-
Call Now
1800-102-2727
Dinitrogen Pentoxide - Structure, Preparation, Properties, Uses, Practice problems and FAQ
Our family of oxides of nitrogen are like a pretty cool bunch of brothers from the same parents. And all of them have their very own wonderful and distinct personalities.
Look at the vibrant boys in the image! Consider they are brothers from the same mother. Isn’t it amazing, how they all have different physiques, and different smiles and when they grow up to become responsible citizens, all of them would shine differently in their own professional fields.

So are the oxides of nitrogen! So different and distinct in their characteristics, structure and properties. Some are inherently beneficial to health, some have medicinal value, and some are even toxic to the environment! Some are solid, some are gaseous; some are acidic and some are even neutral. Strangely they make an ensemble cast for portraying the vividness of chemistry. This page will be dedicated to one such slightly unstable oxide of nitrogen, you may say the eldest in the family-Dinitrogen pentoxide. (Where nitrogen is in its highest positive oxidation state and hence owns the position of the eldest brother!)
Let’s find out more about it!
TABLE OF CONTENTS
- Dinitrogen Pentoxide - Introduction
- Dinitrogen Pentoxide - Structure
- Dinitrogen Pentoxide - Preparation
- Dinitrogen Pentoxide - Physical Properties
- Dinitrogen Pentoxide - Chemical Properties
- Dinitrogen Pentoxide - Uses
- Practice Problems
- Frequently Asked Questions - FAQ
Dinitrogen Pentoxide - Introduction
Dinitrogen pentoxide, sometimes referred to as nitrogen pentoxide or nitric anhydride, is the chemical compound having the formula N2O5. It belongs to the group of binary nitrogen oxides, which simply consists of nitrogen and oxygen.
- Crystals of nitrogen pentoxide are colourless and melt around 41 °C. Its boiling point is 47 °C and sublimes just above room temperature to produce an odourless gas.
- Deville was the first to create N2O5 in the crystalline state by processing silver nitrate (AgNO3) with chlorine, in 1840.
- The anhydrous form of nitric acid, dinitrogen pentoxide, can be found as a colourless crystal.
- Dinitrogen pentoxide is a somewhat unstable nitrogen oxide that is utilised as an oxidiser in a variety of chemical processes.
- Dinitrogen pentoxide is a volatile and possibly toxic oxidant that was once employed as a reagent for nitrations when dissolved in chloroform.
- It is a nitrating substance. However, nitronium tetrafluoroborate (NO2BF4) has mainly superseded it in recent years.
- Dinitrogen pentoxide is highly dissociated in nitric acid to provide a high concentration of nitronium cation.
- Dinitrogen pentoxide is a chemical that is unstable and mildly explosive but has little commercial value at the moment.
Dinitrogen Pentoxide - Structure
Let the oxidation state of N in N2O5 be 'x'.
So,
∴
So, we know that the oxidation state of N in N2O5 is +5.
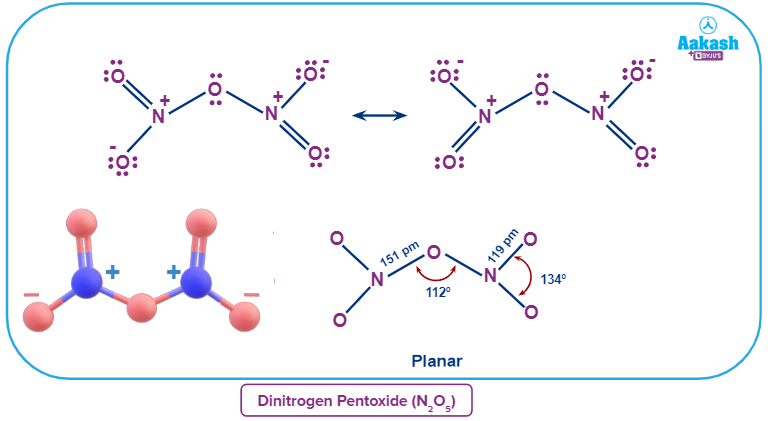
N2O5 in its solid state is a crystalline salt that is made up of separated linear nitronium ions () and planar trigonal nitrate anions (). The oxidation state of both the nitrogen centres is +5.
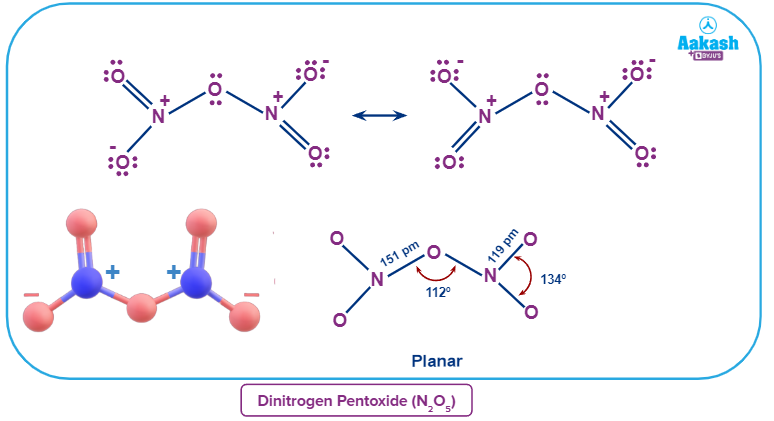
The chemical occurs as covalently linked molecules () in the gas phase or when dissolved in nonpolar solvents such as carbon tetrachloride. The O-N-O angle in each -NO2 wing is around 134° in the gas phase, and the N-O-N angle is about 112°, according to theoretical estimates for the minimum-energy arrangement. The two -NO2 groups are rotated around 35 degrees around the links to the central oxygen in that structure, away from the N-O-N plane. As a result, the molecule bears a propeller form, with one axis rotating at 180 degrees.
The free radicals nitrogen dioxide (NO2) and nitrogen trioxide (NO3) are formed when gaseous N2O5 absorbs UV light. The absorption spectrum is broad, with the highest wavelength of 160 nm.
Dinitrogen Pentoxide - Preparation
- On complete dehydration of nitric acid with diphosphorus pentoxide or oxidising nitrogen dioxide with ozone, dinitrogen pentoxide can be synthesised as white crystals. It is an unstable chemical that decomposes spontaneously into nitrogen dioxide and oxygen at ambient temperatures.
- When a combination of oxygen and nitrogen is pushed through an electric discharge, dinitrogen pentoxide is produced.
- The reaction of lithium nitrate (LiNO3) and bromine pentafluoride (BrF5) in a ratio greater than 3:1 is another laboratory method to produce dinitrogen pentoxide.
- Phosphoryl chloride (POCl3) or nitryl chloride (NO2Cl) interactions with silver nitrate (AgNO3) are another way of preparing dinitrogen pentoxide.
Dinitrogen Pentoxide - Physical Properties
- It is an acidic oxide.
- It is a colourless, deliquescent solid.
- Its dipole moment is 1.39 D
- Its crystal is hexagonal.
- It is an anhydride of nitric acid (HNO3).
- Dinitrogen pentoxide is soluble in chloroform, but its solubility is negligible in CCl4.
- It is soluble in water (reacts to produce nitric acid)
- Its density is
- Its molar mass is
- Its boiling point is (sublimes).
- The melting point of dinitrogen pentoxide is
Dinitrogen Pentoxide - Chemical Properties
- Dinitrogen pentoxide breaks down into NO2 and O2 at ambient temperature. At 0 °C, if the solid is stored in appropriately inert containers, the decomposition is minimal.
- Nitric acid is formed when dinitrogen pentoxide combines with water.
- When dinitrogen pentoxide interacts with sodium hydroxide, sodium nitrate and water are formed.
- Nitric acid (HNO3) and nitryl chloride (NO2Cl) are also produced by the reaction of N2O5 with hydrogen chloride (HCl).
- Between high temperatures of 600 and 1100 K (327–827 °C), dinitrogen pentoxide decomposes in two stoichiometric steps:
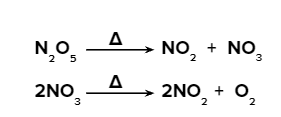
- N2O5 has stoichiometrically broken down into nitrogen dioxide and oxygen in shock waves. Nitrogen dioxide is unstable in relation to oxygen and nitrogen oxide NO at temperatures of 600 K and above. It is known that 0.1 mM nitrogen dioxide thermally decomposes in around two seconds at 1000 K.
- Dinitrogen pentoxide has been used as a reagent to introduce the NO2 functionality in organic compounds, for instance as a solution in chloroform. The following graphic illustrates this nitration reaction:
‘Ar’ denotes an arene moiety.
Dinitrogen Pentoxide - Uses
- Dinitrogen pentoxide is used in high-fuel rockets as a strong oxidizer.
- It is used in solvents that are not reliant on water to readily nitrate molecules that are extremely sensitive to water.
- For various syntheses in organic chemistry, dinitrogen pentoxide is used as a nitrating agent. A nitrating agent made from a combination of N2O5 and HNO3 is also effective.
- Dinitrogen pentoxide determines light intensity variations of different battery cells based on its rate of decomposition.
- N2O5, a strong oxidizer, may combine with ammonium salts and organic substances to create explosives. Nitrogen dioxide gas, which is highly hazardous, is created during dinitrogen pentoxide's breakdown.
Practice Problems
Q. 1. Dinitrogen pentoxide is an
A. Acidic Oxide
B. Basic Oxide
C. Neutral Oxide
D. None of the above
Answer: N2O5 is the most acidic of all the oxides of nitrogen. As the oxidation state of the central atom increases, so does the acidity. In N2O5, N is in its highest possible oxidation state, i.e, +5. Therefore, it is an acidic oxide. This can be further justified by the following reaction with a base. It reacts with a base to produce salt and water.
So, option A) is the correct answer.
Q. 2. What is the covalency of N in N2O5?
A. 2
B. 4
C. 5
D. 3
Answer: Since each nitrogen atom in has four bonds with the adjacent oxygen atoms, (three covalent bonds and one coordinate covalent bond) in dinitrogen pentoxide, nitrogen has a covalency of four.
So, option B) is the correct answer.
Q. 3. Nitric acid anhydride is?
A. N2O5
B. N2O3
C. N2O
D. None of the above
Answer: N2O5 is an acid anhydride of nitric acid. When combined with water, it creates (HNO3).
So, option A) is the correct answer.
Q. 4. Dinitrogen pentoxide can exist both in ionic and covalent forms. Justify the statement.
Answer: N2O5 is a rare instance of a substance that may change its structure based on the environment. Nitronium nitrate is a salt that exists as a solid and is composed of discrete nitronium cations ( ) and nitrate anions (). Yet, in the gas phase, it is a molecule that is covalently bonded.
N2O5 in its solid state is a crystalline salt that is made up of separated linear nitronium ions () and planar trigonal nitrate anions (). Thus, the given statement is justified.
Frequently Asked Questions - FAQ
Q1. Mention the health hazards associated with dinitrogen pentoxide?
Answer: It can cause respiratory tract irritation and eye or skin infection upon exposure. N2O5 is a powerful oxidant that reacts violently with organic molecules and ammonium salts, forming explosive combinations. The highly hazardous nitrogen dioxide gas is produced when dinitrogen pentoxide decomposes.
Q2. Is N2O5 ionic or covalent?
Answer: Dinitrogen pentoxide is a covalent molecule, in which each nitrogen atom has four covalent bonds and one coordinate covalent bond.
In solid-state, dinitrogen pentoxide is ionic. This is evident from the X-ray diffraction studies. Solid dinitrogen pentoxide occurs as (nitronium nitrate). Nitronium nitrate is dinitrogen pentoxide's ionic form. The following reaction may be used to illustrate this:
The atom has a -1 charge, whereas the sodium atom has a +1. is hence the ionic form of N2O5.
Q3. Is dinitrogen pentoxide associated with ozone layer depletion?
Answer: Yes, it is indirectly responsible for ozone depletion. Dinitrogen pentoxide serves as a key storage site for the NOx molecules that cause ozone depletion in the atmosphere. Its production creates a null cycle that keeps NO and NO2 momentarily in an inert state. The gases, later on, undergo a free radical reaction in the stratospheric region, boosting ozone layer depletion.
Q4. Is dinitrogen pentoxide carcinogenic?
Answer: Nitrogen dioxide gas, which is highly hazardous, is created during dinitrogen pentoxide's breakdown Although it is toxic and corrosive, it does not have a carcinogenic effect in particular.


