-
Call Now
1800-102-2727
Denaturation of Protein - Proteins, Sources, Classification, Denatured Protein, Significance, Practice Problems & FAQs
We all must have seen an egg changing its state on heating. But have you ever wondered why this happens? What is there in the egg which is changing its consistency from a gel-like substance to a semi-solid or solid substance on heating?


This disruption of the basic structure of the egg is referred to as Denaturation. Now to answer all your questions and to clear our confusion.
Let’s understand everything about Denaturation!
Table of content:
- Proteins
- Sources
- Classification
- Denaturation
- Significance
- Practice problems
- Frequently asked questions (FAQs)
Proteins:
To understand Denaturation, let's first understand protein.
Proteins are large molecules made up of Peptide bonds between fundamental building blocks called amino acids. In addition to carbon, hydrogen, and oxygen, amino acids contain nitrogen.
Proteins are large, complex molecules made up of twisted and folded amino acid strands. Proteins are extraordinarily complex molecules that play a crucial role in life's most basic and critical functions. Among these include metabolism, motility, defense, cellular communication, and molecular recognition.
Sources of Protein:
Below are the major sources of protein from where intake of protein is mainly considered.
- Beans and legumes
- Eggs
- Greek yogurt
- Milk
- Cottage cheese
- Peanuts
- Lentils

Classification of Proteins:
We know that Proteins are large, intricate molecules made up of twisted and folded amino acid strands. Each amino acid is linked to the next by covalent bonds. Proteins are divided into three categories:
Primary (first level) – Protein structure is a sequence of amino acids in a chain.
Secondary (secondary level) – The protein structure is formed by folding and twisting the amino acid chain.
Tertiary (third level) – Protein structure is formed when the twists and folds of the secondary structure fold again to form a larger three-dimensional structure.
Quaternary (fourth level) – A quaternary structure is a protein that has more than one folded amino acid chain.
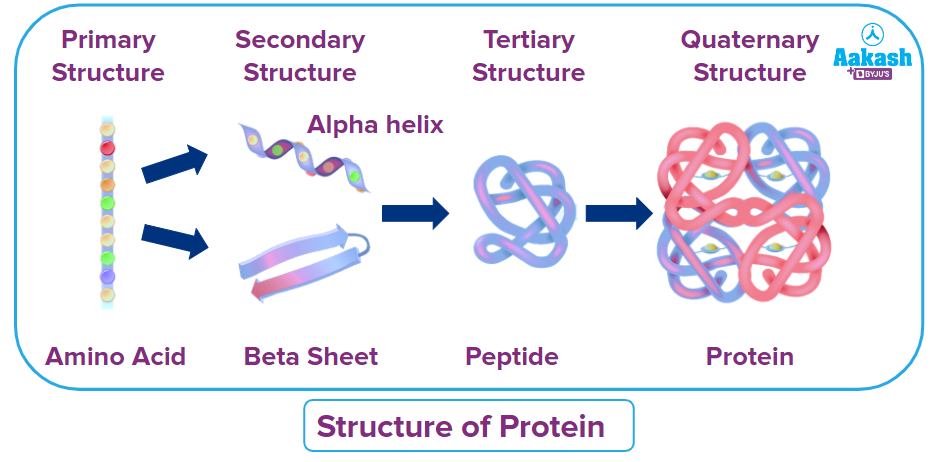
The protein molecule's stability is maintained by a variety of physical and chemical factors. The molecular structure will shift and create disruption if certain circumstances are disrupted. Temperature and pH changes can cause both physical and chemical changes in protein.
Denaturation:
Now comes a very interesting question.
What happens when temperature and pH changes in native protein ?
Here Comes Denaturation!!
Due to change in temperature and pH, H-bonds are disturbed, Globules unfold, Helix uncoils and with the result of which proteins lose their biological activity which can be defined as the process of Denaturation of proteins as shown below.
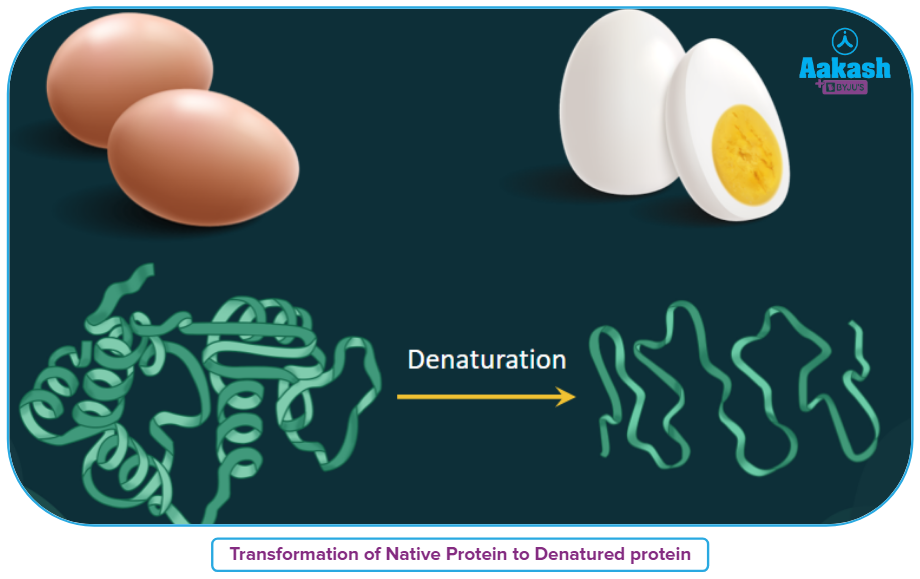
During denaturation only secondary & tertiary structures are destroyed whereas primary structures remain intact.
Denaturation is irreversible in some proteins, and the protein is permanently destroyed. Denaturation of other proteins can be reversed in some cases if the denaturing agent is removed, a process known as renaturation.
Examples of denatured proteins:
- In fresh eggs, the egg white is mostly made up of soluble proteins and is liquid and translucent.
- Adding acids to milk, such as citric acid from lemon juice, denatures milk proteins and causes it to curdle.
- The solid white fraction of the separated whey is denatured protein.
- This process of denaturation is utilized to manufacture yogurt and fresh cheese when it is correctly regulated.
- Furthermore, the presence of denatured proteins in the lens of the eye is one of the leading causes of blindness.
Significance:
- We can use denaturing DNA to amplify and hybridize DNA in a variety of downstream experiments. In addition, it aids in biological research.
- The capacity of nucleic acid strands to separate is used in many laboratory methods. Denaturation is accomplished through knowing the characteristics of nucleic acids. DNA Sequencing is a result of the denaturing process.
Related video:Denaturation of Protein (37:49 to 38:48)
Practice problems:
Q1. Cellulose fibers are similar to _________in terms of proteins, but -amylose is similar to _______.
A. Sheets, Helices
B. Sheets, Hydrophobic core
C. Turns, Helices
D. Helices, Sheets
Answer: A
Solution: Cellulose fibers are similar to sheets in terms of proteins, but -amylose is similar to helices.
Q2. Which of the following statements is true regarding denaturation of proteins?
A. Takes place due to changes in temperature and pH
B. During denaturation, only secondary & tertiary structures are destroyed whereas primary structures remain intact.
C. Denaturation is irreversible in some proteins
D. All of the above
Answer: D
Solution: When a protein's distinctive three-dimensional structure is subjected to alterations, a situation known as denaturation of the proteins results. The hydrogen bonds that are present in proteins become perturbed as a result of changes in temperature, pH, or other chemical activity.
All of the statements given in option A, B and C are correct so option D is the correct answer.
Q3. Which of the following is a result of protein denaturation?
A. Boiling of milk
B. Clotting of blood
C. Formation of yogurt and fresh cheese
D. None of the above
Answer: C
Solution: The process of denaturation is utilized to manufacture yogurt and fresh cheese when it is correctly regulated. So, in this case, option C is the correct answer.
Q4. Which of the following factors determines a protein's three-dimensional shape?
A. Sequence of amino acids
B. Interaction with other polypeptides
C. Molecular interaction
D. Both A and C
Answer: D
Solution: Sequence of amino acids determines a protein's three-dimensional shape. So, option A is the correct answer.. The amino acid sequence, or primary structure, of the protein, governs how it folds and their molecular interaction helps in deciding the 3D-structure of protein. Hence option C is also a correct answer.
Frequently asked questions - FAQs
Q1. What role does protein denaturation have in human health?
Answer: Denaturation is a critical process for all living things. When we eat, the acids in our stomach, specifically HCl, break down the protein molecule components so that the nutrients can be easily consumed by the body. Improve pepsin enzymes' capacity to convert protein to peptides without disturbing secondary and tertiary protein structures. Without protein denaturation, digestion would not have been feasible.
Q2. What’s the difference between globular and fibrous proteins?
Answer: Fibrous Proteins- These proteins are made up of linear thread-like molecules that form fibers when they lie next to each other (parallel). At several sites, hydrogen bonds and certain disulphide bonds hold the polypeptide chains together. As a result, intermolecular attraction forces are exceedingly strong, making fibrous proteins water insoluble. These proteins are also resistant to moderate temperature and pH variations. The primary structural component of animal tissues is fibrous proteins. Collagen in tendons, fibrosis in silk, and myosin in muscles are all examples of keratin, which can be found in skin, hair, nails, and wool.
Globular Proteins: These proteins have a polypeptide chain that is curled around itself, giving the molecule an almost spheroidal form. Hydrophobic (non-polar) components are pushed in whereas hydrophilic (polar) parts are pushed out during folding. Globular proteins are water soluble due to strong interactions between water molecules and polar groups. When compared to fibrous proteins, these proteins are particularly sensitive to tiny changes in temperature and pH. All enzymes, as well as several hormones such as insulin from the pancreas and thyroglobulin from the thyroid gland, fall into this category.
Q3. What impact does denaturation have on the structure of proteins?
Answer: A change in temperature (when heated) or a change in the pH of the medium denatures proteins. As a result, the hydrogen bonding between the proteins is disturbed, and the proteins lose their biological activity, causing them to change their nature. The tertiary and secondary structures of proteins are destroyed during denaturation, but the fundamental structures remain intact.
Q4. What factors contribute to the denaturing of proteins?
Answer: Protein denaturation occurs for a variety of causes. Increased temperature, which ruptures the structure of protein molecules, pH fluctuations, Vigorous shaking, High pressure, protonation of amino acid residues, and UV light and radiation are only a few of them.


