhibits strong reactivity because it has lone pairs of electrons on both oxygen atoms.
- The bond between the two atoms in the dioxygen molecule is covalent in nature.
- Dioxygen is the most common allotrope of oxygen and plays an important role in a sustainable life.
Dioxygen – Preparation
Laboratory Preparation Methods
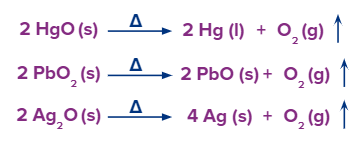
Method – 1: Metal oxides that are lower in the electrochemical series and have low electrode potentials, such as mercury, silver, and lead, break down to release dioxygen gas.
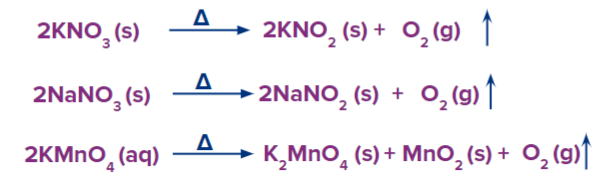
Method – 2: Dioxygen is released during the thermal breakdown of salts that are high in dioxygen, such as nitrates, chlorates, and permanganates.
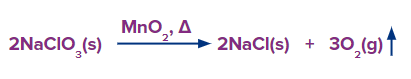
Method – 3: At 420 K, MnO2 acts as a catalyst to accelerate the breakdown of sodium and potassium chlorates. This leads to again the formation of dioxygen.
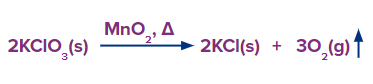
Method – 4: Using electrochemistry, by the Electrolysis of acidified water, dioxygen can also be produced along with hydrogen gas.
Method – 5: Dioxygen is created by the thermal breakdown of peroxides like hydrogen peroxide.
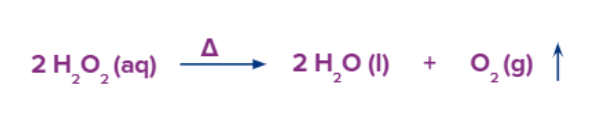
Barium peroxide has been extensively utilised for this purpose commercially.

Industrial Preparation Methods
Method – 1: Adsorption is a technique that involves passing air through zeolite molecular sieves, which adsorb nitrogen to produce 90 – 93% pure dioxygen.
Method – 2: By using the process of fractional distillation of liquefied air, water vapour first condenses and is filtered out using absorbent media. At -190oC , nitrogen liquefies and is drawn out of the atmosphere. When the temperature is lower, dioxygen liquefies at-185oC and is separated.
Dioxygen – Physical Properties
- The diatomic gas dioxygen is colourless, tasteless, odourless, and paramagnetic by nature.
- dioxygen gas is denser than air.
- The boiling point of dioxygen is -183˚C.
- The melting point of dioxygen is -218.4˚C.
- Although dioxygen gas in its pure state is not flammable, it promotes combustion.
Dioxygen – Chemical Properties
- With both metals and non-metals, dioxygen gas can react to form oxides, peroxides, or superoxides.
- Dioxygen can combine with almost every element to form oxides (O2-). It exists as an oxide in an oxidation state of -2.
- In addition to hydrogen, some main group elements and dioxygen can combine to form peroxide (O22-). It exists as peroxide in -1 oxidation state.
- Alkali metals and dioxygen can combine to form superoxide (O2-) generally. It exists as a superoxide in -12 oxidation state.
- Group 1 and Group 2 metals have basic oxides, whereas non-metallic oxides typically have acidic oxides. Amphoteric oxides, which can react with both acids and bases, are created when group 13 metals like gallium and aluminium combine with dioxygen.
Examples of basic oxides include Na2O, CaO, BaO, CuO, etc,.
Examples of acidic oxides: SO2, CO2, Cl2O7, NO2, N2O5, etc,.
Examples of amphoteric oxides: Al2O3, Ga2O3, etc,.
- Some oxides are neutral because they don't react with either acid or base. Examples include CO, N2O, NO, etc.
- Dioxygen is a strong oxidizing agent.
Practice Problems
1. Calculate the oxidation state of Cs in CsO2, Cs2O2, Cs2O.
a. +4, +2, +1
b. +2, +2, +2
c. +2, +4, +1
d. +1, +1, +1
Answer: D
Solution: Since caesium is an alkali metal, it tends to donate electrons. As a result, it is in the +1 oxidation state.
In CsO2, oxygen exists in a superoxide form as O2-. The oxidation state of oxygen in superoxide is - 12. Consider the oxidation state of Cs to be x.
In Cs2O2, oxygen exists in a peroxide form as O22-. The oxidation state of oxygen in peroxide is -1. Consider the oxidation state of Cs to be x.
In Cs2O, oxygen exists in an oxide form as O2-. The oxidation state of oxygen in oxide is -2.
Consider the oxidation state of Cs to be x.
So, option D is the correct answer.
2. Which of the following oxides is acidic in nature?
a. MgO
b. Al2O3
c. NO2
d. Na2O
Answer: C
Solution: Since magnesium and sodium belong to Group 2 and Group 1, respectively, they will form basic oxides. Hence, MgO and Na2O are basic oxides. Al2O3 is an amphoteric oxide as it belongs to Group 13 elements. NO2 is an acidic oxide as it belongs to the main-group elements. Moreover, nitric acid and nitrous acid are produced when NO2 and water react.
So, option C is the correct answer.
3. In the process of corrosion of iron, oxygen acts as a/an
a. Oxidising agent
b. Reducing agent
c. Can act as both oxidising as well as reducing agent
d. Since iron corrosion is not a redox reaction, oxygen won't be reduced or oxidised in the process.
Answer: A
Solution: The corrosion of iron in the presence of water and oxygen is called rusting. In this redox process, iron functions as a reducing agent and gets oxidised, while oxygen acts as an oxidising agent and is reduced.
Here, the oxidation state of O changes from 0 to -2 which reflect oxygen is reduced and acts as oxidising agent.
So, option A is the correct answer.
4. What is/are the cause(s) of dioxygen's anomalous properties?
a. Small atomic radii
b. High electronegativity
c. Lack of vacant d-orbitals
d. All of these
Answer: D
Solution: Small atomic radii, high electronegativity, and the lack of vacant d-orbitals are, the primary causes of dioxygen's anomalous properties. Due to lack of vacant d orbital, it can not expand its octet. Because of smaller radii and high electronegativity, oxygen will form p-p bonds that exist in the gaseous state and can not form higher membered chains or polyatomic molecules whereas other members of group 16 can expand their octet as well as can form polyatomic molecules.
So, option D is the correct answer.
Frequently Asked Questions – FAQ
1. Why does dioxygen exist as gas in the Group 16 while other members exist in a solid state?
Answer: Dioxygen is a gas while others in the group are solid because of weak van der Waals forces in dioxygen and its ability to form p-p bonds.
Though the central atom belongs to the same group, why does H2O exist in a liquid state and H2S in a gaseous state?
Due to its high electronegativity and propensity for hydrogen bonding, the hydride of dioxygen, or water, is a liquid, unlike the hydrides of other elements in group 16 which are gases. That is why
H2O exist in a liquid state and a H2S in a gaseous state.
2. What are the industrial uses of dioxygen?
Answer: In addition to being used in mining, dioxygen is also used in the manufacturing of glass. It is employed in the processes of cutting and welding. The smelting of iron in the steel industry depends heavily on dioxygen.
3. Why is pure oxygen used in spacesuits?
Answer: Normal air, which is composed of 78% nitrogen, 21% oxygen, and 1% other gases, cannot be used in space suits because the low pressure would result in dangerously low oxygen levels in the blood and lungs, much like climbing Mount Everest does. Therefore, the majority of space suits offer a pure oxygen breathing environment.


