-
Call Now
1800-102-2727
Dennis’ Method - Introduction, Physical and Chemical Properties of Fluorine
Suppose you find there is a cavity in your teeth or you get mild gum disease. What will you do in that case? You will surely consult a dentist. But do you know what helps in preventing it? As they say, precaution is always better than cure. It is actually one of the forms of fluorine known as fluoride. Fluoride protects teeth from the bacteria present in plaque. So, fluorine is quite a useful element. For the first time, fluorine was discovered by Scheele. Fluorine is a highly reactive element and so it is not found in nature in its native state but is widely found in combined states. Let’s understand one of the methods where fluorine is isolated from fluoride.
Table of contents
- Introduction to Fluorine
- Dennis method
- Physical Properties of Fluorine Gas
- Chemical Properties of Fluorine Gas
- Practice Problems
- Frequently Asked Questions - FAQs
Introduction to Fluorine
Fluorine element has an atomic number 9 and is the 13th most abundant element of the earth crust and is highly reactive in nature. Fluorine has the highest electronegativity among all the elements present in the earth's crust. It usually combines to form a diatomic F2 which is a toxic gas. Fluorine usually exists as a fluoride ion (F-) in aqueous solutions. It is also present in a small amount in the form of fluoride in teeth, bones of animals and human beings, soil, sea, water etc. Some important minerals containing fluorine include Fluorspar (CaF2), Cryolite(Na3AlF6), potassium hydrogen fluoride (KHF2) etc.
Dennis method
In this method fluorine gas is isolated by electrolysis of a pure and dry fluoride compound [Generally potassium hydrogen fluoride (KHF2)] at a temperature of approximately 520 K. In this electrolytic method hydrogen gas is released at the cathode and fluorine gas is released at the anode. The electrolysis is done in a V-shaped electrically heated copper tube consisting of graphite electrodes. The copper tube placed is insulated properly to prevent heat loss.
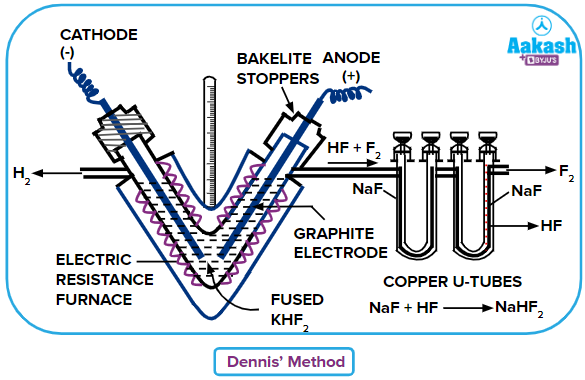
Electrolytic reaction for the isolation of fluorine will be:
H+ ion formed during the process will migrate towards the cathode and F- will migrate towards the anode respectively. Therefore,
Reaction at the cathode: potassium hydrogen fluoride (KHF2)
We know that cathode reduction takes place during an electrolytic process and therefore hydrogen gas will be liberated.
Reaction at the anode:
We know that on anode oxidation takes place during an electrolytic process and therefore fluorine gas will be liberated.
To remove the unwanted hydrogen fluoride prepared during the process as shown in the equation (i). It is then passed through the fused sodium fluoride (NaF) present in the U-tube to to prevent the reaction of hydrogen fluoride (HF) form reacting with the vessel.
Note: Dry fluoride compound should be used during the process rather than moist fluoride compound because in the presence of moisture fluorine gas liberated during the process will react with the water to form oxygen (O2) and ozone (O3).
Physical properties of fluorine gas
Few important physical properties of fluorine include:
- Fluorine is a pale yellow coloured gas with a pungent smell.
- It is heavier than air and is poisonous in nature.
- It can be condensed to a yellow liquid having a boiling point of 86 K.
- It can be solidified into a yellow crystal having a melting point of 53 K.
- It is diamagnetic in nature.
Chemical properties of fluorine gas
Fluorine is a highly reactive diatomic gas containing one 𝜎 bond. It has a high electronegativity value and can react with a large number of substances.
- Reaction with water: Fluorine reacts with water producing oxygen and ozone.
- Reaction with alkali: Fluorine reacts with cold and dilute alkali to form oxygen difluoride (OF2).
When it reacts with hot and concentrated alkali O2(g) is released.
- Reaction with non-metal: Fluorine reacts with almost all elements directly except nitrogen and oxygen under ordinary conditions.
Fluorine can react with hydrogen at a very low temperature of (21 K to 23 K) or even in dark to form HF. This reaction is highly exothermic in nature which shows high-affinity of fluorine towards hydrogen.
- Reaction with metals: Fluorine reacts with almost all the metals forming metal fluoride.
Note: Copper and mercury do not react with the fluorine because CuF2 and HgF2 formed initially on the surface of the metal and prevents the further reaction.
- Reaction with halide: Fluorine is the strongest oxidising agent and oxidises all halides into corresponding halogens from its compound.
- Reaction with ammonia: Fluorine reacts with excess ammonia to liberate nitrogen gas but when excess fluorine is allowed to react with ammonia, nitrogen trifluoride is formed.
- Oxidising properties: Fluorine is the strongest oxidising agent and can oxidise several compounds and itself get reduced.
- Reaction with hydrocarbons: Fluorine also reacts with hydrocarbons violently to form fluorinated forms of hydrocarbons.
Practice problems
Q. Select the correct option for the product that cannot be formed when fluorine reacts with water.
a. O2
b. HF
c. O3
d. H2
Answer: (D)
Solution Fluorine reacts with water to form hydrogen fluoride along with oxygen (O2) or ozone (O3).
Q. In the reaction the oxidation state of fluorine changes from
a. 0 to -1
b. 0 to 1
c. -1 to 1
d. No change in oxidation state
Answer: (A)
Solution: Fluorine has the highest electronegative value among the elements present in a periodic table and therefore it cannot exhibit the +ve oxidation state. In the above reaction initially, fluorine is present in the form of a diatomic molecule (F2) where the oxidation state of fluorine is 0. In the products, fluorine is present in the form of HF the molecule and its oxidation state can be calculated as:
We know, that the oxidation state of hydrogen, when combined with more electronegative element, is 1.
Let the oxidation state of fluorine be X.
Sum of the oxidation states of all the elements should be equal to the net charge present on the molecule. Therefore,
So, the oxidation state of fluorine changes from 0 to -1 in the given reaction.
Q. In Dennis' method of isolation of fluorine the possible electrolyte that can be used to obtain fluorine gas is:
a. KHF2
b. HF
c. KClO3
d. NaF
Answer: (A)
Solution: In this method, fluorine gas is isolated by electrolysis of a pure and dry sodium or potassium hydrogen fluoride compound [Generally potassium hydrogen fluoride (KHF2)] at a temperature of approximately 520 K.
Q. Which compound is formed other than water and sodium fluoride when fluorine reacts with hot and concentrated sodium hydroxide solution?
a. OF2
b. H2
c. O2
d. Both A and C will be formed
Answer: (C)
When fluorine reacts with hot and concentrated sodium hydroxide solution it liberates oxygen gas (O2) along with the sodium fluoride and water.
Frequently Asked Questions-FAQs
Q. What are the health hazards of fluorine?
Answer: Fluorine gas is extremely irritating to the skin, eyes and lungs. This substance is extremely dangerous. Fluorine gas has an effect on the eyes and nose at low doses. If inhaled, it causes breathing problems and may damage the lungs caused to excessive fluoride concentrations. Also, can result in the death of an individual if inhaled in larger quantities.
Q. Why fluorine element is not obtained in the free state from nature?
Answer: Fluorine element has the highest electronegativity value and it is highly reactive in nature as it contains 7 electrons in its valence shell and needs only one electron to complete its octet and can react with almost all the metals and non-metals directly therefore it is not found in the free state rather obtained in combined form.
Q. How do we determine the presence of fluorine elements in organic compounds?
Answer: Fluorine in organic mixtures can be measured by combustion of the organic mixture in the presence of air at 900°C in a quartz tube with a platinum catalyst, then titrating the combustion products using the principles of acid-base titration.
Q. What is the reason for anomalous behaviour of fluorine element?
Answer: Fluorine element shows anomalous behaviour as compared with the other elements present in the same group because of the following reasons:
- It possesses the highest electronegativity.
- It has the smallest size as compared with the other elements present in the group i.e.,
chlorine (Cl), bomine (Br) and iodine (I) elements.
- It doesn't have vacant d-orbitals in its valence shell.
- It has the highest positive reduction potential value.


