-
Call Now
1800-102-2727
Ethers: Classification, Nomenclature, Preparations, Practice Problems and Frequently Asked Questions
Because of their olfactory and organoleptic properties, ethers are widely used in the perfumery and aroma industry.fragrance & scent industry They add flavor to a wide range of foods and medications, including fruit-based foods and beverages, desserts, fruit jams, yogurts, ice cream, chewing gum, and some pharmaceutical preparations.desserts,fruit jams,yogurts,ice cream,chewing gum,fruit based foods and beverages, and some pharmaceutical preparations Consider the following examples:
Some new ether compounds, such as diethyl ether, are particularly useful in the preparation of perfumes or perfumed articles,in the making of fragrances or fragrant items such as soaps, detergents, and everyday household materials such as essential oils, medicines, and so on.
1-methoxy-3-hexanethiol imparts grapefruit flavor, giving beverages a fruity-green, exotic flavor.
1-methoxy-3-hexanethiol adds grapefruit taste to beverages, giving them a fruity-green, exotic flavor.
Some ethers are also used to impart salty aromas, particularly meat-like aromas, to food such as soups and broths, seasonings, snacks, sauces, or ready-to-eat non-veg meals.
Methoxy benzene (anisole) is a common constituent used to impart fragrance to anise seed essential oil.
Table of content:
- Introduction of ether
- Classification of ethers
- Nomenclature of ethers
- Preparations of ethers
- Practice problems
- Frequently asked questions
Introduction of ethers:
An ether is a type of organic compound that mainly contains an ether group in which the oxygen atom is bonded to two alkyl or aryl groups. The word Ether is derived from the Latin word "ether" which means "to burn". At room temperature and under high pressure,Under high pressure and at room temperature ethers are usually flammable. The general formula of the ether is given by ROR, ROR', ROAr or ArOAr where R represents an alkyl group and Ar represents an aryl group.
Ether is a Latin word. The meaning of this word according to latin is “to burn”. Meaning of this word was assigned as Ethers can catch fire (flammable) under normal room temperature (298 K) and high pressure.
General formula of ether :
ROR, ArOAr, ROR', ROAr
Where R= alkyl group
Ar=Aryl group
We often come across this topic in organic chemistry and it is also a widely covered topic in biochemistry, where we find common bonds between carbohydrates and lignin.
We frequently encounter this subject in organic chemistry, and it is also a subject that is extensively covered in biochemistry. Here, we discover that lignin and carbohydrates share common bonds. Whereas if we look at the structure of the ethers, they basically have bent C-O-C bonds.
Classification of ethers:
Ethers can be classified into two broad categories based on the substituent group attached to its oxygen atom: symmetrical ethers and asymmetrical ethers.
1. Symmetrical ethers or simple ethers: These ethers have the same alkyl or aryl groups attached on either side of the -O- functional group.
Example: C2H5 - O - C2H5 (diethyl ether), CH3 - O - CH3 (dimethyl ether)
2. Unsymmetrical ethers or mixed ethers: These ethers have different alkyl or aryl groups attached on either side of the -O- functional group.
Example: C2H5 - O - CH3 (ethyl methyl ether), CH3 - O - C6H5 (methyl phenyl ether).
Nomenclature of ethers:
The ether is named simply by the name of the two alkyl/aryl groups bonded to the oxygen and the addition of the word "ether". These alkyl groups are listed in alphabetical order.
For example, t-butyl methyl ether, ethyl methyl ether
If only one alkyl/aryl group is given in the name of a particular ether, it implies two identical groups, as in the ethyl ether for the diethyl ether. The ether naming adopted by IUPAC uses a more complex group as the parent name, with the oxygen atom and the smaller named group as the alkoxy substituent. Thus, in the IUPAC system, ethers are alkoxy alkanes.
Examples: ethoxyethane (diethyl ether), methoxyethane (ethyl methyl ether), 2- methoxy 2- methylpropane (methyl tert-butyl ether), phenoxybenzene (diphenyl ether).
Preparations of ether:
1. Dehydration of alcohols:
Alcohols undergo dehydration in the presence of protic acids (sulfuric acid, phosphoric acid) to yield alkenes and ethers under different conditions. The formation of reaction products depends on the reaction conditions.
Under various circumstances, alcohols dehydrate in the presence of protic acids (sulfuric acid, phosphoric acid) to produce alkenes and ethers. The conditions where the reaction was carried out, determine how reaction products are formed.
For example, ethanol is dehydrated to ethylene at 443 K in the presence of sulfuric acid. On the other hand, ethanol gives ethoxyethane in the presence of sulfuric acid at 413 K.

2. Intermolecular dehydration of alcohol:
The formation of ether by intermolecular dehydration of alcohol occurs by an SN2mechanism.
Mechanism:
Step-1: Protonation of alcohol

Step-2: Attack of nucleophile
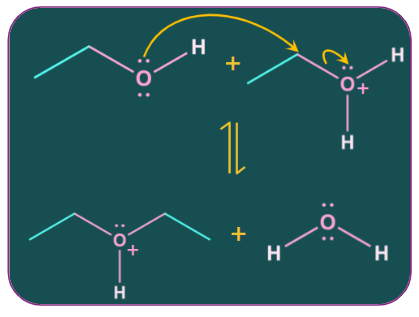
Step-3: Deprotonation

Limitations of intermolecular dehydration:
- Synthesis of ethers using secondary alcohol is usually unsuccessful because alkenes are formed very easily.
- Synthesis of ethers with tertiary alkyl groups lead exclusively for the formation of alkenes.
- Not useful for the preparations of unsymmetrical ethers from primary alcohols because reaction leads to formation of mixture of products.
3. Williamson ether synthesis:
This is an important method for the preparation of symmetric and asymmetric ethers in the laboratory. In the Williamson synthesis, an alkyl halide is reacted with a sodium alkoxide, resulting in the formation of an ether.
In the Williamson synthesis, an ether is created when an alkyl halide reacts with a sodium alkoxide.
This reaction involves the SN2 attack of an alkoxide ion on an alkyl halide. We know that alkoxides are very strong bases and that they frequently react with alkyl halides and thus participate in elimination reactions.
The substrate must be unhindered (1o or 2o alkyl halide) and bear a good leaving group.
In the case of the primary alkyl halides, the Williamson synthesis shows a higher yield.
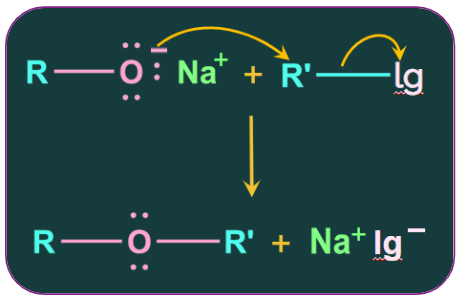
4. Reaction of alkyl halide with dry silver oxide:
When alkyl halide is treated with dry silver oxide, ether is produced
2 R - X + Ag2O → R - O - R + 2 AgX
Practice problems:
Q1. The formation of dimethyl ether from methanol is based on:
(A) Dehydrogenation reaction
(B) Hydrogenation reaction
(C) Dehydration reaction
(D) Heterolytic fission reaction
Answer: (C)
Solution: Formation of dimethyl ether from methanol is an example of dehydration reaction. The reaction is as follows:
CH3OH + H2SO4 → CH3 - O - CH3
Q2. Which one is formed when sodium phenoxide is heated with alkyl halide?
(A) Alkyl phenyl ether (B) Dimethyl ether
(C) Diphenyl ether (D) None of these
Answer: (A)
Solution: In carrying out a Williamson synthesis of ether:
- Less hindered alkyl group should be provided by the alkyl halide.
- More hindered alkyl group should come from the alkoxide.
To prepare alkyl phenyl ether, phenoxide ion is treated with alkyl halide.The reaction is as follows:

Q3. Reaction of tert-Butoxide ion with methyl bromide gives
(A)methyl tert- butyl ether
(B) tert-ethyl methyl ether
(C) tert-phenyl methyl ether
(D) None of the above
Answer: (A)
Solution: In carrying out a Williamson synthesis of ether:
- Less hindered alkyl group should be provided by the alkyl halide.
- More hindered alkyl group should come from the alkoxide.
Reaction of tert-Butoxide ion with methyl Bromide gives methyl tert-butyl ether. The reaction is as follows:

Q4. Which of the following reactants will give anisole?
(A) CH3CHO,RMgX
(B) C6H5OH, NaOH, CH3I
(C) C6H5OH, neutral FeCl3
(D) None of the above
Answer: (B)
Solution: Phenol on reaction with sodium hydroxide gives phenoxide ion and that phenoxide ion reacts with methyl iodide via williamson ether synthesis to give anisole. The reaction is as follows:
C6H5OH + CH3I + NaOH → C6H5 - O - CH3 + NaI + H2O
Frequently asked questions:
Question 1. Is ether is acidic or basic?
Answer: The ether acts like a Lewis base due to the presence of two lone pairs of electrons on the oxygen atom. Therefore, they form salts with strong acids.
The presence of two lone pairs of electrons on the oxygen atom causes the ether to behave like a Lewis base. As a result, they combine to produce salts with strong acids.
Question 2. Which method of preparing an unsymmetrical ether is the best, and why?
Answer: Williamson synthesis can be used to prepare unsymmetrical ethers because, in this method of preparation, alkyl halide and sodium alkoxides react to form ether. Depending on the type of unsymmetrical Ether to be prepared, the alkyl groups in both reactants can be selected.
Because alkyl halide and sodium alkoxides combine to form ether in this method of preparation, Williamson synthesis can be utilised to prepare unsymmetrical ethers. The alkyl groups in both reactants can be selected depending on the type of unsymmetrical Ether to be prepared.
CH3ONa + C2H5Br → CH3 - O - C2H5 + NaBr
Question 3. Which intermolecular forces act on the ether?
Answer: Putting the two ethyl ether molecules together, the opposite partial charges attract each other. This type of intermolecular force is called dipole-dipole interaction or dipole-dipole attraction because it occurs in polar molecules with dipoles.
Question 4. Is epoxide an ether?
Answer: Epoxide is a cyclic ether with a three-membered ring. The basic structure of an epoxide contains an oxygen atom attached to two adjacent carbon atoms of a hydrocarbon. The strain of the three-membered ring makes an epoxide much more reactive than a typical acyclic ether.
Related Topics:
|
Ethers-physical and chemical properties |
Ethanol |
|
Pinacol-pinacolone rearrangement |
Phenol |
|
Lucas test |
Methanol |


