-
Call Now
1800-102-2727
Concept of Equivalence - Equivalent Weight, Valency Factor, Practice Problems and FAQs
Let's imagine that you visited a factory that produces motorcycles in order to learn more about how they are created and what essential components go into each one.
Thrilling, isn't it?
Let's imagine a worker asked you how many wheels, seats, and frames are required to create a single motorcycle and handed you 4 wheels, 2 seats, and 2 frames.

Therefore, the correct response is that a motorcycle must have two wheels, a seat, and a frame.
Right??
Equivalence theory is comparable to what you said above. The concept of equivalency will provide you with more precise information about a motorcycle, including which pieces and how many of each are needed to complete it, whereas the concept of moles will only provide you with information about the motorcycle as a whole and not the specifics.
Table of contents
- Concept of Equivalence
- Valency factor or n-factor
- Practice problems
- Frequently asked questions-FAQs
Concept of Equivalence:
An equivalent concept is the "gram equivalent weight" or "gram equivalent" concept. It is based on the concept of equivalent weight.
The equivalent weight of an element is the number of parts by mass of that element that reacts with or displaces from a combination, such as 1.008 parts by mass of hydrogen, 8 parts by mass of oxygen, or 35.5 parts by mass of chlorine.
Equivalent weight of Al for its oxide formation :
From the above equation we can say,
316 g of oxygen combined with 227 g of Aluminium
1 g of oxygen combined with 5448 g of Aluminium
8 g of oxygen combined with of Aluminium
Equivalent weight of Aluminium = 9 g
We can define formulas for acid, base, salt and ions for a better understanding.
|
Different Components |
Formulas |
|
Elements or ions |
Equivalent weight = |
|
Acids |
Equivalent weight = |
|
Bases |
Equivalent weight = |
|
Equations and in general |
Equivalent weight = |
Valency factor or n-factor:
In redox reactions, the n-factor or valency factor is used to describe the number of electrons gained or lost per atom during a reaction.
For Acids:
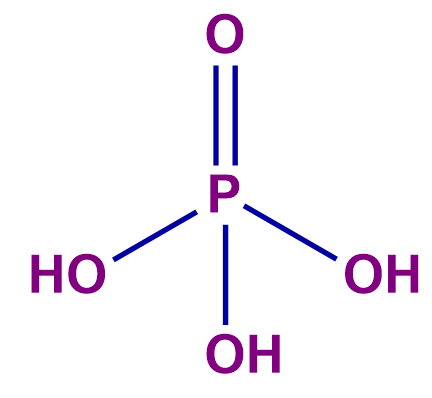
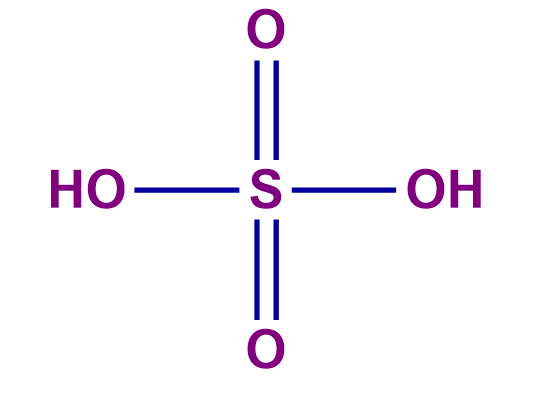
n-factor of acid = number of H+ ions released per molecule of an acid which is also known as Basicity.
|
Acid |
n-factor |
Structure |
|
number of H+ ions = 1 (n-factor) |
|
|
|
number of H+ ions = 3 (n-factor) |
|
|
|
number of H+ ions = 2 (n-factor) |
|
|
|
number of H+ ions = 2 (n-factor) |
|
|
|
number of H+ ions = 1 (n-factor) |
|
|
|
number of H+ ions = 1 (n-factor) |
|
For Bases:
n-factor of base = number of OH- ions released per molecule of base and also known as acidity.
|
Base |
n-factor |
|
number of OH- ions = 1 (n-factor) |
|
|
number of OH- ions = 2 (n-factor) |
|
|
number of OH- ions = 3 (n-factor) |
For salts:
n-factor of salt = total charge present on an ion
|
Salt |
n-factor |
|
charge present on ion = 1 (n-factor) |
|
|
charge present on ion = 2 (n-factor) |
|
|
charge present on ion = 6 (n-factor) |
For redox reactions:
In redox reactions, the n-factor is calculated based on the change in oxidation state.
Case 1: Oxidation: SO32-SO42-
|
Element |
Initial O.S |
Final O.S |
|
S |
+4 |
+6 |
|
O |
-2 |
-2 |
Oxidation state of Sulfur changes from +4 to +6
Case 2: Reduction: MnO4-Mn2+
|
Element |
Initial O.S |
Final O.S |
|
Mn |
+7 |
+2 |
|
O |
-2 |
-2 |
Oxidation state of Manganese changes from +7 to +2
Case 3: If an atom from a chemical species undergoes either oxidation or only reduction and then appears in two different products in two different oxidation states.
Example: MnO4-Mn2++MnO2
|
Element |
Initial O.S |
Final O.S |
|
Mn |
+7 |
+2 for Mn2+ & +4 in MnO2 |
|
O |
-2 |
-2 |
In a balanced equation: 2MnO4-+12H++8e-Mn2++MnO2+6H2O
Case 4: When two or more atoms are oxidized as a result of oxidation.
|
Element |
Initial O.S |
Final O.S |
|
Cr |
+2 |
+3 |
|
C |
+3 |
+4 |
Case 5: When two or more atoms are oxidized or reduced as a result of either oxidation or reduction.
NaNO3NaNO2+12O2
|
Element |
Initial O.S |
Final O.S |
|
N |
+5 |
+3 |
|
O |
-2 |
0 |
n-factor for oxidation = 1|0-(-2)|=2 (only one oxygen get oxidized)
n-factor for reduction = 1|5-3|=2
You can consider n-factor by oxidation or reduction of any one of them.
Case 6: Disproportionation reaction; in this reaction, one type of atom gets oxidized as well as reduced.
Type-1: When the number of electrons gained or lost is the same.
|
Element |
Initial O.S |
Final O.S |
|
N |
+4 |
+5 in HNO3 & +3 in HNO2 |
Reduction: NO2HNO2
Oxidation: NO2HNO3
Type-2: When the number of electrons gained or lost is different.
|
Element |
Initial O.S |
Final O.S |
|
Br |
0 |
-1 in Br- & +5 in BrO3- |
Reduction: Br2Br-
Oxidation:
Related Topic Link: Some Basic Concepts of Chemistry - What is Equivalent Concept? | JEE | NEET | Aakash EduTV
Practice problems:
Q 1. Calculate the n-factor for the given reaction-
Answer: (D)
Solution:
|
Element |
Initial O.S |
Final O.S |
|
Cl |
0 |
-1 in Cl- & +5 in ClO3- |
Reduction: Cl2Cl-
Oxidation:
Hence, the correct answer is option (D).
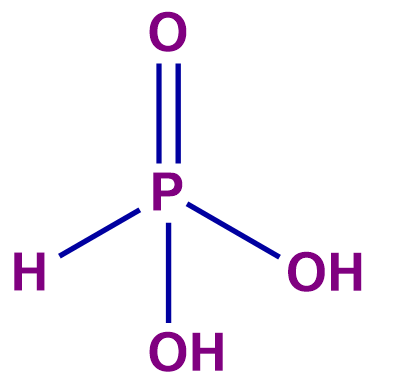
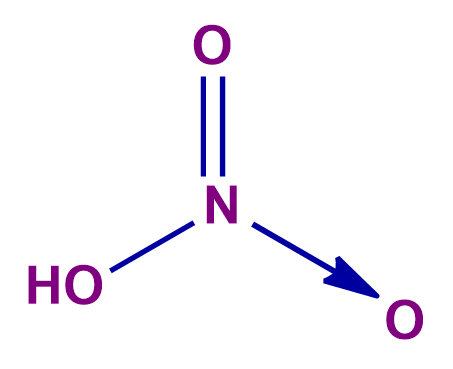
Q 2. Calculate the equivalent weight of HNO2 for reaction ; if the molar mass of HNO2 is M.
- None of these
Answer: (C)
Solution:
|
Element |
Initial O.S |
Final O.S |
|
N |
+3 |
+5 in HNO3 and +2 in NO |
Reduction: HNO2HNO3
Oxidation: HNO2NO
Equivalent weight of
Hence, the correct answer is option (C).
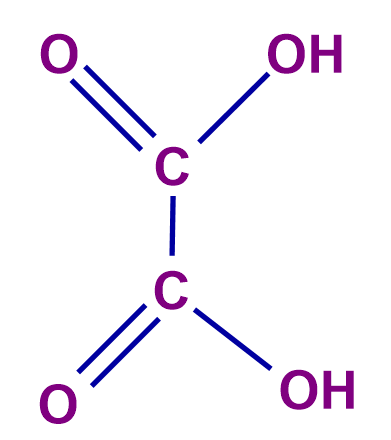
Q 3. Calculate the equivalent weight for oxalic acid.
- 45 g
- 90 g
- 30 g
- None of these
Answer: (A)
Solution:
|
Acid |
n-factor |
Structure |
|
number of H+ ions = 2 (n-factor) |
|
Molecular Mass of Oxalic acid = 90 g
Equivalent weight of Oxalic acid =
Q 4. Calculate n-factor for the given reaction
- 1
- 6
- 3
- 4
Answer: (B)
Solution:
When two or more atoms oxidize or reduce as a result of oxidation or reduction.
|
Element |
Initial O.S |
Final O.S |
|
Br |
+5 |
-1 |
|
O |
-2 |
0 |
You can consider n-factor by oxidation or reduction.
Hence, the correct answer is option (B).
Frequently asked questions-FAQs:
1. What exactly is normality?
Answer: Normality is a concentration measure that equals the gram equivalent weight of solute per liter of solution. Gram equivalent weight is a measure of a molecule's reactive capacity. The normality unit is eq/L. The letter "N" represents normality.
Normality=Number of gram equivalentsVolume of solution (mL)1000
2. Why does normality matter more than molarity?
Answer: While molarity refers to the concentration of a compound or ion in a solution, normality refers to the molar concentration of the acid or base components of solution only. Thus, normality provides a more comprehensive understanding of solution concentration in acid-base reactions.
3. What is the distinction between equivalent and molecular weight?
Answer: The key distinction between gram molecular weight and gram equivalent weight is that the former refers to the mass of a molecule in gram, which is numerically equal to its molecular weight, whereas the latter refers to the mass of one equivalent in gram. Equivalent weight is basically concerned with the valency of the atom or ion.
4. Are there any conditions when equivalent weight and molecular weight become equal to each other?
Answer: If we can analyze the relation between equivalent weight and molecular weight, it says that
Now, if in any the value of n-factor become equals to 1, then equivalent weight becomes equal to molecular weight.
If, n-factor=1
Equivalent weight=Molecular weight