-
Call Now
1800-102-2727
Chemical Properties of Ketones: Introduction, Chemical Properties of Ketones, Practice Problems & Frequently Asked Questions
Have you heard about the terms ketosis, ketonic meal plan etc? These are related to the intake of ketones as a supplement. The advantages are that it helps reduce carbohydrate-rich food, and changes the metabolism from a carbohydrate conversion to fat conversion. This is of help for reducing weight, heart disease, cancer cure and type-2 diabetes.

Ketones are also the most useful solvents in the chemical world.
Acetone is a component of many other beauty products in addition to being a key component in nail paint remover. In the beauty sector, it is often used as a denaturant and solvent. Acetone is a typical chemical
used in production, from wet wipes to hair colours.
After a home remodelling, acetone is useful for cleaning painting spots that have been splashed on glass panels. Additionally, it can be used to take care of the worn-in appearance that develops on leather shoes, purses, handbags, etc. after repeated use.
Spilled superglue can be removed from a surface more easily by dissolving it in acetone. Acetone can also be used to get stains out of some of the smallest spaces, including between the keys of a keyboard, with the use of a cotton swab.
Here you will know more about the properties of such organic molecules called ketones.
Table of Contents:
- Introduction of Ketones
- Chemical Properties of Ketones
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Ketones:
Ketones have a carbonyl group, as a functional group containing a double bond between carbon and oxygen. In ketones, the carbonyl group's shape is trigonal planar, and the carbon atom shows sp2 hybridizations. The carbon atom in the carbonyl group with two sp2 orbitals forms two sigma bonds with other carbon atoms in molecules. The third sp2 hybrid orbital establishes a bond with the oxygen atom. The unhybridized p orbital on the carbon atom in the carbonyl group overlaps a p orbital on the oxygen atom to create a π- bond of the double bond.
Chemical Properties of Ketones:
Chemical properties of ketones arise from the reactions of the attached alkyl groups or the ketonic functional group.
Reactions of the Ketonic Group:
A. Nucleophilic addition reactions:
A nucleophile is an electron-rich atom or group seeking electron-deficient or positively charged atoms or groups.
A nucleophile with many electrons targets an electrophile with few electrons in a nucleophilic addition reaction.
The carbonyl group is polar with carbon getting a partial positive charge and oxygen having a partial negative charge. Hence, the positively charged carbon acts as an electrophile. A nucleophile will attack this carbonyl carbon. The ketone being planar structured, the nucleophilic attack occurs perpendicular to the plane of the ketone.
As a result, the hybridization of the carbon atom changes from sp2 to sp3, leading to the formation of a tetrahedral alkoxide intermediate. An electrically neutral product is created when the intermediate generated takes a proton from the medium of the reaction. The entire procedure adds Nu- and H+ even with the carbon-oxygen double bond.
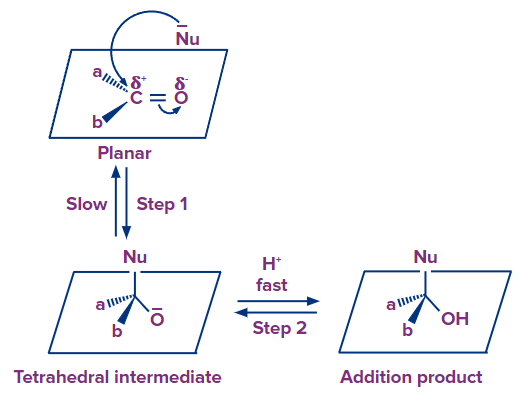
Aldehydes are usually more reactive than ketones in nucleophilic addition reactions due to electronic and steric reasons.
(i) Electronically, the electrophilicity of the carbonyl group is lessened by the two alkyl groups in ketones than by the single alkyl group in aldehydes.
(ii) Ketones have two very big groups that prevent the nucleophile from attacking the electrophilic carbonyl atom. Aldehydes do not display this steric barrier because they only contain one such substituent.
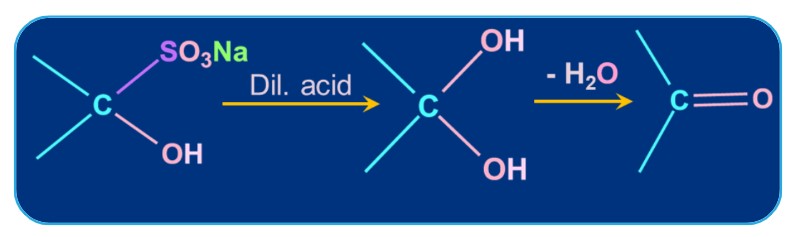
1. Addition of Sodium Hydrogen Sulphite:
Aldehydes and ketones, which are carbonyl compounds, react with sodium hydrogen sulphite to create the corresponding addition products. The process is irreversible, and the product is crystalline. For steric considerations, the equilibrium position for the most of ketones is significant to the left while that for aldehydes is large to the right.

Metal bisulphite reacts with aldehydes and ketones react to produce a water-soluble byproduct. The by-product can be converted back into the original carbonyl compound by acidifying with dilute alkali or mineral acids. For separating and purifying aldehydes, this reaction is most helpful.
2. Addition of Hydrogen Cyanide (HCN):
Ketones and hydrogen cyanide combine to form cyanohydrins, which are useful intermediates for the synthesis of other compounds. The cyanide ion (CN-) is produced through a base-catalysed reaction. By combining carbonyl compounds with the greater nucleophile cyanide ion (CN-), it is simple to create the corresponding cyanohydrin.

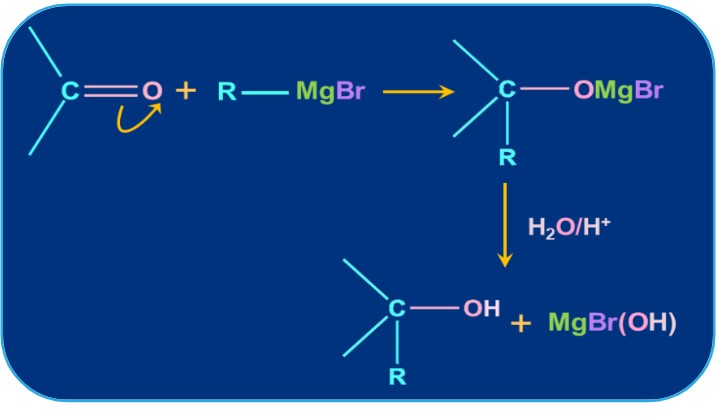
3. Addition of Grignard Reagent:
Tertiary alcohols are created when ketones and Grignard reagents interact. The reaction involved is a two-step process. Through a nucleophilic addition reaction, the Grignard reagent adds to the carbonyl group in the first phase of the reaction. As a result, an adduct forms. The second stage involves hydrolyzing the adduct to produce alcohol.
4. Addition of Alcohol:
When dry hydrogen chloride is present, a monohydric alcohol molecule interacts with one molecule of ketone to create an intermediate called alkoxy alcohol. This intermediate, known as hemiketals, combines with one more alcohol molecule to form the gem-alkoxy compound ketal.

Ethylene glycol ketals are cyclic compounds that are created when ketone and ethylene glycol combine in the vicinity of dry hydrogen chloride.

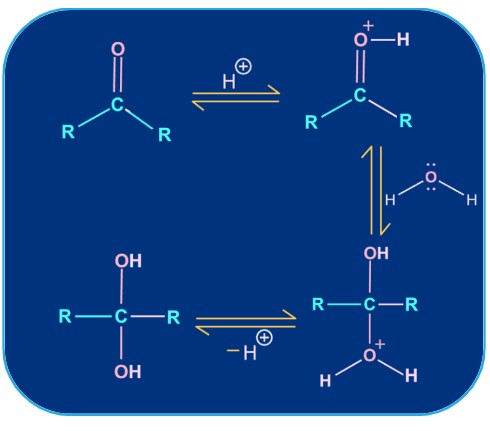
5. Addition of water:
Gem diols are produced when water and ketones react. Since water is a weak nucleophile, the carbonyl group is added rather slowly. However, the rate of the reaction can be accelerated with an acid catalyst. The equilibrium of the reversible water reaction tends to shift in the direction of >C=O.
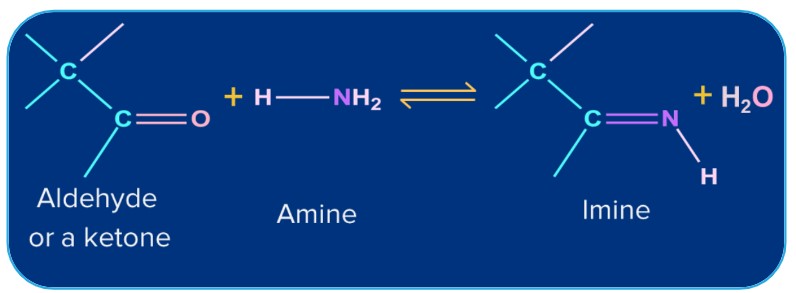
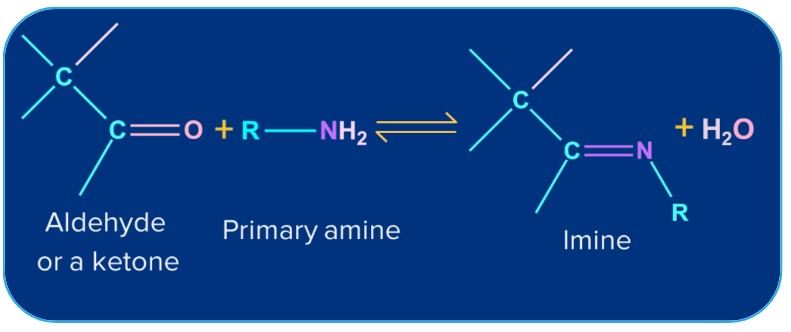
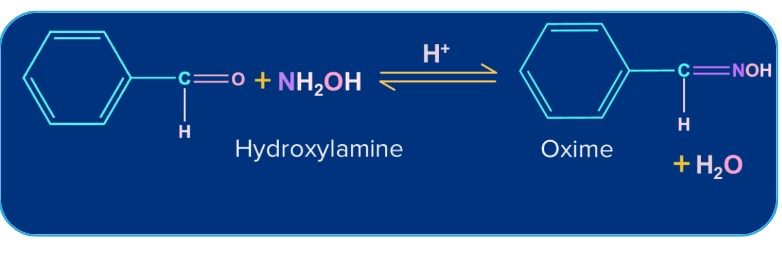
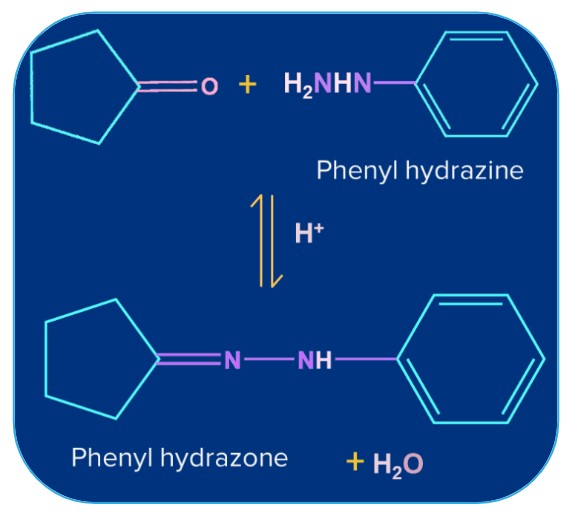
6. Addition of Ammonia and its Derivatives:
Ammonia and its derivatives H2N-Z (Z= Alkyl, Aryl, OH, C6H5NH,NHCONH2 etc.) combine with the carbonyl group of ketones. It is a reversible acid-catalysed process. Because of the quick dehydration of the intermediate to form >C=N-Z, the equilibrium supports the formation of the product.

|
If Z is |
Reagent Name |
Products |
|
-H |
Ammonia |
Imine |
|
If Z is |
Reagent Name |
Products |
|
Alkyl group (-R) |
Primary amine |
Substituted imine(Schiff’s Base) |
|
If Z is |
Reagent Name |
Products |
|
-OH |
Hydroxylamine |
Oxime |
|
If Z is |
Reagent Name |
Products |
|
-NH2 |
Hydrazine |
Hydrazone |

|
If Z is |
Reagent Name |
Products |
|
Phenyl Hydrazine |
Phenyl Hydrazone |
|
If Z is |
Reagent Name |
Products |
|
2,4- Dinitro Phenyl Hydrazine (or) Brady’s Reagent (or) 2,4-DNP |
2,4- Dinitro Phenyl Hydrazone (orange precipitate) |
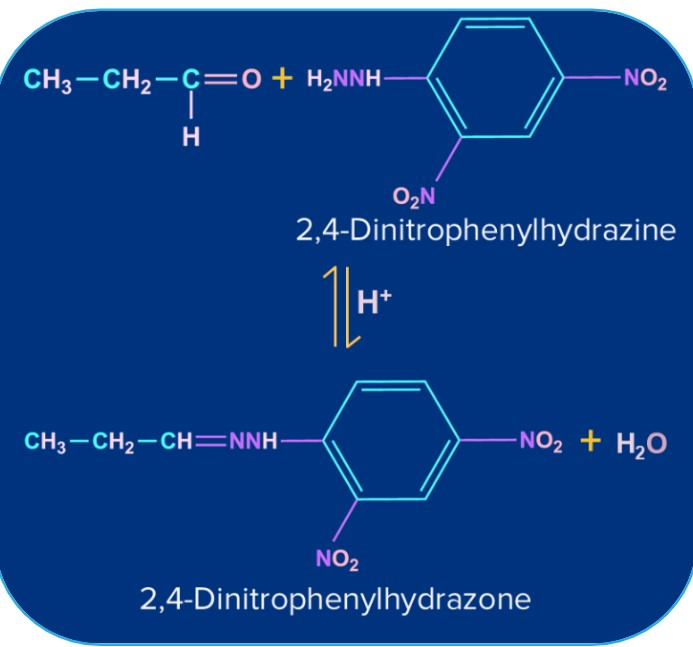
|
If Z is |
Reagent Name |
Products |
|
semicarbazide |
Semicarbazone |
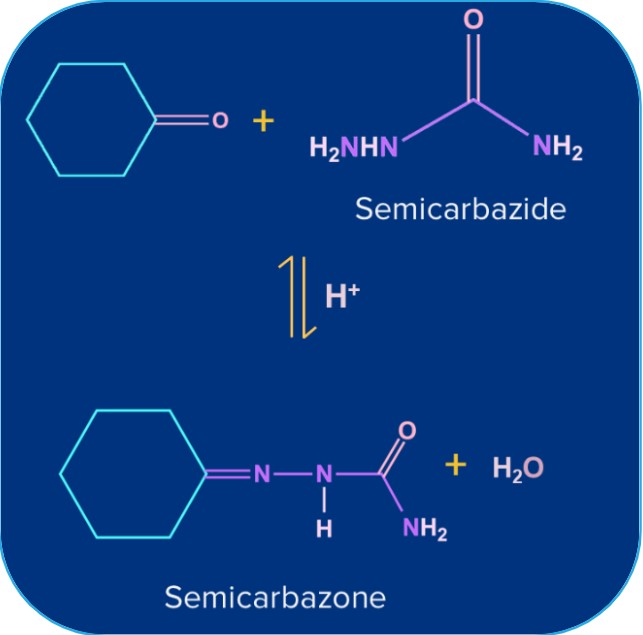

B. Reduction to Alcohols:
Ketones are converted to their corresponding alcohols via catalytic hydrogenation. Catalysts such as palladium, platinum or nickel are employed in this method to add hydrogen. This reaction also happens in the presence of lithium aluminium hydride (LiAlH4) and sodium borohydride (NaBH4). Secondary alcohols are produced by ketones, whereas primary alcohols are produced by aldehydes.

C. Reduction to Hydrocarbons:
(i) Wolff-Kishner Reaction:
The Wolf-Kishner reduction process changes the carbonyl groups on ketones into -CH2 groups. Carbonyl compounds are treated with hydrazine and sodium or potassium hydroxide and subsequently heated in a solvent with a higher boiling point, such as ethylene glycol.
(ii) Clemmensen Reduction:
The carbonyl group of ketones is changed to the -CH2 group using the Clemmensen reduction procedure, which uses zinc amalgam and strong hydrochloric acid (HCl).
D. Oxidation Reaction:
In order to perform oxidation processes, ketones often need strong oxidising reagents and high temperatures. They undergo carbon-carbon bond cleavage during oxidation, resulting in a variety of carboxylic acids with reduced carbon atoms than the parent ketone.
E. Aldol Condensation:
Aldehyde and ketone "hydrogens" refer to the - hydrogen atom that is located on the carbon atom next to the carbonyl carbon. Aldehydes and ketones include - hydrogen atoms that are acidic by nature and engage in a variety of chemical reactions. The acidity of the - hydrogen atoms in carbonyl compounds is explained by the strong electron-withdrawing impact of the carbonyl group and resonance stabilisation of the conjugate base.
As a catalyst, dilute alkali causes a reaction to occur between carbonyl molecules like aldehydes and ketones that include at least one - hydrogen. This process is referred to as the "aldol condensation reaction" and produces either - hydroxy aldehydes (aldol) or - hydroxy ketones (ketol).
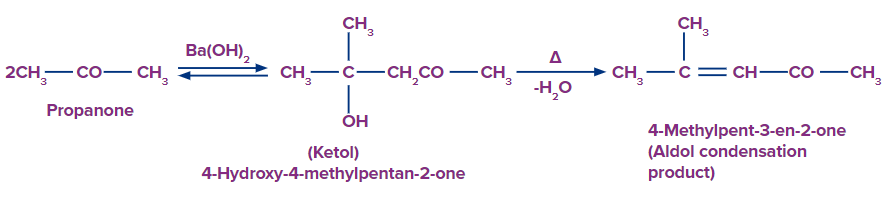
F. Haloform Reaction:
Ketones that have at least one methyl group attached to the carbonyl atom are known as methyl ketones. Sodium hypohalite converts these methyl ketones into sodium salts of the corresponding carboxylic acids. There is one reduced carbon atom in these carboxylic acids than in the original carbonyl molecule. Haloform is created by the methyl group. If the molecule contains a carbon-carbon double bond, it is unaffected by this oxidation.
The detection of CH3CO groups or CH3CH(OH) groups, which oxidise to CH3CO groups, is also done using the iodoform reaction with sodium hypoiodite.
The reaction is considered a chloroform reaction if chlorine is the halogen employed. The chloroform reaction example is shown below:
The reaction is considered a bromoform reaction if bromine is the halogen employed. The bromoform reaction example is shown below:
The reaction is considered an iodoform reaction if iodine is the halogen employed. The iodoform reaction example is shown below:
G. Aromatic Electrophilic Substitution Reaction:
In the ring of aromatic aldehydes and ketones, where electrophilic substitution takes place, the carbonyl group functions as a deactivating and meta-directing group.
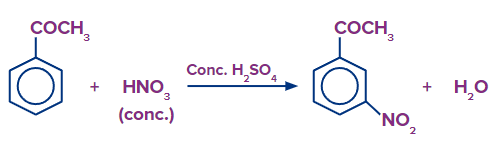
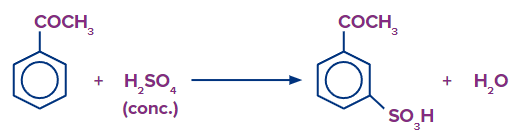
H. Sodium Nitroprusside Test:
The existence of a ketonic (> C=O) group is demonstrated by the appearance of red colour in the solution when a newly produced sodium nitroprusside solution is added to a ketone. The mixture is agitated thoroughly and basified by adding sodium hydroxide solution drop by drop.
The nitroprusside ion and the anion of ketone produced by alkali combine to form a complex that is red in colour and shows the presence of the ketonic group.
Practice Problems:
Q1. Which of the following reacts the least favourably to a nucleophilic attack?
(A) Methanal
(B) Ethanal
(C) Acetone
(D) Di Tert-butyl ketone
Answer: (D)
Solution: When analysing these substances, the steric effects are taken into account. Two very large tert-butyl groups, one on either side of the carbonyl carbon, are present in ditert-butyl ketone. This reduces reactivity and increases the barrier to the approaching nucleophile.
Q2. Determine the catalyst for the nucleophilic addition of hydrogen cyanide (HCN)to acetone.
(A) Sodium hydroxide
(B) Hydrochloric acid
(C) Sodium cyanide
(D) Sodium chloride
Answer: (A)
Solution: A cyanohydrin of acetone is produced when acetone and hydrogen cyanide (HCN) react. With pure hydrogen cyanide, (HCN) however, this reaction moves along very slowly. As a result, the reaction is carried out in a basic medium, which causes hydrogen cyanide (HCN) to lose its proton and generate the CN- ion, a stronger nucleophile and makes the reaction faster.
Q3. Propan-2-ol is produced by catalytic hydrogenation of -
(A) Acetone
(B) Acetaldehyde
(C) formaldehyde
(D) None of the above
Answer: (A)
Solution: Acetone is reduced to propan-2-ol, secondary alcohol, in the catalyst presence like nickel (Ni), Platinum (Pt) or Palladium (Pd). Lithium aluminium hydride (LiAlH4) and Sodium borohydride (NaBH4) can also reduce it through a chemical reaction. Aldehydes yield primary alcohols in a similar reaction.
Q4. Which of the following substances can be identified from the iodoform test?
(A) Acetaldehyde and acetone
(B) Acetophenone and benzaldehyde
(C) Acetophenone and benzophenone
(D) Benzophenone and benzaldehyde
Answer: (C)
Solution: To determine whether a substance contains a CH3CO or CH3CH(OH) group, an iodoform test involves reacting the molecule with sodium hypoiodite. A positive test results in the formation of a yellow precipitate called CHI3 (iodoform). Formaldehyde (HCHO), benzophenone (Ph(CO)Ph), and benzaldehyde (Ph-CHO) do not undergo this test because they do not contain a CH3CO group in their compounds. Acetone (CH3COCH3) Acetaldehyde (CH3-CHO) and acetophenone (Ph(CO)CH3) produce yellow ppt in this test.
Frequently Asked Questions(FAQs):
Q1. Why don't ketones react with the tollens reagent?
Answer: Aldehydes and ketones are distinguished from one another using reagents, one of which being Tollen's reagent. An oxidation reaction occurs when aldehydes are mixed with Tollen's reagent. Ketones are difficult to oxidise, whereas aldehydes are oxidised easily.
Ammoniacal silver nitrate solution is the Tollen's reagent. With this reagent, silver ions are converted to metallic silver when an aldehyde is treated, and a brilliant silver mirror is created on the inside of the test tube. You need an alkaline medium for this oxidation.
An aldehyde molecule (R-CHO) possesses an H-atom, which makes it easy for them to oxidise, but ketones (RCOR') lack an H-atom and are hence resistant to oxidation. Only very powerful oxidising agents, such as potassium permanganate, may oxidise ketones.
Q2. Why is benzaldehyde less reactive than para-nitro benzaldehyde in nucleophilic addition reactions?
Answer: The nitro (NO2) group is a potent electron-withdrawing group in p-nitrobenzaldehyde. By using both inductive and resonance effects, it pulls away electrons, lowering the density of electrons surrounding the carboxyl group's carbon atom. This makes it easier for the nucleophile to attack it, making it more reactive than benzaldehyde.
Q3. What makes acetone such an excellent solvent?
Answer: In the industry, acetone is an excellent solvent. Acetone, for example, dissolves both polar and nonpolar pigments, making it the preferred solvent for chromatography. It is crucial to understand that when acetone dissolves in water, its molecules totally reunite to produce a heterogeneous mixture rather than splitting off into ions. The acetone molecule contains two non-polar methyl groups that dissolve non-polar compounds, but the carbonyl group in the middle of the molecule is polar and dissolves polar compounds.
Q4. Why do ketones oxidise only in the presence of potent oxidising agents like potassium permanganate (KMnO4)?
Answer: Ketones are resistant to oxidation because they do not have a hydrogen atom connected to their carbonyl carbon. Ketones can only be oxidised by extremely potent oxidising agents like potassium permanganate solution. With cleavage, however, this strong oxidation takes place, rupturing carbon-carbon bonds to produce two carboxylic acids. This reaction is rarely used due to its destructive effects.


