-
Call Now
1800-102-2727
Dehydrohalogenation- Dehydrohalogenation to Alkene and Alkyne, Saytzeff or Zaitsev’s Rule, Practice Problems and Frequently Asked Questions
As we all know, artificial ripening of fruits is now very common, though it is prohibited because of causing health problems. Do you know how to identify naturally ripened fruit from artificial ripened?
In spite of the bad effect, fruit sellers introduce chemicals to accelerate the artificial ripening process.
Do you know which chemical is commonly used in this process?
Ethylene gas which can be easily produced is the answer. Ripening fruits and vegetables emit naturally ethylene, which causes them to ripen further. The gas (C2H4) produced by plants is known as the "ripening hormone" because it promotes fruit ripening. This means that this alkene is extremely useful.
So, let us look at a new method for producing alkene and alkyne also via dehydrohalogenation.

Table of Contents
- Dehydrohalogenation to Alkenes
- Saytzeff or Zaitsev rule
- Dehydrohalogenation to Alkynes
- Practice Problems
- Frequently Asked Question
Dehydrohalogenation to Alkenes
It is one of the methods for producing alkenes from alkyl halides via dehydrohalogenation, which means the removal of HX. Alcoholic KOH is the reagent used in this process. The elimination reaction is named after the removal of hydrogen and halogen present in the molecule. The hydrogen atom opposite the halogen atom attached to the carbon (carbon atom next to the halogen atom) is removed. As a result, it is referred to as anti-elimination.
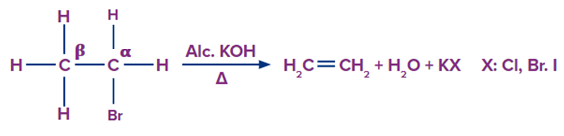
Reaction mechanism
Step 1: Removal of Hydrogen
The hydroxide ion derived from alcoholic KOH functions as a strong base. It attacks the β-H atom, which is naturally slightly acidic, and separates it from the alkyl halide molecule.
Step 2 Removal of halogen
The electrons shared by the broken hydrogen-carbon bond are drawn to the ⍺-carbon atom, which has a slight electron deficiency due to its attachment to the electronegative halogen atom. The halogen atom breaks as these electrons approach the ⍺-carbon atom, resulting in the formation of the double bond.
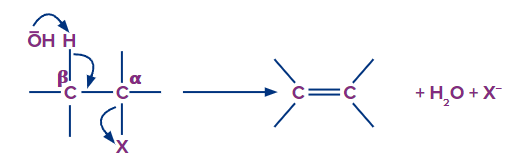
Saytzeff or Zaitsev rule
Zaitsev's rule, Saytzev's rule, and Z-rule are all variations of Saytzeff's rule. Alexander Zaitsev, a Russian chemist, studied various elimination reactions and discovered a general pattern in the resulting alkenes. Based on this analysis, Zaitsev concluded that stable alkenes are formed when hydrogen is removed from -carbon with a low number of hydrogen substituents.
Saytzeff's Rule enters the picture during elimination reactions. The most substituted product is the most stable and preferred. This rule only applies to the regiochemistry of the elimination reaction and does not generalise about the product stereochemistry.
The number of alkyl groups attached to the alkene predicts the major and minor products.
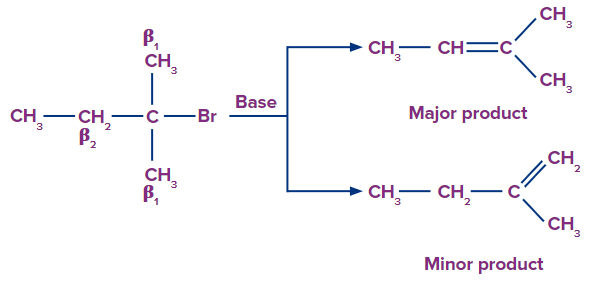
Rate of Elimination
Rate: Tertiary alkyl halide > Secondary alkyl halide > Primary alkyl halide
The rate of elimination reaction directly depends upon the stability of alkene.
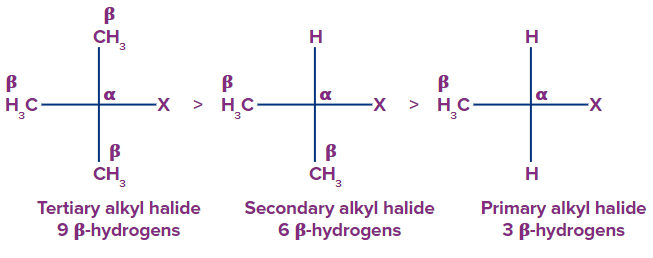
This is because tertiary halides have more number of β-hydrogens as compared to secondary and
primary halides.
Dehydrohalogenation of RX
Reactivity: R−I > R−Br > R−Cl > R−F
This is because iodide is better leaving group due to its large size.
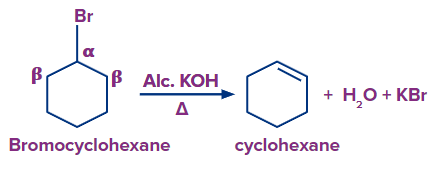
Dehydrohalogenation to Alkynes
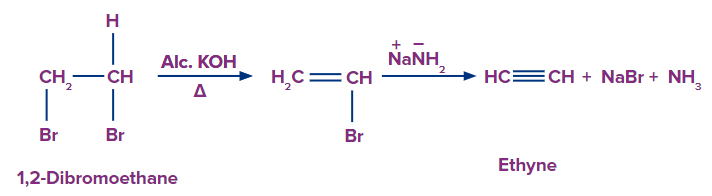
Vicinal dihalides undergoes dehydrohalogenation when treated with alcoholic potassium hydroxide to release one HX to an alkenyl halide. On further treatment with a stronger base, one more molecule of hydrogen halide is eliminated from the alkenyl halide, yielding an alkyne. Dehydrohalogenation refers to the elimination of hydrogen along with a halogen in order to obtain an alkyne.
Practice Problems
Q1. What is the rate of dehydrohalogenation in increasing order?
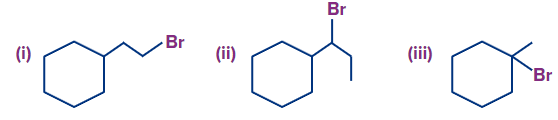
A. (I)<(II)<(III)
B. (II)<(I)<(III)
C. (III)<(II)<(I)
D. (I)<(III)<(II)
Solution: We know that the rate of dehydrohalogenation of alkyl halide is as follows:
Tertiary alkyl halide > Secondary alkyl halide > Primary alkyl halide
The primary alkyl halide is (I), the secondary alkyl halide is (II), and the tertiary alkyl halide is (III). As a result, the following will be the order: (I) <(II)< (III).
The rate of dehydrohalogenation reaction is increasing in the following order: (I) (II) (III).So, the correct answer is option (A).
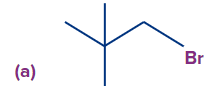
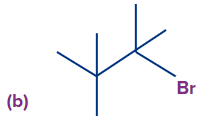
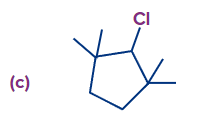
Q2. Find the possible alkenes after dehydrohalogenation and the types of β-carbons in the following molecules respectively.
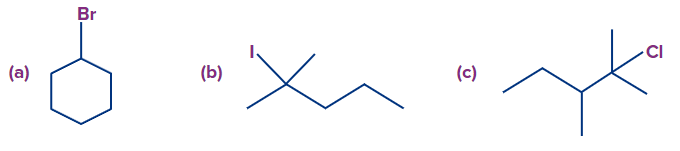
A. (a): 2,2 ; (b): 2,2 ; (c): 2,2
B. (a): 1,2 ; (b): 2,2 ; (c): 2,1
C. (a): 2,1 ; (b): 2,1 ; (c): 2,2
D. (a): 1,1 ; (b): 2,2 ; (c): 2,2
Solution: In compound (a), there is only one type of β-hydrogen and therefore, only one alkene will be
formed (cyclohexene).
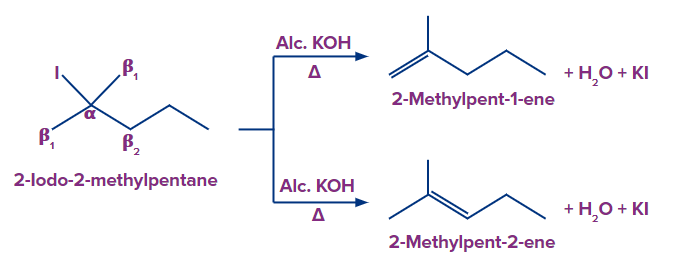
In compound (b), we have two types of β-hydrogen and therefore, two alkenes will be formed
(2-methylpent-1-ene and 2-methylpent-2-ene).
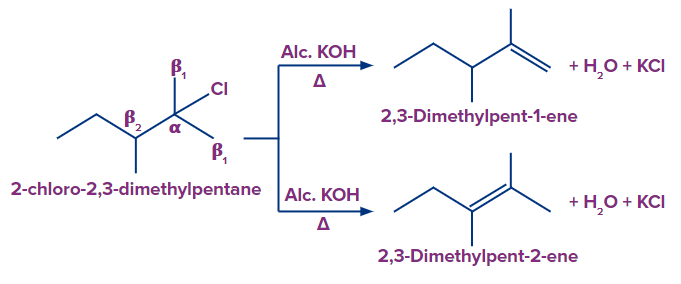
In compound (c), we have two types of β-hydrogen and therefore, two alkenes will be formed
(2,3-dimethylpent-2-ene and 2,3-dimethylpent-1-ene).
Hence, the correct answer is option (D).
Q3. Which of the given compounds produce a β-elimination product?
Solution: The presence of a β-hydrogen is required for the dehydrohalogenation (β-elimination) reaction to occur.
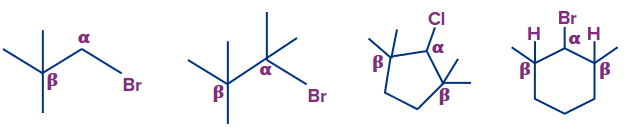
Because the given compound (A), (B) and (C) contains no β-hydrogen, it does not undergo dehydrohalogenation. Whereas in option (D), there are 2 β-hydrogen present in the molecule which leads to the desired product.
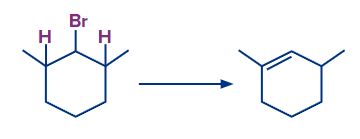
Hence, the correct answer is option (D).
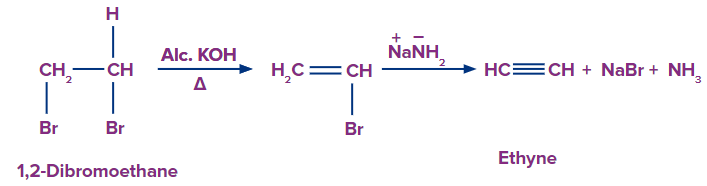
Q4. Pick up the true statement about the product formed when 1,2 dibromoethane is treated with alcoholic potassium hydroxide followed by soda amide?
A. Its common name is Acetylene
B. It is used in welding and cutting processes
C. It is an extremely flammable gas which can easily react with atmospheric oxygen result in fire and explosion
D. All of these
Solution: When 1,2 dibromo ethane is treated with alcoholic potassium hydroxide, it undergoes dehydrohalogenation. One molecule of hydrogen bromide is eliminated during sodamide treatment to form ethenyl bromide, which yields ethyne. The common name of ethyne is acetylene and it is used in welding and cutting processes. It is also a highly flammable gas that, when combined with atmospheric oxygen, can easily ignite and explode. Hence, all the statements are correct which means the appropriate option is (D).
Frequently Asked Question
Q1. Why is Soda amide used in dehydrohalogenation of vicinal dihalide?
Answer: For removing hydrogen from alkenyl halide, alc. KOH is not used. A stronger base is used, i.e.,
NaNH2, as hydrogen has to be removed from an sp2 hybridised carbon atom, forming a strong
Bond.
Q2. What is the difference in the function of Aqueous KOH and alcoholic KOH?
Answer: Simply put, alcoholic KOH causes the formation of alkenes, which is an elimination reaction, whereas aqueous KOH causes the formation of alcohols, which is a substitution reaction. While alcoholic KOH is commonly used to form Alkene from Alkyl Halides, aqueous KOH is used to form alcohol from Alkyl Halides. Dehydrohalogenation is accomplished using alcoholic KOH.
Q3. Which is the more strong base, Alcoholic KOH or Aqueous KOH?
Answer: In water, alcoholic KOH dissociates to form RO- ions, which are strong bases. It abstracts hydrogen, causing elimination in the reaction, and Aqueous KOH, being a weak base, causes substitution in the reaction.
Q4. What are the applications of Alkene?
Answer: Alkenes have numerous applications in manufacturing. As starting materials, they are used in the synthesis of alcohols, plastics, lacquers, detergents, and fuels. In the chemical industry, ethene, propene, and 1,3-butadiene are the three most significant alkenes.