-
Call Now
1800-102-2727
Coordination Compounds - Introduction, Important Terms, Properties, Classification, Ligands, IUPAC Nomenclature, Isomerism and Applications
You must have heard about the diagnosis which is done in the laboratory which is a haemoglobin test.
But do you know why it is important to maintain a particular range of haemoglobin in the body?
Haemoglobin is a kind of globular protein found in red blood cells (RBCs) that transports oxygen throughout our bodies via blood. It is a respiratory pigment that aids in the transport of oxygen from the lungs to various parts of the body as oxyhaemoglobin. Carbon dioxide is also transported back through haemoglobin as carbaminohaemoglobin.
But do you know what the structure of haemoglobin is?
Haemoglobin is made up of “Haeme” complex-ions with tetrapyrrole porphyrin ring structures and a central Fe2+ ion. Now, this compound is a coordination compound.
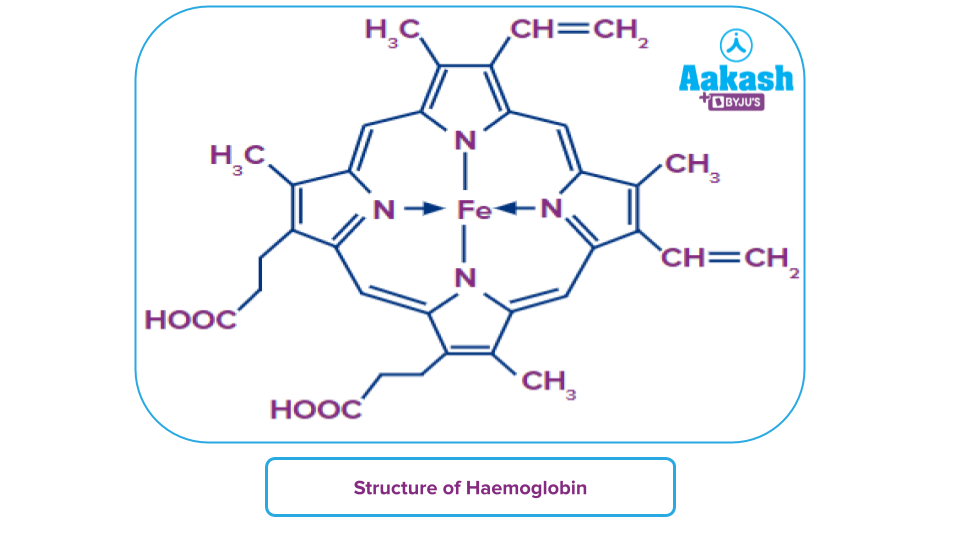
Let us learn more about the coordination complex compounds in this article.
Table of Contents
- Introduction to Addition Compounds
- What is a Coordination Compound?
- Important Terms
- Properties of Coordination Compounds
- Classification of Coordination Compounds
- Classification of Ligands
- Iupac nomenclature of coordination compounds
- Isomerism in coordination compounds
- Werner’s Theory
- Magnetic Properties of Coordination Compounds
- Stability of Coordination Compounds
- Colour of Coordination Compounds
- Application of Coordination Compounds
- Practice Problems
- Frequently Asked Questions - FAQs
Introduction to Addition Compounds
When an acid and base combines together it forms a salt. Generally, the salts are of two types:
- Simple Salt
- Addition Salt
Simple salt: A simple salt is produced when an acid is neutralised by a base. For example-
NaCl, NaNO3, NaHCO3, Na2CO3 etc.
A simple salt normally ionises in water and produces ions in the solution. The simple salt solution exhibits the properties of its constituent ions.
Let us consider an example in which acid and base react together to form salt and water.
When KCl is dissolved in water it water to form K+(aq) and Cl-(aq) ions.
Addition salt: It is also known as molecular salt. These are molecular compounds formed by the addition of two (or more) simple salts in stoichiometric proportions. For example: (potash alum), (Mohr’s salt), (potassium ferrocyanide), (potassium ferricyanide) etc.
Addition salts are further classified into two types:
Double salt: It is the type of salt which is formed by the addition of two or more molecular compounds combined in simple stoichiometric proportion and ionises completely when dissolved in an aqueous solution.
Note: It gives a test for all the ions, simple salt retains its identity and is stable in solid-state but loses its identity in aqueous solution.
For example:
In an aqueous solution:
Complex salt: It is the type of addition compound which does not ionise completely in an aqueous solution and dissociate into simple ions and complex ions.
Note: It does not give a test for all the ions. During the formation of complex salt, the simple salt loses its identity. It is stable in solid as well as in the aqueous solution.
For example-
In an aqueous solution:
The solution of , gives the identification test of K+ ions but does not give identification test because it can not dissociate into Fe2+ and CN- ions in an aqueous solution.
What is a Coordination Compound?
Coordination compounds are the compounds in which central atoms are linked with molecules and ions by coordinate covalent bonds. Another name for coordination compounds is coordination complexes. They are the addition compounds that retain their identity (do not lose it) in solutions.
In coordination compounds, usually, transition elements are the central atoms. In these compounds, the central atom is the centre of the coordination sphere/compound.
Important Terms
The coordination compound can be represented as:
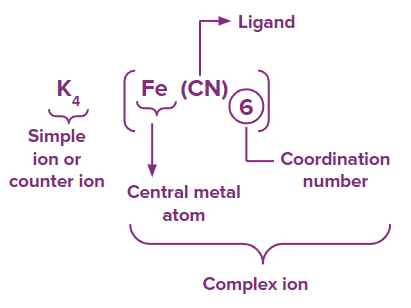
Coordination entity: A coordination entity is a complex compound in which the centre ion or atom (or coordination centre) is linked to a number of ions, atoms, or molecules.
In the above example is a coordination entity.
Central atom: As previously stated, the central atoms and ions are the atoms and ions with which a specific number of atoms, molecules, or ions are associated. In coordination compounds, the central atoms or ions are usually Lewis acids, which can act as electron-pair acceptors.
Ligands: Ligands are atoms, molecules, or ions that are bound to the coordination centre or the central atom/ion by a coordinate covalent bond.
These ligands can be simple ions or molecules such as -Cl , -NH3 & -CN or relatively large molecules such as ethane-1,2-diamine (-NH2-(CH2)2-NH2-).
Coordination number: The coordination number of the coordination complex refers to the total number of sigma bonds by which the ligands are bound to the coordination centre.
In the above example coordination number is 6 as six ligands are attached to the central atom ).
Coordination sphere: The non-ionizable fraction of a complex chemical is composed of a core transition metal ion surrounded by square bracketed atoms or groups. The coordination sphere is formed by the coordination centre, the ligands connected to the coordination centre, and the net charge of the chemical complex as a whole when written together. This coordination sphere frequently contains a counter ion (the ionizable groups that attach to charged coordination complexes).
Oxidation number: When all of the electron pairs provided by the ligands are removed from the central atom, the charge associated with it can be computed to determine its oxidation number.
For example, in case of -;
Assume the oxidation number of Fe be x.
So,
⇒ x=2
So, the oxidation number of Fe is +2.
Properties of Coordination Compounds
- The coordination compounds of transition metals are coloured. For example, the complexes of Iron (II) are green in colour, whereas complexes of iron (III) are brown in colour.
- The coordination compounds also have magnetic properties due to the unpaired electrons present in the central metal atoms.
- Coordination molecules' chemical reactivity varies. They can participate in electron transfer reactions in both the inner and outer spheres.
- Complex compounds containing specific ligands can act as catalysts.
Classification of Coordination Compounds
On the basis of types of ligand, coordination compounds are classified into two types:
- Homoleptic coordination compound: It is defined as the type of complex compound in which all the ligands are the same. For example- etc.
- Heteroleptic coordination compound: It is defined as the complex compound in which all the ligands are not the same. For example- etc.
On the basis of the stability of the compound, complex compound are classified into two types:
- Perfect compound: It is defined as the complex compound which is stable and does not dissociate completely in an aqueous solution. For example,
In an aqueous solution:
- Imperfect compound: It is defined as the complex compound which is unstable and dissociates completely like a double salt in an aqueous solution. For example- the complex of cadmium.
(unstable)
In an aqueous solution,
On the basis of the charge, the complex compound is classified into four different types:
- Cationic complexes: Coordination sphere forming a cation in the coordination complex. For example,
- Anionic complexes: Coordination sphere forming an anion in the coordination complex. For example,
- Neutral complexes: There are no cations or anions in this coordination sphere. For example,
- Cation and anion complex: Both cations and anions are coordination spheres. For example,
Classification of Ligands
Ligands are the atoms, ions, and molecules that surround the central transition metal ion. They act as Lewis bases, donating electron pairs to the transition metal ion, forming a dative bond between the ligands and the transition metal ion.
On the basis of the charge, ligands are classified into three types:
- Positively charged ligands: Those ligands which consist of a positive charge are known as positively charged ligands. For example- Hydrazinium (), nitrosonium (NO+), nitronium () etc.
- Negatively charged ligands: Those ligands which are negatively charged are known as negatively charged ligands. For example- etc.
- Neutral ligands: It is the type of ligand which does not contain any charge. For example: H2O, NH3 etc.
On the basis of denticity, ligands are classified into nine different types:
Denticity: It is defined as the number of lone pairs donated by the ligand to the central metal atom.
- Monodentate ligand: It is defined as the type of ligand which donates one lone pair to form one dative bond. For example ammine, aqua, carbonyl, nitrosyl etc.
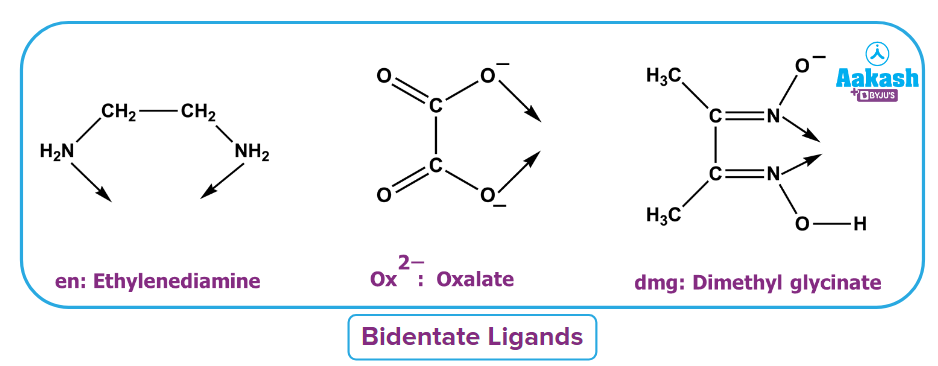
- Bidentate ligand: It is defined as the type of ligand which donates two lone pairs to form two dative bonds. For example, ethylenediamine, oxalate ion, dimethyl glycinate, acetylacetonato etc.
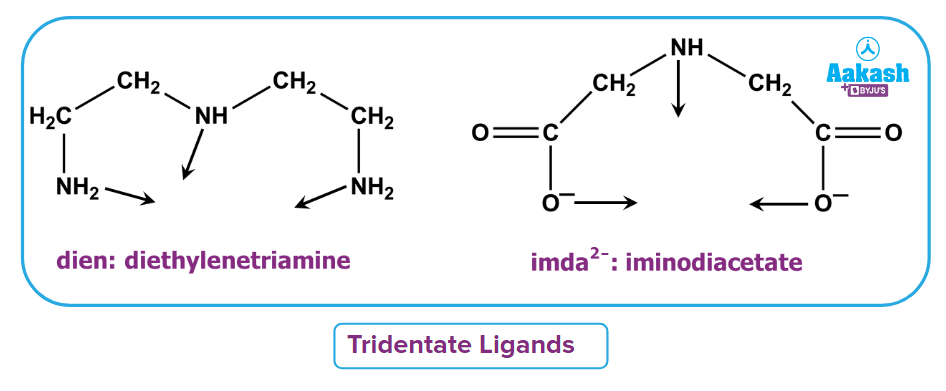
- Tridentate ligand: It is defined as the type of ligand which donates three lone pairs to form three dative bonds. For example, diethylene triamine, iminodiacetate ion etc.
- Tetradentate ligand: It is defined as the type of ligand which donates four lone pairs to form four dative bonds. For example- triethylene, tetraammine, nitrilotriacetate ion etc.
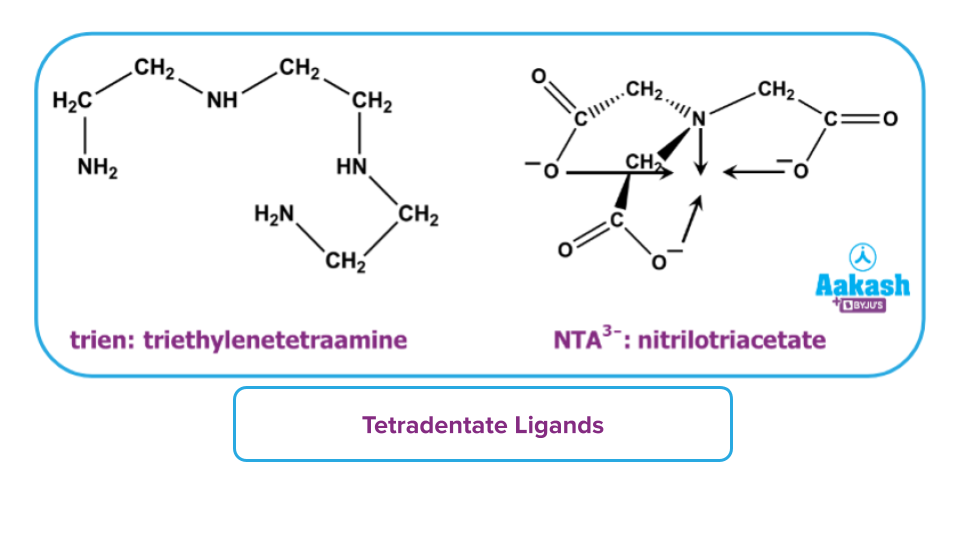
- Pentadentate ligand: It is defined as the type of ligand which donates five lone pairs to form five dative bonds. For example- ethylenediamine triacetate (EDTA)3-
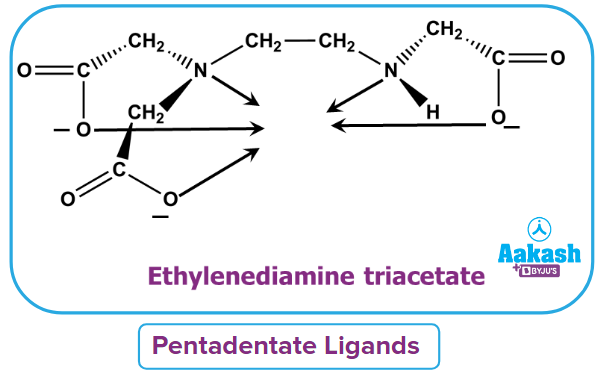
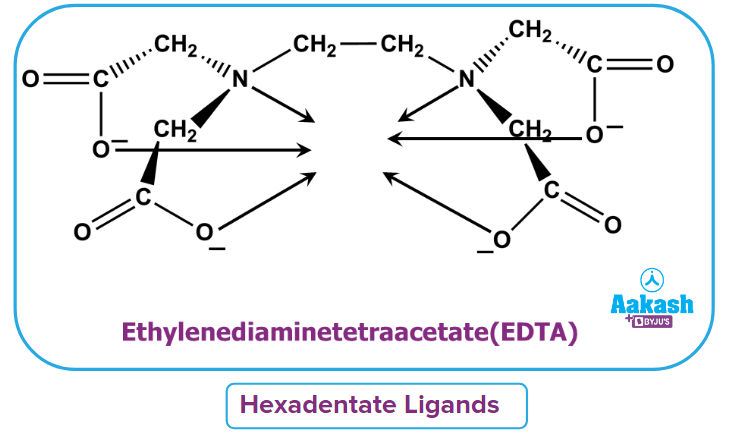
- Hexadentate ligand: It is defined as the type of ligand which donates six lone pairs to form six dative bonds. For example- ethylenediamine tetraacetate (EDTA)4-
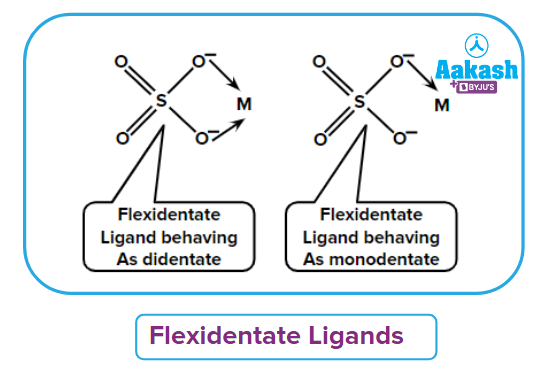
- Flexidentate ligand: It is defined as the ligands which have flexibility in their denticity. For example, (EDTA)4- can behave as hexadentate, pentadentate and tetradentate ligand. generally behave as a monodentate ligand but can also behave as bidentate ligand.
- Ambidentate ligand: A ligand which has two different donor sites but at a time donates only one lone pair is known as ambidentate ligand. For example:
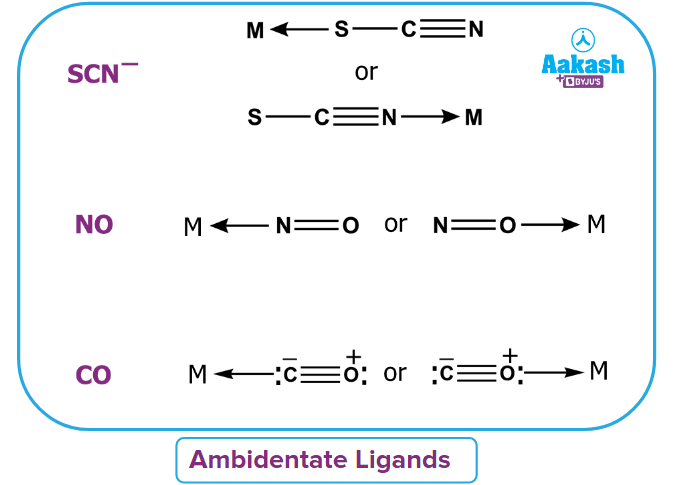
- Chelating ligands: It is defined as the ligands which have denticity of more than one and forms ring-like structure with the central metal atom is known as a chelating ligand.
IUPAC Nomenclature of Coordination Compounds
The conventional guidelines for coordination compound nomenclature are outlined below.
- In the IUPAC nomenclature of the coordination compounds, the central metal atom/ion is always written after the ligands.
- The names of the ligands are provided in an alphabetical order that is unaffected by the numerical prefixes that must be applied to the ligands when the coordination centre is attached to more than one.
- When a coordination molecule has a large number of monodentate ligands, the prefixes di-, tri-, tetra- etc are used to signify the number of ligands.
- The prefixes bis-, tris-, and so on are used when there are numerous polydentate ligands linked to the core metal ion.
- Anions in a coordination compound must have names that conclude with the letter 'o,' which usually replaces the letter 'e.' As a result, the nitrate anion must be written as 'nitrato' and the bromide anion to be written as 'bromido'.
- In coordination compounds, the neutral ligands NH3 (ammine), H2O (aqua or aquo), CO (carbonyl), and NO (nitrosyl) are given special names.
- The name of the core metal atom is written after the ligands have been named. The suffix '-ate' is used if the complex has an anionic charge connected with it.
- If the Latin name of the metal exists, it takes precedence when writing the name of the central metal atom in an anionic compound (with the exception of mercury).
- The oxidation state of the central metal atom/ion must be indicated using roman numerals wrapped in a pair of parentheses.
- The cationic entity must be written before the anionic entity if the coordination molecule is accompanied by a counter ion.
For example,
- Potassium hexacyanoferrate (II)
- triamminetriaquachromium (III) chloride
Isomerism in Coordination Compounds
Structural isomerism: Coordination compounds with the same molecular formula but a different arrangement of ligands around the central metal atom exhibit structural isomerism. These are further classified into five different types:
- Linkage isomerism: This isomerism is shown by the compounds which have the same molecular formula but contain an ambident ligand. For example,
- Ionisation isomerism: This isomerism is shown by the compounds which have the same molecular formula but give different ions in an aqueous solution as ions outside and inside the coordination complex is replaced.
For example- and
- Coordination isomerism: It is the type of isomerism shown by those compounds in which the cationic and anionic part both are complex in nature. For example- and
- Hydrated isomerism: Compounds differ based on the number of solvent molecules directly bonded to the metal ion in this special case of ionisation isomerism. For example-
Note: If there is no counter ion then it won't be able to hold the water molecule with the help of ion-dipole interaction and therefore the compound won’t exist. For example, cannot exist.
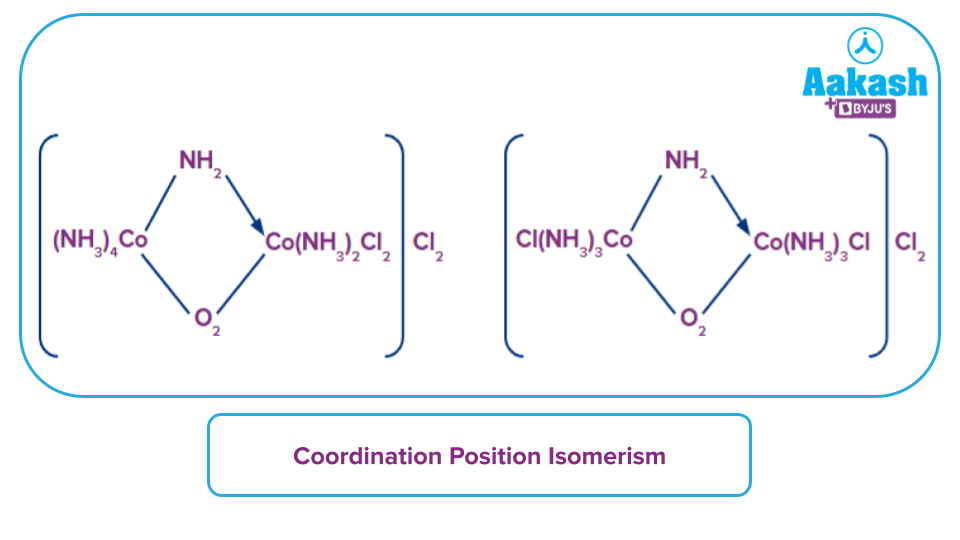
- Coordination position isomerism: It is the type of isomerism in which ligands change the position from one metal to another metal in the same coordination sphere. For example,
Stereoisomerism: Stereoisomers are coordination compounds that have the same chemical formula and chemical bonds but a different spatial arrangement of ligands around the central metal atom. There are two types of isomerism: optical isomerism and geometrical isomerism.
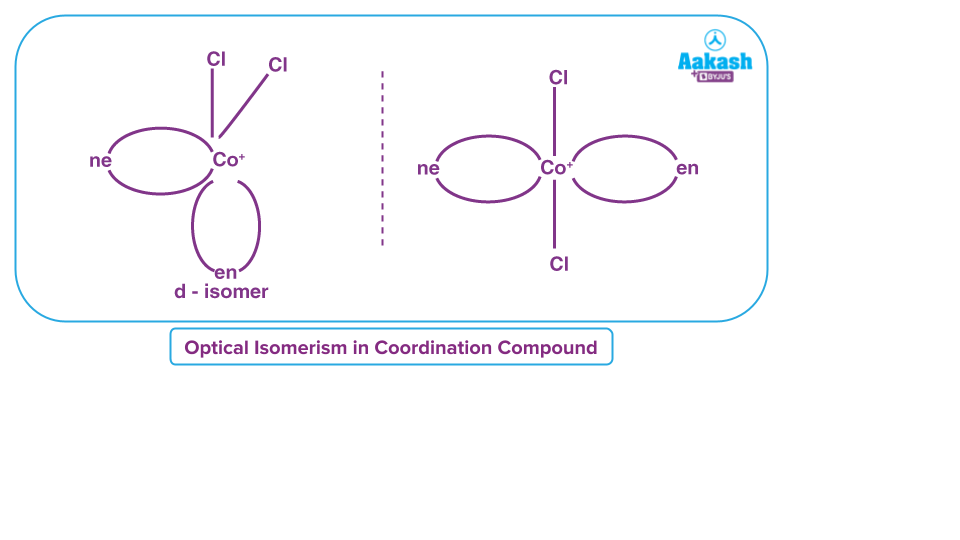
Optical Isomerism: Optical isomers are the isomers that rotate the plane polarised light to either a clockwise or anti-clockwise direction. There are two kinds of these.
The isomer that rotates plane-polarised light clockwise is known as the dextro, 'd,' or '+' isomer.
Laevo isomer is the isomer that rotates plane-polarised light anticlockwise. It is also known as ‘l’ isomer or ‘-’ isomer.
Optical isomers have non-superimposable mirror images.
Condition for optical isomerism:
- Compounds should rotate plane polarised light in any particular direction.
- Compounds should not contain any element of symmetry which includes (plane of symmetry, centre of symmetry and axis of symmetry)
- Mirror image of the compound should be non-superimposable in nature.
Geometrical isomer: Due to the various geometric arrangements of the ligands, geometrical isomerism is observed in heteroleptic complexes (complexes with more than one type of ligand). This behaviour is most noticeable in coordination compounds with coordination numbers between 4 and 6. Complexes with coordination number 4 with type ML4 do not exhibit geometric isomerism because the relative position of all the four ligands are similar with respect to each other and do not change.
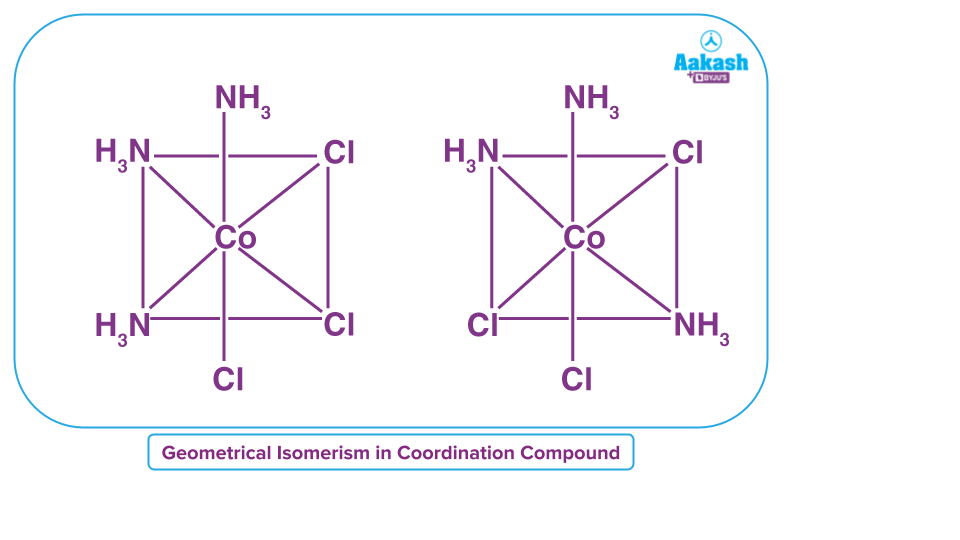
Werner’s Theory
Alfred Werner proposed Werner's theory of coordination compound structures in 1898.
Werner's Experiment: When AgNO3 (silver nitrate) was combined with CoCl3.6NH3, all three chloride ions were converted to AgCl (silver chloride). When AgNO3 was combined with CoCl3.5NH3, however, only two moles of AgCl were formed. Furthermore, when CoCl3.4NH3 was mixed with AgNO3, one mole of AgCl was formed.
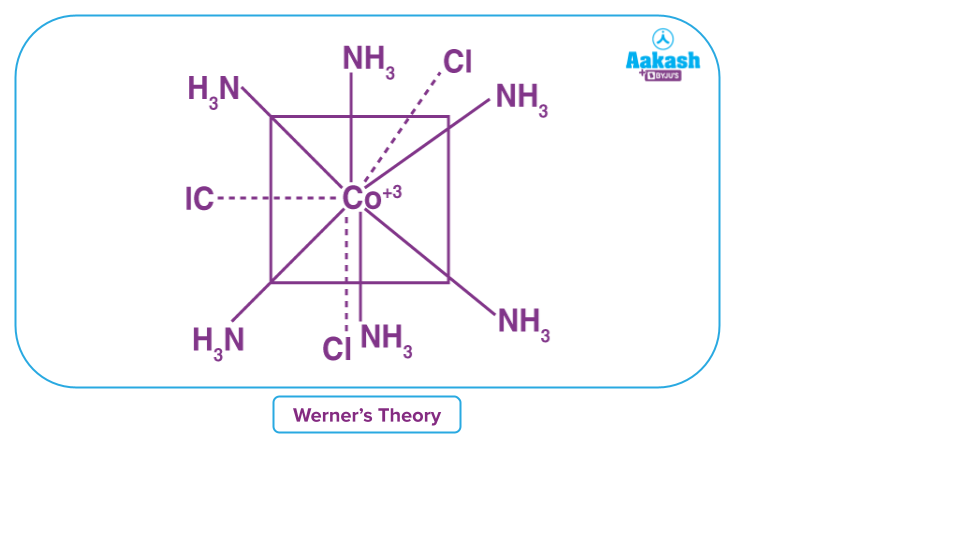
Based on this observation, Werner proposed the following postulates:
- The coordination compound's central metal atom has two types of valencies: primary and secondary linkages or valencies.
- Ionizable primary linkages are satisfied by negative ions.
- Primary valency is non-directional in nature and is equal to the oxidation state of the metal.
- Secondary valency cannot be ionisable. These are satisfied by ligands.
- Secondary valency is directional in nature and secondary valency of any metal is fixed and equal to the coordination number.
- Coordination polyhedra are the spatial arrangements of the metal atoms and ligands. Coordination complexes are the terms inside the square brackets, and counter-ions are the ions outside the square brackets.
Werner's Theory's Limitations:
- It fails to account for the magnetic, colour, and optical properties of coordination compounds.
- It did not explain why all elements do not form coordination compounds.
- It did not explain the directional properties of coordination bonds.
- This theory does not explain the complex's stability.
- The nature of complex ions could not be explained by this theory.
Magnetic Properties of Coordination Compounds
Due to the presence of unpaired electrons in the central metal atom, the coordinate complex shows paramagnetic properties (strongly attracted by the magnet). The complexes where there are no unpaired electrons in the central metal atom are diamagnetic (repelled in the presence of a magnet) in nature.
The formula given below is used to calculate the magnetic moment of the complex:
M = BM
BM stands for Bohr Magneton.
'M’ signifies the magnetic moment of the complex compound.
'n' signifies the number of unpaired electrons present in the metal atom.
Factors affecting the magnetic moment of the complex:
- Hybridization type.
- The central metal ion's oxidation state.
- The number of electrons that are unpaired.
Stability of Coordination Compounds
The complex is built up in different stages. Each step of the process is reversible, and the equilibrium constant is referred to as the stepwise formation constant. Consider the formation of a complex ML4.
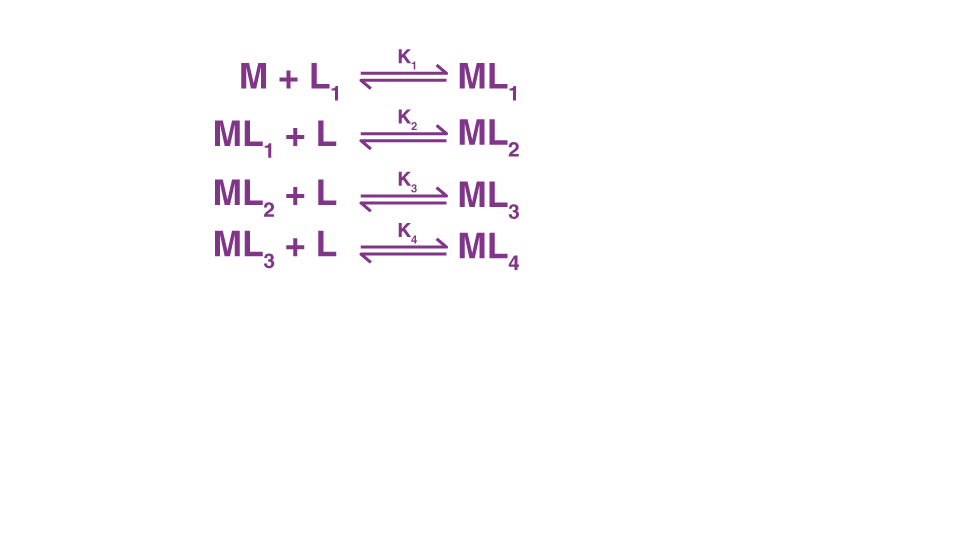
The overall formation constant, also known as the stability constant, is = and 1/ = Instability constant.
Here, '' represent the stability constant.
Factors influencing complex stability:
- The central transition metal ion should have a smaller size and a high nuclear charge for complex stability.
- The amount of crystal field stabilisation energy (CFSE) should increase.
- Chelating ligand-containing complexes are more stable.
- Octahedral complexes are more stable than tetrahedral complexes.
Colour of Coordination Compounds
Complexes with unpaired electrons in the central transition metal ion exhibit colour. The transition is known as 'd - d' transition. Other reasons for the transition metal to exhibit colourful compounds can be explained by polarisation and charge transfer spectra.
For example, the TiCl3 is violet colour compound due to d-d transition. Whereas is orange colour compound due to charge transfer spectra.
Factors affecting the colour of complexes are:
- Unpaired electrons in a transition metal ion.
- The composition of ligands.
- The central transition metal ion's oxidation state.
- Light wavelengths absorbed and emitted.
- The number of ligands present in the coordination sphere.
Application of Coordination Compounds
The unique properties of coordination compounds make them particularly useful in a wide range of processes and industries. Some of the applications of coordination compounds are listed below:
- Coordination compounds containing transition metals are widely used in industries for material colouration due to their colour. They are used in the dye and pigment industries.
- Some complex molecules with cyanide as a ligand are used in the electroplating process. These chemicals are also useful in the photography industry.
- Coordination complexes can be used to extract many metals from their ores. Nickel and cobalt, for example, can be extracted from ores using hydrometallurgical procedures.
- Water hardness is calculated using complexes of Ca++, Mg++, and EDTA.
- In medicine, cisplatin is used to treat cancer.
- In photography, film development is a complex formation.
- In metallurgy, the Mac Arthur process involves a complex of cyanide ions in the extraction of gold and silver.
Recommended Video Link: https://youtu.be/bDzsIVKEWIs
Practice Problems
Q1. NH2-NH2 is a _______ ligand.
A. Ambidentate
B. Monodentate
C. Chelating
D. All of the above is correct
Answer: (B)
Solution: Monodentate ligand is defined as the type of ligand which donates one lone pair to form one dative bond. NH2-NH2 is a monodentate ligand because one lone pair can be donated by the molecule to form a dative bond. The stability of the three-membered ring is very low due to structural constraints such as angle strain. Chelation is thus less efficient in this case if compared with four-membered (or higher) rings. As a result, it will not act as a chelating ligand. Therefore, option (B) is the correct answer.
Q2. From the given complexes, select the complex which does not give white ppt with AgNO3.
Answer: (D)
Solution: From the given complexes, will not form a precipitate when AgNO3 solution is added to it as there is no free chloride ion in the compound but in case of , , and when AgNO3 solution is added it will form 3 mol, 1 mol and 2mol of white AgCl precipitate respectively. So, the correct answer is option (D).
Q3. Which among the given pair of compounds represents the hydrated isomerism?
- and
- and
- and
Answer: (B)
Solution: Compounds differ based on the number of solvent molecules directly bonded to the metal ion in this special case of ionisation isomerism known as hydrate isomerism. and pair represent the hydrate isomerism as both the compound has the same molecular formula but there is a different number of solvent molecules directly bonded to the metal ion or is simply present in the crystal lattice as free solvent molecules. Therefore, option (B) is the correct answer.
Q4. Which among the given complex will not exhibit the geometrical isomerism?
- Both A and C is correct
Answer: (D)
Solution: Complexes with coordination number 4 with type ML4 does not exhibit geometric isomerism because the relative position of all the four ligands are similar with respect to each other and does not change. Therefore -and will not exhibit the geometrical isomerism and option (D) is correct. So, the correct answer is option (D).
Frequently Asked Questions-FAQs
Q1. What is the difference between primary and secondary valency?
Answer:
|
Primary Valency |
Secondary Valency |
|
It is equal to the oxidation state of the metal. |
It is equal to the coordination number of the metal. |
|
Primary valency is satisfied by the anions present in the complex compounds. |
Secondary valency is satisfied by the ligands present in the complex compounds. |
|
It is ionizable in nature (i.e. It can be dissociated into ions). |
It is non-ionizable in nature (i.e. it cannot be dissociated into ions) |
|
It is non-directional in nature and is represented by (………..) lines in a complex compound. |
It is direction in nature and is represented by the ( _______ ) lines in a complex compound. |
Q2. Why is given in case of lead poisoning?
Answer: In the case of lead poisoning is given because EDTA will form a soluble complex with lead and will be excreted out from the body.
Q3. What is a spectrochemical series?
Answer: A spectrochemical series is a list of ligands arranged in ascending order of ligand’s strength.It is an experimentally obtained series based on light absorption by complexes with various ligands. Ligands at the left end of this spectrochemical series are commonly considered to be weaker ligands and generally cannot pair the unpaired electron present in the orbital of the central metal atom. Whereas the ligands at the right end are generally stronger ligands that form inner orbital complexes after forceful pairing of electrons present in the orbital of the metal. The order of strength of ligands is given below:
Q4. What is the difference between d-d transition and charge transfer spectra?
Answer: d-d transition takes in a compound which has unpaired electrons then by absorbing visible light, electrons (e-) transfer from t2g to eg orbitals and when the electron return back from eg to t2g , it releases energy which lies in the visible region only and reflects their complementary colour. For example, in the case of TiCl3 which is violet in colour because it absorbs yellowish-green colour light.
Whereas there are some compounds in which unpaired electrons are absent but are still colourful due to the transfer of charge from anion to metal and is known as charge-transfer spectra. For example, is a orange colour compound due to charge transfer phenomena.


