-
Call Now
1800-102-2727
Column Chromatography – Principle, Elution, Procedure, Types, Applications, Practice Problems and FAQ
Have you ever had a chance to take a glance at the vast, infinite fields of flowers like the tulip gardens or the rose fields in Gulmarg? It truly gives your heart a scope to expand your imaginations for a while on– “What if life would have been a bed of roses!!”
Coming back to reality now! Imagine a mixed swarm of honeybees and wasps flying from atop this bed of flowers. It is difficult to physically separate or distinguish these groups!

The wasps would not be attracted to the flowers, and they would fly past the flower bed soon enough to reach the end of the field. The wasps would fly ahead of the bees if one were to view from a spot beyond the flowerbed. Hence, the wasps would become separated from the bees. This is symbolic to what exactly happens in column chromatography, or in fact in chromatography at large!
Chromatography is a separation technique used to separate various analytes from a sample mixture.
In this example, the analytes to be separated are represented by the bees and wasps, the stationary phase is symbolised by the flowers, and the mobile phase is represented by the air. The different affinities between the analyte, stationary phase, and mobile phase are the key to the separation.
So, let's take a deeper insight into this wonderful separation technique called column chromatography!
TABLE OF CONTENTS
- What is Chromatography?
- Column Chromatography – Introduction
- Column Chromatography – Principle
- Column Chromatography – Elution
- Column Chromatography – Preparation & Instrumentation
- Column Chromatography – Stationary Phase
- Column Chromatography – Mobile Phase
- Column Chromatography – Experiment & Procedure
- Column Chromatography – Types
- Column Chromatography – Applications
- Column Chromatography – Disadvantages
- Practice Problems
- Frequently Asked Questions – FAQ
What is Chromatography?
Chromatography is a laboratory method used in chemical analysis to separate a mixture into its constituent parts. The mixture is dissolved in a liquid solvent known as the mobile phase, which transports it through a system where a substance known as the stationary phase is fixed. While the stationary phase can either be a solid or a liquid, the mobile phase can either be a liquid or a gas.

The separation of components of a mixture takes place on the basis of differential adsorption on the adsorption column. After the separation, the substances are extracted from the adsorbent using a suitable solvent which is called eluent.
Adsorption is the phenomenon of attracting and retaining the molecules of a substance on the surface (adsorbent) of a liquid or solid, resulting in a higher concentration of the molecules on the surface.
Column Chromatography – Introduction
In column chromatography, substances are separated into fractions based on the differential adsorption of the compounds to the adsorbent as the compounds pass along the column at varying speeds. Purifying materials that can be utilised in upcoming research can be done on a large-scale or small-scale using this approach. This technique is a subset of adsorption chromatography.
The method is applicable at scales ranging from micrograms to kilograms. The fundamental benefit of column chromatography is the stationary phase, which is inexpensive and easily disposed of after usage. The latter stops recycling-induced stationary phase deterioration and cross-contamination. Both gravity and compressed gas can be used in column chromatography to force the solvent through the column.

Column Chromatography – Principle
- The stationary phase is tightly packed inside a glass or metal column during column chromatography.
- The mobile phase, also known as the eluent, is then transported through the column either by using a pumping system or by applying gas pressure once the combination of analytes has been added.
- The stationary phase is either placed as a thin film on the interior of the column wall or coated onto discrete tiny particles (the matrix) and packed into the column.
- The analytes segregate based on their distribution coefficients as the eluent passes through the column and emerges separately in the eluate as it exits the column.
- Different parts of the mixture move at varying speeds when the mobile phase and the mixture that needs to be separated are introduced from the top of the column.
- Compared to components with greater adsorption and affinity to the stationary phase, those with lower adsorption and affinity move more quickly and are first removed from the column.
- The elements that move quickly (low adsorption) are eliminated first, while the elements that move slowly (higher adsorption) are eliminated last.
The reversible adsorption of solute molecules to the column takes place. In column chromatography, the analytes' movement rate is stated as follows using the parameter RF, which is commonly known as the retention factor and is inversely proportional to the retardation factor.
RF= Amount of Time the analyte was retained in stationary phaseAmount of Time the analyte is retained in mobile phase
Column Chromatography – Elution
Elution is a chemical process that includes exchanging ions with another substance to remove the ions from a substance. It is the chromatographic process of utilising a solvent to extract an adsorbate from a solid adsorbing medium. The solvent or mobile phase that travels through the column is known as the eluent. The molecules in the sample desorb from the adsorbent and dissolve in the eluent when the polarity of the eluent matches the polarity of the molecules in the sample.
Eluent is the term for the portion of the mobile phase that carries the components of the sample. Eluate refers to the combination of solute and solvent that leaves the column. The mobile phase and analytes make up the eluate. a material that separates and pushes mixture components through a chromatograph's column. In gas chromatography, the eluent is a carrier gas, in liquid chromatography, the eluent is a liquid solvent.
Column Chromatography – Preparation & Instrumentation
- The column is prepared by putting a solid adsorbent inside of a glass or plastic tube with a cylindrical shape. The quantity of the substance that is being separated will determine the size.
- To hold the solid (stationary) phase in place, the base of the tube includes a filter, a cotton or glass wool stopper, or a glass frit. The top of the column may have a solvent reservoir attached to it.
- The dry method and the wet approach are typically employed to prepare columns.
- For the dry technique, dry stationary phase powder is added to the column first. Next, a mobile phase is added, which is then flushed through the column until it is entirely wet and is never allowed to run dry beyond this point.
- For the wet technique, the stationary phase powder and eluent are mixed into a slurry, which is then gently put into the column.
- The top of the silica should be level, and it might be covered with sand to prevent damage. To move the organic material along in the column, the eluent is gradually fed through it.
- Columns are typically 25-50 cm long, 4 mm in diameter, and made of stainless steel for liquid chromatography, whereas they are 1-3 m long, 2-4 mm in diameter, and either made of glass or stainless steel for gas chromatography.
- Either the traditional type, which is filled with the stationary phase or the microbore type, which coats the stationary phase directly on the inside wall of the column, is acceptable.
- To deliver the sample for testing, an injector system is used.
- While moving through the column with the eluent at various speeds, the distinct components are held by the stationary phase differently and apart from one another.
- They elute one at a time towards the column's end. The eluent is collected during the entire chromatography procedure in a number of fractions.
- By using fraction collectors, fractions may be automatically gathered.

Detectors: Running many columns at once will boost chromatography's productivity. Multi-stream collectors are employed in this situation. Each fraction is examined for dissolved chemicals using analytical chromatography, UV absorption spectra, and fluorescence to monitor the effluent flow's composition. Through the glass wall, coloured compounds may be seen as moving bands (or fluorescent compounds with the help of a UV light).
A peak of chart readout in the form of a recorder is present for representing separated analytes.
Column Chromatography – Stationary Phase
The stationary phase is a solid that must fulfil the following criteria and have strong adsorption properties.
- Particle size and shape: Particles should be between 60-200 μ in diameter and have a consistent shape.
- The particles in the stationary phase should be mechanically stable and inert.
- It should be affordable, readily available, and colourless.
- It should allow the mobile phase to move freely.
- It must be capable of separating combinations of different substances.
- In column chromatography, the stationary phase or adsorbent is solid. Silica gel is the most used stationary phase for column chromatography, followed by alumina. In the past, cellulose powder has frequently been employed.
Column Chromatography – Mobile Phase

Composed of solvents or a combination of solvents, the mobile phase or eluent is utilised to transport the chemicals across the column. To cut down on the time and eluent required to conduct the chromatography, it is chosen such that the retention factor value of the target chemical is about between 0.2 and 0.3.
This phase serves the following purposes:
- It functions as a sample-solvent combination that may be added to the column.
- It serves as a developing agent and aids in the sample's components being separated into bands.
- It serves as an eluting agent, removing the separated components from the column during the experiment.
- Based on their polarity, some solvents used as mobile phases include ethanol, acetone, water, acetic acid, pyridine, etc.
Column Chromatography – Experiment & Procedure
As the top level of the mobile phase and the stationary phase should coincide, the solvent is used to moisten the stationary phase.
Solvents or a combination of solvents make up the mobile phase or eluent. Without disturbing the top level, the compound mixture that has to be separated is introduced in the first step from the top of the column. The adsorption process starts on the silica surface as soon as the tap is opened.
The stationary phase solvent mixture is progressively introduced by lightly contacting the glass column's sides. As needed, the solvent is added during the experiment.
To start the movement of the compounds in the mixture, the tap is turned on. The polarity of the molecules in the sample is what causes the movement. When compared to the polar components, the non-polar components move more quickly.
For instance, if a compound combination contains the colours red, blue, and green, and the order of the compounds according to polarity is blue>red>green, the green compound will move first since it has a lower polarity. It is collected in a sterile test tube when it reaches the column's end. The red compound is then collected after which the blue compound is collected. These are gathered in several test tubes.
Column Chromatography – Types
- Adsorption Column Chromatography - Adsorption chromatography is a separation technique where the mixture's components are adsorbed on the surface of the adsorbent.
- Partition Column Chromatography - In this method, both the stationary phase and the mobile phase are liquids.
- Size Exclusion Chromatography - This process is also known as molecular sieve chromatography. The molecules in a solution are sorted according to their size and, in certain circumstances, molecular weight. Proteins and commercial polymers and macromolecular complexes use this technique to be separated.
- Gel Column chromatography - With this type of chromatography, the separation is accomplished using a gel-filled column. A solvent is used to hold the stationary phase in the solvent's space.
- Ion Exchange Column Chromatography - It is a type of chromatography in which ion exchange resin is always used as the stationary phase. Using this technique, polar molecules and ions are divided according to how well they adhere to the ion exchanger. It operates on nearly all types of charged molecules. To separate charged biological components like proteins, peptides, amino acids, or nucleotides, ion exchange chromatography is frequently utilised.
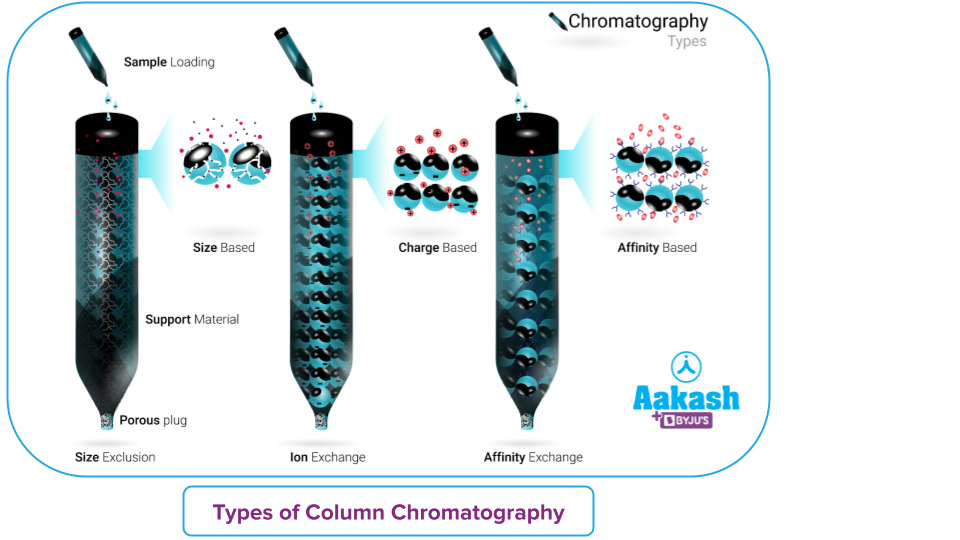
- Affinity Chromatography - An immobilised ligand and its binding partner engage in a particular binding interaction, which forms the basis of the separation technique known as affinity chromatography. The strong affinity of a ligand immobilised on a stationary phase with one or more molecules in a sample is the foundation of affinity column chromatography. Reversible interaction exists between the binding partners.
Examples include interactions between an enzyme and a substrate, an enzyme and an inhibitor.
- Gas Chromatography (GC) - An immensely useful technique this is. By injecting a gaseous or liquid sample into a mobile phase, which is sometimes referred to as the carrier gas and passing the gas through a stationary phase (in the column), gas chromatography is a method for separating chemicals in mixtures. Inert gas or an unreactive gas, such as helium, argon, nitrogen, or hydrogen, often makes up the mobile phase.
Chemicals that can be vaporised without decomposing are used to separate and analyse other compounds in analytical chemistry.
Column Chromatography – Applications
- It is used in the separation of a chemical mixture.
- It is used in the purification or the process of removing contaminants.
- It is used in the isolation of the active elements.
- It is used in removing metabolites from bodily fluids.
- It is used in the estimation of pharmaceutical compounds or crude extracts.
- It is used immensely in food and beverage testing, and testing for adulteration.
- It is used in drug purification and analytical testing
- It is used in forensic testing, and as a crime detection tool.
Column Chromatography – Disadvantages
- The compounds must be separated more slowly.
- In comparison to more sophisticated separation methods, column chromatography has a poor separation power/efficiency.
- Solvents must be used in larger, more expensive volumes.
- The automated method becomes expensive and complex.
Practice Problems
- Silica gel used in column chromatography is used as a/an
- Eluent
- Mobile Phase
- Stationary Phase
- Column
Answer: C)
Solution: In column chromatography, the stationary phase or adsorbent is solid. Silica gel is the most used stationary phase for column chromatography, followed by alumina. In the past, cellulose powder has frequently been employed.
So, option C) is the correct answer.
- In column chromatography, the analytes of a mixture are eluted in which of the following orders?
- Increasing polarity
- Decrease in polarity
- Not dependent on polarity
- Molecular structure
Answer: A)
Solution: In column chromatography, the analytes of a mixture are eluted in order of increasing polarity. The degree of polarity in a solvent as a whole, the polarity of the stationary phase, and the make-up of the sample all affect how well it can elute.
So, option A) is the correct answer.
- Column Chromatography is based on the principle of
- Sieving
- Absorption
- Differential Adsorption
- van der Waals force
Answer: C)
Solution: In column chromatography, substances are separated into fractions based on the differential adsorption of the compounds to the adsorbent as the compounds pass along the column at varying speeds. Purifying materials that can be utilised in upcoming research can be done on a large-scale or small-scale using this approach. This technique is a subset of adsorption chromatography.
So, option C) is the correct answer.
- Ion-exchange chromatography uses what kind of stationary phase?
Solution: A cross-linked polymer resin, typically divinylbenzene cross-linked polystyrene, with covalently connected ionic functional groups serves as the stationary phase in ion-exchange chromatography (IEC). This polymeric resin is applied to a column after being slurried with water. The inorganic salt is dissolved in a suitable solvent and is part of the mobile phase, which is applied to the column.
Frequently Asked Questions – FAQ
1. What are the advantages of column chromatography?
Answer: The stationary phase is less costly and easier to dispose of since it is recycled, which is the major benefit of this chromatography process. Also, the procedure entails separating almost any combination, contaminants from any combination are separated. Besides that, any amount or bulk of separation is feasible. The procedure is simple to grasp and entails little expense and any type of solvent can be used in the procedure to get the desired outcome. Big laboratories automate the entire procedure to a great extent.
2. Which components come out of the column first (as eluent)?
Answer: Non-polar compounds come out first or are eluted first. The more polar chemicals are absorbed more strongly because the adsorbents are polar. As a result, non-polar substances are eluted first. In contrast to non-polar molecules, polar compounds will strongly adsorb onto silica.
3. What quantity of silica should be packed in a column?
Answer: Silica is often the go-to sorbent for generally non-polar chemicals. The size and homogeneity of the silica grains will determine how much silica is required. The silica to compound ratio is taken into consideration for determining the amount of silica needed.
The most commonly used silica grain size in column chromatography is around 40 μ- 63 μ. The exact amount of silica needed is variable for the types of mixtures used. It is calculated based on the estimated silica to compound weight ratio. The most common method for determining the ratio of silica to a compound is to use the estimated weight of the mixture's most abundant component.
For example, let there be a mixture of 2 g of A and 3 g of B. The silica desirable (for adsorption) is 50:1 (which means just one compound is adsorbed and the other goes away with eluent), the optimum amount of silica needed would be (350)= 150 g and not (3+2)50= 250 g.
4. Mention the reason that a column in column chromatography should never run dry?
Answer: If you allow the column to become completely dry, the silica will start to break and the compounds won't be separated well. Never allow the solvent level in the column to go below the silica gel level while running it to avoid getting subpar results. As a result, the column must stay moist after the solvent has been applied. Never let the column dry; instead, keep adding solvent to it. The separation will be uneven if the column dries up, producing unreliable findings.


