-
Call Now
1800-102-2727
Cyclic Compounds: Definition. Classification, Nomenclature and Uses of Cyclic Compounds, Practice Problems & Frequently Asked Questions
Consider some substances that have a powerful scent for a moment.
What types of ideas cross your mind?
Vanilla, perfume, or maybe even cinnamon? These are all fragrant or aromatic in some way. Despite having quite varied smells, these substances do share one thing in common: they are all composed of aromatic compounds.
What exactly are aromatic chemicals then? Aromatic compounds are molecules that have a chemical structure that consists of one or more rings with alternate single and double bonds. There are some substances that are chemically aromatic but do not have a recognisable scent, despite the fact that many aromatic compounds in nature have an odour.
Table of Contents:
- Definition of Cyclic Compounds
- Classification of Cyclic Compounds
- Nomenclature of Cyclic Compounds
- Uses of Cyclic Compounds
- Practice Problems
- Frequently Asked Questions(FAQs)
Definition of Cyclic Compounds:
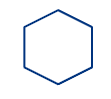
Cyclic compounds are also referred to as "Ring compounds". Cyclic compounds, as their second name implies, are those in which one or more atoms are linked together to form one or more closed rings. These compound rings don't necessarily need to be the same size on either side.
Examples :
|
Structure |
Name |
|
|
Cyclohexane |
|
|
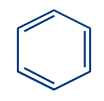
Benzene |
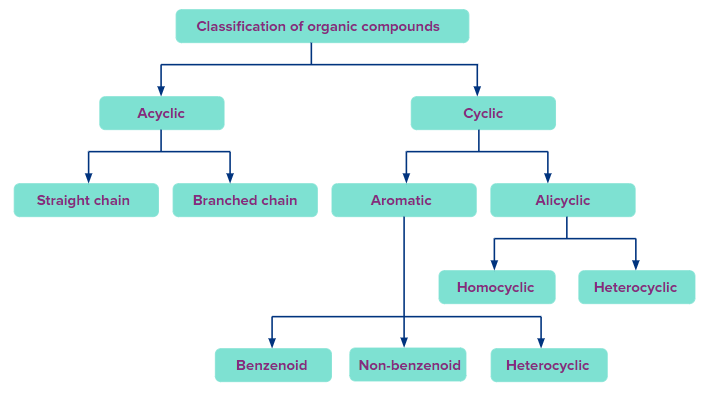
Classification of Cyclic compounds:
Following is a general classification of organic compounds based on structure:
A) Alicyclic compounds: Organic compounds with one or more cyclic rings, not having continuous conjugated multiple bonds are referred to as alicyclic compounds.
Examples: Cyclopropane
Homocyclic and heterocyclic compounds are subcategories of acyclic compounds:
(i) Homocyclic or carbocyclic compounds: The atoms that make up this ring structure will be of the same element, which in this case is carbon. We refer to these as carbocyclic or homocyclic compounds. In this molecule, rings are formed only by carbon.
Example: Cyclohexane is a six-membered ring constituting only carbon atoms hence it is a homocyclic or carbocyclic compound.
(ii) Aliphatic Heterocyclic compounds: Heterocyclic compounds are substances where the ring is formed by carbon and one or more non-carbon atoms, such as S, O, N, and others. For instance
Example: 1. Tetrahydrothiophene
B) Aromatic Compounds: The conjugated planar ring systems with delocalized pi-electron clouds that replace individual alternating double and single bonds in chemical compounds are known as aromatic compounds. They are known by the names arenes or aromatics as well.
(i) Benzenoid Compounds: The finest examples are toluene and benzene. Aromatic compounds are cyclic unsaturated molecules that possess aromaticity. Aromatic compounds should adhere to Huckel's Rule. These rules must be adhered to for a chemical to be considered aromatic.
- The molecule needs to be coplanar.
- Complete delocalization of the pi-electron in the ring (conjugated ℼ--electron arrangement).
- The presence of (4n+2) ℼ-electrons in the ring, where n is an integer (n=0,1,2,3...).
The aromatic compounds are further divided into the following categories based on the type of ring:
The term "benzenoid aromatic compounds" refers to aromatic compounds that have a minimum one benzene ring, which is an alternating double-bond structure made up of six carbon atoms and having
4n +2, pi-electrons.
Examples: Naphthalene and Toluene etc.
(ii) Non-Benzenoid compounds: Non-benzenoid aromatic compounds are highly unsaturated aromatic compounds without a benzene ring in their ring structure. Conjugated systems based on planar cyclic structure are present in these substances. Azulene contains 10 electrons since it possesses 5 double bonds, as seen below For instance
Example: Azulene
(iii) Aromatic heterocyclic compounds: These are cyclic organic compounds satisfying the aromatic conditions and forming a ring with one or more atoms of O, N and S along with carbon. Not only are these compounds heterocyclic, but they are also aromatic heterocyclic molecules.
Sigma bonds and delocalized electrons between atoms that are part of a ring system are found in aromatic compounds. These substances are referred to as aromatic compounds because of their pleasant aroma.
Aromatic compounds are defined as organic substances that exhibit -electron delocalization and follow Huckel’s rule of aromaticity.
Examples: Pyrrole
Nomenclature of Cyclic Compounds:
- Nomenclature of Alicyclic compounds
Rule 1:
(a) If a ring has the same or more carbon atoms than the alkyl groups linked to it, the alkyl groups are considered as substituents and so named alkyl cycloalkane as per the IUPAC nomenclature.
(b) If a ring has fewer carbon atoms than the alkyl groups linked to it, the cyclic group is considered as a substituted and known as cycloalkyl alkane.

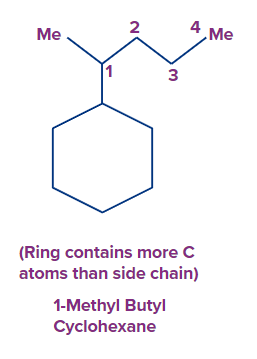
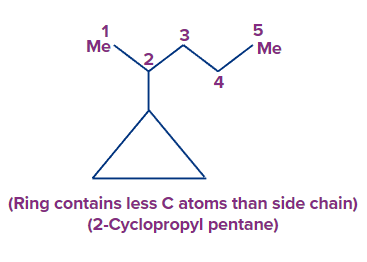
Rule 2: If the ring has multiple bonds as well as additional substituents, the numbering is done in such a way that the multiple bonds get the lowest number.
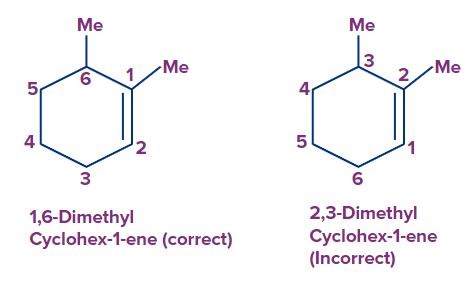
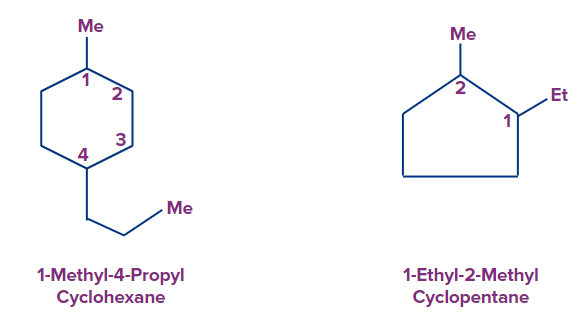
Rule 3:
If the ring contains two or more alkyl groups or other substituents, their positions are denoted by 1, 2, 3, etc. The lowest number is assigned to the substituent that comes first in the alphabetical list.
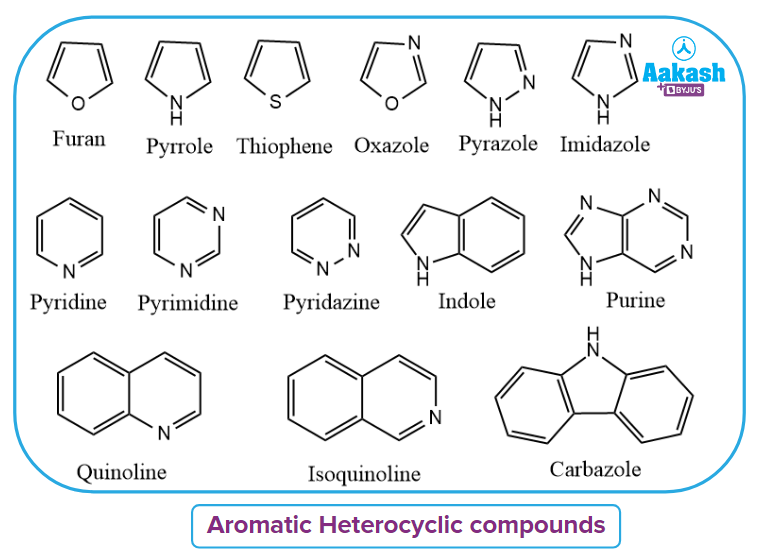
Aromatic heterocyclic compounds:
Aromatic heterocyclic compounds are ones that have a ring structure plus one or more extra elements, such as oxygen, nitrogen, phosphorus, or sulphur, as well as sigma bonds and delocalized electrons between the carbon atoms. In this system, the ring system contains atoms of multiple types.
Some examples are given as:
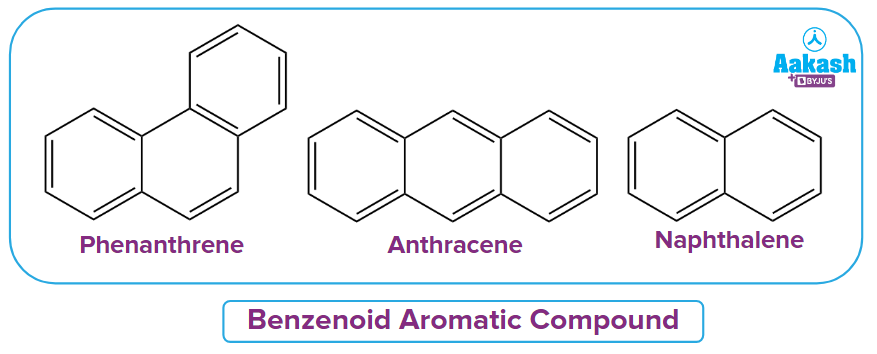
Aromatic benzenoid compounds:
Two or more fused rings and at least one benzene ring are characteristics of benzenoid aromatic systems, which are aromatic compounds.
The entire system may be regarded as aromatic even if just one component (ring) is aromatic.
Here are a few examples: phenanthrene, anthracene, and naphthene.
Uses of Cyclic Compounds:
- In the synthesis of pharmaceuticals for use in medicine, cycloalkanes are employed as an organic solvent.
- A cycloalkane called cyclopropane is employed as an anaesthetic in the medical industry.
- Cycloalkanes are employed in the production of hair care items.
- They are employed by the food industry.
- They are also employed in the petroleum sector.
- Carboplatin, a component of the cycloalkane cyclobutane, is employed in cancer treatment.
- Cyclobutane is employed for pigmentation purposes.
- In the industries that manufacture perfumes, cyclobutane is employed to create fragrances.
- As steroids, cyclobutane can also be present in the tissues of both plants and mammals.
Practice Problems:
Q1. Which of the following cyclic compounds has the highest degree of stability in accordance with Baeyer's strain theory?
(A) Cyclopentane
(B) Cyclopropane
(C) Cyclobutane
(D) None of the above
Answer: (A)
Solution: All three carbon atoms in cyclopropane are located in the corners of the triangle's equilateral shape. Thus, cyclopropane has a 60o C-C-C bond angle, cyclobutane has a 90o C-C-C bond angle and cyclopentane has a 108o C-C-C bond angle. Because of its close to tetrahedral bond angles and less angle strain, cyclopentane is the most stable cyclic molecule and cyclopropane is the least stable cyclic molecule.
Q2. Which among the following is an example of an acyclic organic compound?
(A) Benzene
(B) Furan
(C) Cyclopropane
(D) Isobutane
Answer: (D)
Solution: Benzene, furan and cyclopropane all are cyclic organic compounds, isobutane is a branched acyclic organic compound.
Q3. Among the following, which has a higher melting point?
(A) Cyclopentane
(B) Cyclooctane
(C) Cyclopropane
(D) They all have equal melting points
Answer: (B)
Solution: Cycloalkanes are a category of alkanes that contain one or more rings of carbon atoms. In general, as the number of carbons rises, cycloalkanes' melting point, boiling temperature, and density rise. This pattern develops because larger membered rings have a greater number of bonds, making the bonds more difficult to break and occurs because the molecule is effectively under the influence of more London dispersion forces, which makes it much more difficult for it to melt or boil.
Q4. Cyclobutane is an example of which type of organic molecule?
(A) Homocyclic
(B) Non-benzenoid
(C) Aromatic Heterocyclic
(D) Acyclic
Answer: (A)
Solution: The atoms that make up this ring structure all come from the same element, which in this case is carbon. We refer to these as carbocyclic or homocyclic compounds. In this molecule, only carbon may be present. Cyclobutane is a four-membered ring constituting only carbon atoms hence it is a homocyclic or carbocyclic compound.
Frequently Asked Questions(FAQs):
Q1. Why does ring strain exist in cycloalkanes?
Answer: In cycloalkanes, the carbons are sp3 hybridised, which means that they do not have the predicted ideal bond angles of 109.5o. This leads to ring strain, which is brought on by the need for the carbons to be located at the ideal bond angle.
The tetrahedral carbon figure showed the cause of ring strain. The ideal C-C-C bond angle is 109.5o, while the C-C-C bond angles in cyclopropane and cyclobutane are 60o and 90o, respectively. Cyclopropane and cyclobutane have a high ring strain as a result of this bond angle. However, because the bond angle between the carbons is much closer to 109.5o in compounds like cyclohexane and cyclopentane, they would have a significantly smaller ring strain.
Q2. What exactly are London Dispersion Forces and how do they relate to cycloalkanes?
Answer: Although alkanes and cycloalkanes are identical, the ring configuration of a cycloalkane provides a larger area of contact, leading to higher London Dispersion forces. Additionally, ring strain makes certain cycloalkanes more reactive. The attractive or repulsive forces between molecules or between atoms within a single molecule are known as London Dispersion Forces. London dispersion forces for cycloalkanes are the repelling forces between the molecules that result in ring strain.
Q3. How do cycloalkanes handle ring strain?
Answer: Conformers are formed by some cycloalkanes, such as cyclohexane, to deal with ring strain. Conformers are stereoisomer molecules that exist as distinct isomers of the same connectivity and formula, in this case to reduce ring strain. The rotations around the sigma bonds reduce ring strain in conformers.
Q4. Why do cycloalkanes differ from regular alkanes in terms of their physical characteristics?
Answer: Because they have more London dispersion forces than regular alkanes, cycloalkanes have unusual physical characteristics. Additionally, cycloalkanes have the potential to have higher steric hindrance, which raises their relative energy level.